Amarin - The Science Of Lipid Therapy (Seite 117)
eröffnet am 03.01.14 20:10:32 von
neuester Beitrag 04.04.24 15:47:54 von
neuester Beitrag 04.04.24 15:47:54 von
Beiträge: 1.840
ID: 1.190.027
ID: 1.190.027
Aufrufe heute: 0
Gesamt: 156.359
Gesamt: 156.359
Aktive User: 0
ISIN: US0231112063 · WKN: A0NBNG · Symbol: EH3A
0,8657
USD
+0,74 %
+0,0064 USD
Letzter Kurs 02:00:00 Nasdaq
Neuigkeiten
22.04.24 · globenewswire |
15.04.24 · globenewswire |
Amarin Highlights Key Data Providing Mechanistic Insights into Eicosapentaenoic Acid (EPA) at ACC.24 08.04.24 · globenewswire |
06.04.24 · globenewswire |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 3,5800 | +922,86 | |
| 0,8800 | +95,56 | |
| 29,10 | +21,25 | |
| 0,9650 | +14,88 | |
| 7,9900 | +14,14 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 14,510 | -32,32 | |
| 1,3700 | -32,84 | |
| 1,4100 | -32,86 | |
| 3,6400 | -38,62 | |
| 0,7000 | -61,85 |
Beitrag zu dieser Diskussion schreiben
Antwort auf Beitrag Nr.: 61.933.355 von bernie55 am 15.11.19 14:40:50
Ich denke das Amarin natürlich weiss das sie ihr Marktpotential nur dann umfangreich nutzen können wenn sie eine Partnerschafr mit BP eingehen. Es ist wichtig in möglichst kurzer Zeit einen grossen Fussabdruck im Markt zu hinterlassen sichert höhere Einnahmen über Jahre. Ausserdem brauchen sie einen grossen Partner für Produktion usw. jedoch wie gesagt Buyout sehe ich nicht, Amarin kann so die Zügel in der Hand behalten und hat eine ideale Verhandlungsposition. Denke das auch die Nachricht einer Partnerschaft in Kombination mit hoffentlich full label expansion nochmal ordentlich Dampf macht. Sehe im ersten Quartal nächsten Jahres solch eine Entscheidung als realistisch. Sie müssen nichts überstürzen und können ideal verhandeln...
Zitat von bernie55:Zitat von flyingbeef: ...
Wenn ich mich richtig erinnere:
Amarin hat aber immer gesagt Übernahme "nicht mit uns" daher ich glaube die wollen die Zügel auch langfristig in der Hand haben sollte trotzdem ordentlich nach oben rauschen.
Sollte es zu einem BuyOut-Bidding von verschiedensten BPs ( Pfizer ? Amgen ? xxxx ? etc. ) kommen und ein sehr hohes Buyoutangebot vorliegen , dann würde ich das " nicht mit uns " nicht mehr unterschreiben.
Ich denke das Amarin natürlich weiss das sie ihr Marktpotential nur dann umfangreich nutzen können wenn sie eine Partnerschafr mit BP eingehen. Es ist wichtig in möglichst kurzer Zeit einen grossen Fussabdruck im Markt zu hinterlassen sichert höhere Einnahmen über Jahre. Ausserdem brauchen sie einen grossen Partner für Produktion usw. jedoch wie gesagt Buyout sehe ich nicht, Amarin kann so die Zügel in der Hand behalten und hat eine ideale Verhandlungsposition. Denke das auch die Nachricht einer Partnerschaft in Kombination mit hoffentlich full label expansion nochmal ordentlich Dampf macht. Sehe im ersten Quartal nächsten Jahres solch eine Entscheidung als realistisch. Sie müssen nichts überstürzen und können ideal verhandeln...
Ich sitze hier mit Popcorn echt wahr 

Sorry war jetzt nicht sehr informativ aber an solchen Tagen darf man auch mal etwas albern sein 🙃


Sorry war jetzt nicht sehr informativ aber an solchen Tagen darf man auch mal etwas albern sein 🙃
Antwort auf Beitrag Nr.: 61.935.875 von Magnetfeldfredy am 15.11.19 18:43:48Hier die ScriptZahlen von Sam81:
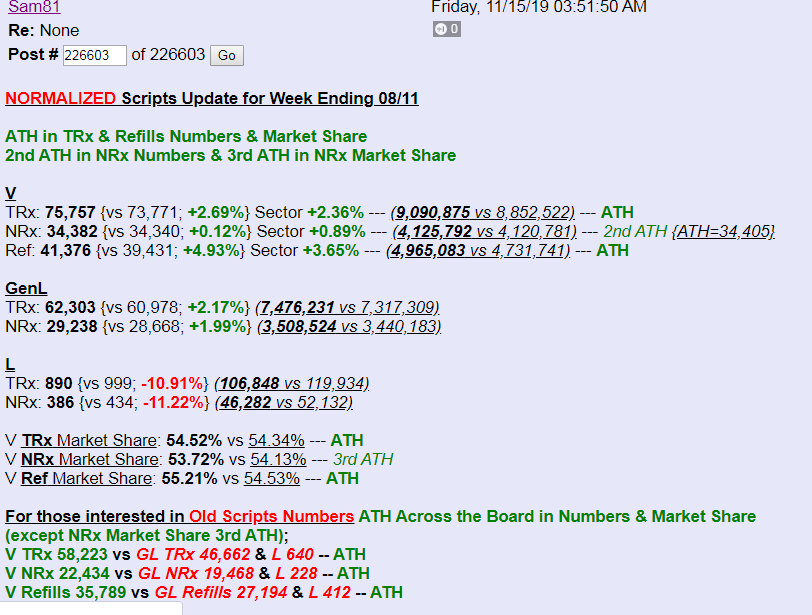
Antwort auf Beitrag Nr.: 61.935.875 von Magnetfeldfredy am 15.11.19 18:43:48Gute Zusammenfassung, ich denke auch, dass wir mit der gestrigen Entscheidung stetig steigen werden. Es wird auch zu Rücksetzern kommen, gerade wenn der Gesamtmarkt in die Knie geht, aber die Richtung ist jetzt klar aufwärts
Meinung von "Sam" aus Investorshub, toller Mensch, der jeden Freitag die scripts meldet:
Sam81 Friday, 11/15/19 04:19:07 AM
Re: None 0
Post # of 226871
Dear all,
I wasn't able to post last night after i reached my limit.
Congrats to all
Anyhow, what a day, up and down with all discussions but finally science prevailed and we got our 16-0 vote
In addition with regard to primary indication, i would say panelists were 10 for - 4 against & 2 undecided (10-4-2) and that bodes well for us.
Personally, and after watching and listening to all panelists after the vote, i would say we will get a label for both primary & secondary, TG 150 and above, 40 Years onward & of course diabetic and with one or more CVD risk factors.
And as with statins, the prerequisites with time will be loosened after Real World Data & follow up
Finally, and all my personal opinion, i am strongly against a BO. Why burn your cards and miss opportunities for your shareholders by taking a premium NOW of 100% - 200% (based on Wednesday close of $21.49) when in the future (2021-2022) your company will be generating billions if not tens of billions of income and your valuation will be at the level on its own and then you can decide to get a premium on top of that or remain an independent big firm just like others.
Congrats to all once again and let's enjoy 2020 and more💰
Sam81 Friday, 11/15/19 04:19:07 AM
Re: None 0
Post # of 226871
Dear all,
I wasn't able to post last night after i reached my limit.
Congrats to all
Anyhow, what a day, up and down with all discussions but finally science prevailed and we got our 16-0 vote

In addition with regard to primary indication, i would say panelists were 10 for - 4 against & 2 undecided (10-4-2) and that bodes well for us.

Personally, and after watching and listening to all panelists after the vote, i would say we will get a label for both primary & secondary, TG 150 and above, 40 Years onward & of course diabetic and with one or more CVD risk factors.
And as with statins, the prerequisites with time will be loosened after Real World Data & follow up
Finally, and all my personal opinion, i am strongly against a BO. Why burn your cards and miss opportunities for your shareholders by taking a premium NOW of 100% - 200% (based on Wednesday close of $21.49) when in the future (2021-2022) your company will be generating billions if not tens of billions of income and your valuation will be at the level on its own and then you can decide to get a premium on top of that or remain an independent big firm just like others.
Congrats to all once again and let's enjoy 2020 and more💰
(Bloomberg) -- The unanimous panel backing to expand the label for Amarin Corp.’s heart drug Vascepa sparked debate across Wall Street as analysts and investors weighed whether the drug could be sold to as many as 15 million Americans.
The divide comes after members of an advisory panel to the U.S. Food and Drug Administration “offered different opinions when it came to specific recommendations for a potential Vascepa label,” according to analysts at Roth Capital Partners. That may leave billions in sales hanging in the balance. But Jefferies’ Michael Yee disagreed, saying the “label debate doesn’t really matter to the thesis” for investors. He believes doctors will use the drug “widely across patients.”
Amarin shares climbed as much as 11% Friday after being halted all day Thursday. With the stock briefly nearing $24 a share, it traded about a nickel short of a July peak.
Here’s what analysts had to say about the news.
Roth Capital Partners, Yasmeen Rahimi
“With unanimously favorable response for label expansion, question moves to which patients make it on label, with 10/16 docs voting in favor of both” atherosclerotic cardiovascular disease groups. Roth highlighted that some committee members “were caught up with analysis that showed a 12% relative CV risk reduction” without significant benefit for patients who had not had something like a heart attack or stroke and are at a lower risk.
Counted 10 of the 16 panelists “who were receptive to Vascepa label expansion including primary and secondary prevention patients” with the six others wanting the drug’s label to include patients who have had a stroke, or have cardiovascular disease.
Says there was “lots of safety talk, but vote indicated AdCom members felt the two signals could be adequately managed through label, thus meaning no patient restrictions.”
Maintains buy rating with $31 price target.
Jefferies, Michael Yee
“Our thesis and valuation are not based on specific ‘details’ of a label as we think docs will use this widely across patients...if stock was down, we think it bounces since label details don’t matter in big picture.”
Notes that while Amarin got the positive vote for approval, “investors will debate whether the labeled indication will include a more broad ‘primary prevention’ (high-risk) population which is patients w/o established CV disease but have diabetes and at least one other major” cardiovascular risk factor.
Highlights that the “stock has already run-up a lot and we wouldn’t be surprised if it’s volatile on this labeling discussion but we aren’t changing numbers as the populations are huge already.”
Maintains buy rating, $30 price target.
Cantor Fitzgerald, Louise Chen
“There were few-to-no surprises brought up during the meeting, in our view, and we think Vascepa is well on-track now for expected approval by year-end.”
Notes that most of the meeting “focused on concerns about the impact of mineral oil, as well as on which patient populations should be included in the label. Although some AdCom members believe mineral oil may have affected results somewhat, they agreed that the REDUCE-IT data are meaningful enough to warrant approval.”
Highlighted that panelist discussions on how high patients’ triglycerides, a type of fat in the blood, must be to benefit were “less clear-cut compared to the mineral oil debate.”
Maintains overweight rating with a $35 price target.
Citi, Joel Beatty
Amarin is unlikely to reach a market value above $10 billion over the next several months with its market value at $7.7 billion as of Wednesday’s close.
Sees the drug as “likely to be used in high-risk primary prevention patients regardless of” the exact label wording, though there will likely be some debate over the coming weeks regarding the label.
“We believe the exact wording of the indication matters relatively little compared to other drugs, because we believe physicians will still use Vascepa in very high risk” primary prevention patients.
Maintains buy rating and $23 price target.
Stifel, Derek Archila
“While the panel was in agreement Vascepa should be approved for secondary prevention patients (our base case), the key question that remains is whether or not Vascepa will receive a broader label inclusive of primary prevention patients, which would be upside to our model.”
“We believe approval in secondary prevention alone offers an addressable market that is at least ~10-15 million patients for Vascepa, making us comfortable with our ~$3 billion in peak sales estimate.”
Rates shares at buy with a $26 price target.
(Updates share movement in third paragraph.)
To contact the reporter on this story: Bailey Lipschultz in New York at blipschultz@bloomberg.net
To contact the editors responsible for this story: Catherine Larkin at clarkin4@bloomberg.net, Scott Schnipper, Richard Richtmyer
For more articles like this, please visit us at bloomberg.com
The divide comes after members of an advisory panel to the U.S. Food and Drug Administration “offered different opinions when it came to specific recommendations for a potential Vascepa label,” according to analysts at Roth Capital Partners. That may leave billions in sales hanging in the balance. But Jefferies’ Michael Yee disagreed, saying the “label debate doesn’t really matter to the thesis” for investors. He believes doctors will use the drug “widely across patients.”
Amarin shares climbed as much as 11% Friday after being halted all day Thursday. With the stock briefly nearing $24 a share, it traded about a nickel short of a July peak.
Here’s what analysts had to say about the news.
Roth Capital Partners, Yasmeen Rahimi
“With unanimously favorable response for label expansion, question moves to which patients make it on label, with 10/16 docs voting in favor of both” atherosclerotic cardiovascular disease groups. Roth highlighted that some committee members “were caught up with analysis that showed a 12% relative CV risk reduction” without significant benefit for patients who had not had something like a heart attack or stroke and are at a lower risk.
Counted 10 of the 16 panelists “who were receptive to Vascepa label expansion including primary and secondary prevention patients” with the six others wanting the drug’s label to include patients who have had a stroke, or have cardiovascular disease.
Says there was “lots of safety talk, but vote indicated AdCom members felt the two signals could be adequately managed through label, thus meaning no patient restrictions.”
Maintains buy rating with $31 price target.
Jefferies, Michael Yee
“Our thesis and valuation are not based on specific ‘details’ of a label as we think docs will use this widely across patients...if stock was down, we think it bounces since label details don’t matter in big picture.”
Notes that while Amarin got the positive vote for approval, “investors will debate whether the labeled indication will include a more broad ‘primary prevention’ (high-risk) population which is patients w/o established CV disease but have diabetes and at least one other major” cardiovascular risk factor.
Highlights that the “stock has already run-up a lot and we wouldn’t be surprised if it’s volatile on this labeling discussion but we aren’t changing numbers as the populations are huge already.”
Maintains buy rating, $30 price target.
Cantor Fitzgerald, Louise Chen
“There were few-to-no surprises brought up during the meeting, in our view, and we think Vascepa is well on-track now for expected approval by year-end.”
Notes that most of the meeting “focused on concerns about the impact of mineral oil, as well as on which patient populations should be included in the label. Although some AdCom members believe mineral oil may have affected results somewhat, they agreed that the REDUCE-IT data are meaningful enough to warrant approval.”
Highlighted that panelist discussions on how high patients’ triglycerides, a type of fat in the blood, must be to benefit were “less clear-cut compared to the mineral oil debate.”
Maintains overweight rating with a $35 price target.
Citi, Joel Beatty
Amarin is unlikely to reach a market value above $10 billion over the next several months with its market value at $7.7 billion as of Wednesday’s close.
Sees the drug as “likely to be used in high-risk primary prevention patients regardless of” the exact label wording, though there will likely be some debate over the coming weeks regarding the label.
“We believe the exact wording of the indication matters relatively little compared to other drugs, because we believe physicians will still use Vascepa in very high risk” primary prevention patients.
Maintains buy rating and $23 price target.
Stifel, Derek Archila
“While the panel was in agreement Vascepa should be approved for secondary prevention patients (our base case), the key question that remains is whether or not Vascepa will receive a broader label inclusive of primary prevention patients, which would be upside to our model.”
“We believe approval in secondary prevention alone offers an addressable market that is at least ~10-15 million patients for Vascepa, making us comfortable with our ~$3 billion in peak sales estimate.”
Rates shares at buy with a $26 price target.
(Updates share movement in third paragraph.)
To contact the reporter on this story: Bailey Lipschultz in New York at blipschultz@bloomberg.net
To contact the editors responsible for this story: Catherine Larkin at clarkin4@bloomberg.net, Scott Schnipper, Richard Richtmyer
For more articles like this, please visit us at bloomberg.com
Antwort auf Beitrag Nr.: 61.933.181 von flyingbeef am 15.11.19 14:22:08
Sollte es zu einem BuyOut-Bidding von verschiedensten BPs ( Pfizer ? Amgen ? xxxx ? etc. ) kommen und ein sehr hohes Buyoutangebot vorliegen , dann würde ich das " nicht mit uns " nicht mehr unterschreiben.
Zitat von flyingbeef:Zitat von Magnetfeldfredy: John Thero hat nur 5 % seiner Aktien verkauft, das hat mich nicht beunruhight, 16:0 und Evaportate vor der Tür lassen auf viel mehr hoffen, bzw. eine Übernahme für US Dollar 50!
Wenn ich mich richtig erinnere:
Amarin hat aber immer gesagt Übernahme "nicht mit uns" daher ich glaube die wollen die Zügel auch langfristig in der Hand haben sollte trotzdem ordentlich nach oben rauschen.
Sollte es zu einem BuyOut-Bidding von verschiedensten BPs ( Pfizer ? Amgen ? xxxx ? etc. ) kommen und ein sehr hohes Buyoutangebot vorliegen , dann würde ich das " nicht mit uns " nicht mehr unterschreiben.
Antwort auf Beitrag Nr.: 61.933.145 von Magnetfeldfredy am 15.11.19 14:19:44
Wenn ich mich richtig erinnere:
Amarin hat aber immer gesagt Übernahme "nicht mit uns" daher ich glaube die wollen die Zügel auch langfristig in der Hand haben sollte trotzdem ordentlich nach oben rauschen.
Zitat von Magnetfeldfredy: John Thero hat nur 5 % seiner Aktien verkauft, das hat mich nicht beunruhight, 16:0 und Evaportate vor der Tür lassen auf viel mehr hoffen, bzw. eine Übernahme für US Dollar 50!
Wenn ich mich richtig erinnere:
Amarin hat aber immer gesagt Übernahme "nicht mit uns" daher ich glaube die wollen die Zügel auch langfristig in der Hand haben sollte trotzdem ordentlich nach oben rauschen.
Antwort auf Beitrag Nr.: 61.933.103 von Magnetfeldfredy am 15.11.19 14:16:43Hehe das wäre ganz grosses Kino und ja der nächste Squeeze darf gerne kommen 😎
Antwort auf Beitrag Nr.: 61.933.085 von flyingbeef am 15.11.19 14:15:30John Thero hat nur 5 % seiner Aktien verkauft, das hat mich nicht beunruhight, 16:0 und Evaportate vor der Tür lassen auf viel mehr hoffen, bzw. eine Übernahme für US Dollar 50!
Amarin - The Science Of Lipid Therapy





















