GILEAD SCIENCES 885823 (Seite 315)
eröffnet am 20.02.06 13:18:36 von
neuester Beitrag 13.02.24 21:17:08 von
neuester Beitrag 13.02.24 21:17:08 von
Beiträge: 3.632
ID: 1.041.671
ID: 1.041.671
Aufrufe heute: 9
Gesamt: 481.690
Gesamt: 481.690
Aktive User: 0
ISIN: US3755581036 · WKN: 885823 · Symbol: GILD
60,80
EUR
-3,03 %
-1,90 EUR
Letzter Kurs 21:19:41 Tradegate
Neuigkeiten
| Gilead Sciences Aktien ab 5,80 Euro handeln - Ohne versteckte Kosten!Anzeige |
24.04.24 · Shareribs Anzeige |
22.04.24 · Shareribs Anzeige |
18.04.24 · Shareribs Anzeige |
16.04.24 · Shareribs Anzeige |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 0,7300 | +25,11 | |
| 6,0000 | +25,00 | |
| 9,5000 | +18,75 | |
| 0,6400 | +18,52 | |
| 1,0900 | +14,74 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 0,8410 | -17,06 | |
| 9,7200 | -19,60 | |
| 4,0000 | -27,27 | |
| 2,7280 | -29,14 | |
| 14,510 | -32,32 |
Beitrag zu dieser Diskussion schreiben
Zitat von dax5000: Was ist heute mit Gilead los ? trotz possitiven Ausblich von Moodys
http://www.onvista.de/news/gilead-bepreist-vorrangig-ungesic…
Was ist heute mit Gilead los ? trotz possitiven Ausblich von Moodys
gilead ist so geil 


nichts kann diesen trendfolger stoppen
Rating Action:
Moody's rates Gilead's notes Baa1; positive outlook
https://www.moodys.com/research/Moodys-rates-Gileads-notes-B…



nichts kann diesen trendfolger stoppen
Rating Action:
Moody's rates Gilead's notes Baa1; positive outlook
https://www.moodys.com/research/Moodys-rates-Gileads-notes-B…
3 Things to Watch at Gilead
http://www.fool.com/investing/general/2014/02/24/gilead-gild…
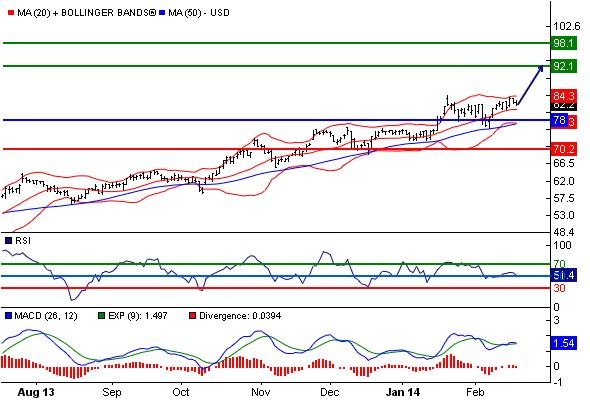
Pre Market grün zZ.83.79$ Wiederstand bei 92.1
Oberkassler
http://www.fool.com/investing/general/2014/02/24/gilead-gild…
Pre Market grün zZ.83.79$ Wiederstand bei 92.1
Oberkassler
Gilead's Hepatitis C Pill Takes Off Like A Rocket
http://www.forbes.com/sites/matthewherper/2014/02/21/gileads…
Heute Long Signal.
Unser Pivot-Punkt liegt bei 78.
Unsere Meinung: solange die Unterstützung bei 78 hält, ist die Aufwärtstendenz intakt.
Alternatives Szenario: unter 78 lauten die Kursziele 73.1 und 70.2
Analyse: der RSI liegt über der Neutralitätszone von 50. Der MACD liegt über der Signallinie und ist positiv. Die Konfiguration ist positiv. Es wird darauf hingewiesen, dass die Volumina seit einigen Tagen fallen.
Source:CC
Oberkassler
http://www.forbes.com/sites/matthewherper/2014/02/21/gileads…
Heute Long Signal.
Unser Pivot-Punkt liegt bei 78.
Unsere Meinung: solange die Unterstützung bei 78 hält, ist die Aufwärtstendenz intakt.
Alternatives Szenario: unter 78 lauten die Kursziele 73.1 und 70.2
Analyse: der RSI liegt über der Neutralitätszone von 50. Der MACD liegt über der Signallinie und ist positiv. Die Konfiguration ist positiv. Es wird darauf hingewiesen, dass die Volumina seit einigen Tagen fallen.
Source:CC

Oberkassler
CURx Pharmaceuticals Announces License Agreement with Gilead Sciences
CURx Pharmaceuticals today announced it has entered into a global license agreement with Gilead Sciences, Inc. for development of Fosfomycin:Tobramycin for Inhalation (FTI) to treat Pseudomonas aeruginosa lung infection in cystic fibrosis (CF) patients. FTI is ready for Phase III clinical trials, having successfully completed a Phase II trial in CF patients.
"This is a great acquisition for CURx Pharmaceuticals which enables us to expand our portfolio and also to provide another inhaled therapeutic to the cystic fibrosis community", commented Dinu Sen, CEO and founder of CURx. "Clinical trials in CF patients with Pseudomonas aeruginosa infection have demonstrated FTI's safety and efficacy. Future trials may pave the way for its use against a range of bacterial infections in CF - there is an unmet need for antibiotics with such characteristics."
"Lung infections continue to be a serious problem for individuals with cystic fibrosis," commented Dr. Drucy Borowitz, Director of the Cystic Fibrosis Center of Western New York. "If further studies confirm that (FTI) is safe and efficacious it will be a significant addition to improving the health of people with CF." Dr. Patrick Flume, Director of the Cystic Fibrosis Center at Medical University of South Carolina added "Currently we have two drugs approved in the US for use in CF patients, but there remains a need for more options. The CF community welcomes the development of new treatment options such as FTI."
About FTI
FTI is a combination of fosfomycin and tobramycin, antibiotics well characterized for safety and anti-bacterial activity. FTI is formulated as a liquid for inhalation with an aerosol device. In Phase II clinical trials FTI was shown to be safe and to effectively maintain improvement in lung function achieved with Cayston in CF patients with Pseudomonas aeruginosa infection. FTI has demonstrated antibiotic activity against multiple pathogenic bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), in preclinical studies.
About cystic fibrosis
Cystic fibrosis is a hereditary disease affecting the lungs and digestive system of 30,000 children and adults in the US and 70,000 worldwide. Chronic bacterial infections in the lungs of CF patients are common and can be life threatening if not adequately treated. Pseudomonas aeruginosa infects the lungs of half of all CF patients, while methicillin-resistant Staphylococcus aureus (MRSA) infects the lungs of about a quarter of CF patients.
About CURx Pharmaceuticals
San Diego-based CURx Pharmaceuticals specializes in developing late-stage therapeutics with high medical need. In August 2013 CURx Pharmaceuticals licensed intravenous topiramate from Ligand Pharmaceuticals, Inc. for treatment of hospitalized epilepsy patients unable to take oral medication. Please visit www.curxpharma.com for more information.
Forward-Looking Statements
This news release contains forward-looking statements. These statements are subject to known and unknown risks and uncertainties that may cause actual future experience and results to differ materially from the statements made. Factors that might cause such a difference include, among others, the completion of clinical trials, the FDA and other foreign review processes and other governmental regulation, CURx Pharmaceuticals' ability to successfully develop and commercialize drug candidates, competition from other pharmaceutical companies, the ability to effectively market products, and other factors. CURx Pharmaceuticals undertakes no duty to update forward-looking statements.
SOURCE CURx Pharmaceuticals
Oberkassler
CURx Pharmaceuticals today announced it has entered into a global license agreement with Gilead Sciences, Inc. for development of Fosfomycin:Tobramycin for Inhalation (FTI) to treat Pseudomonas aeruginosa lung infection in cystic fibrosis (CF) patients. FTI is ready for Phase III clinical trials, having successfully completed a Phase II trial in CF patients.
"This is a great acquisition for CURx Pharmaceuticals which enables us to expand our portfolio and also to provide another inhaled therapeutic to the cystic fibrosis community", commented Dinu Sen, CEO and founder of CURx. "Clinical trials in CF patients with Pseudomonas aeruginosa infection have demonstrated FTI's safety and efficacy. Future trials may pave the way for its use against a range of bacterial infections in CF - there is an unmet need for antibiotics with such characteristics."
"Lung infections continue to be a serious problem for individuals with cystic fibrosis," commented Dr. Drucy Borowitz, Director of the Cystic Fibrosis Center of Western New York. "If further studies confirm that (FTI) is safe and efficacious it will be a significant addition to improving the health of people with CF." Dr. Patrick Flume, Director of the Cystic Fibrosis Center at Medical University of South Carolina added "Currently we have two drugs approved in the US for use in CF patients, but there remains a need for more options. The CF community welcomes the development of new treatment options such as FTI."
About FTI
FTI is a combination of fosfomycin and tobramycin, antibiotics well characterized for safety and anti-bacterial activity. FTI is formulated as a liquid for inhalation with an aerosol device. In Phase II clinical trials FTI was shown to be safe and to effectively maintain improvement in lung function achieved with Cayston in CF patients with Pseudomonas aeruginosa infection. FTI has demonstrated antibiotic activity against multiple pathogenic bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), in preclinical studies.
About cystic fibrosis
Cystic fibrosis is a hereditary disease affecting the lungs and digestive system of 30,000 children and adults in the US and 70,000 worldwide. Chronic bacterial infections in the lungs of CF patients are common and can be life threatening if not adequately treated. Pseudomonas aeruginosa infects the lungs of half of all CF patients, while methicillin-resistant Staphylococcus aureus (MRSA) infects the lungs of about a quarter of CF patients.
About CURx Pharmaceuticals
San Diego-based CURx Pharmaceuticals specializes in developing late-stage therapeutics with high medical need. In August 2013 CURx Pharmaceuticals licensed intravenous topiramate from Ligand Pharmaceuticals, Inc. for treatment of hospitalized epilepsy patients unable to take oral medication. Please visit www.curxpharma.com for more information.
Forward-Looking Statements
This news release contains forward-looking statements. These statements are subject to known and unknown risks and uncertainties that may cause actual future experience and results to differ materially from the statements made. Factors that might cause such a difference include, among others, the completion of clinical trials, the FDA and other foreign review processes and other governmental regulation, CURx Pharmaceuticals' ability to successfully develop and commercialize drug candidates, competition from other pharmaceutical companies, the ability to effectively market products, and other factors. CURx Pharmaceuticals undertakes no duty to update forward-looking statements.
SOURCE CURx Pharmaceuticals
Oberkassler
Next Resistance Level for Gilead Sciences (GILD) is $83.78
http://www.mysmartrend.com/news-briefs/news-watch/next-resis…
Gilead Sciences: A Great Buy Despite Competition From AbbVie
http://seekingalpha.com/article/2027821-gilead-sciences-a-gr…
Unser Pivot-Punkt liegt bei 86.7.
Unsere Meinung: solange 86.7 einen Widerstand bildet, sind 73.1 das Ziel
Alternatives Szenario: über 86.7 liegen die nächsten Ziele bei 91.6 und 94.5
Analyse: der RSI liegt unter der Neutralitätszone von 50. Der MACD liegt unter der Signallinie und ist positiv. Der MACD muss die Null-Linie überwinden, um weiteres Abwärtspotenzial erwarten zu können. Es wird darauf hingewiesen, dass die Volumina seit einigen Tagen ansteigen.
Source:CC
Oberkassler
http://www.mysmartrend.com/news-briefs/news-watch/next-resis…
Gilead Sciences: A Great Buy Despite Competition From AbbVie
http://seekingalpha.com/article/2027821-gilead-sciences-a-gr…
Unser Pivot-Punkt liegt bei 86.7.
Unsere Meinung: solange 86.7 einen Widerstand bildet, sind 73.1 das Ziel
Alternatives Szenario: über 86.7 liegen die nächsten Ziele bei 91.6 und 94.5
Analyse: der RSI liegt unter der Neutralitätszone von 50. Der MACD liegt unter der Signallinie und ist positiv. Der MACD muss die Null-Linie überwinden, um weiteres Abwärtspotenzial erwarten zu können. Es wird darauf hingewiesen, dass die Volumina seit einigen Tagen ansteigen.
Source:CC
Oberkassler
The Hidden Gems in Gilead Sciences' Pipeline
http://www.fool.com/investing/general/2014/02/13/the-hidden-…
Ein sehr interessanter Artikel zu den Wettbewerbern zu GILD - und deren Präperate, Indikationen, Umsatz, Stati und Analysen.
Oberkassler
http://www.fool.com/investing/general/2014/02/13/the-hidden-…
Ein sehr interessanter Artikel zu den Wettbewerbern zu GILD - und deren Präperate, Indikationen, Umsatz, Stati und Analysen.
Oberkassler
Mahlzeit, bin seid paar Minuten dabei.
GILEAD SCIENCES 885823





















