Paratek Pharmaceuticals, Inc. (PRTK) - Antibiotikawert mit Potential - Kurs am 07.11.2018 - 7$ (Seite 3)
eröffnet am 07.11.18 20:23:41 von
neuester Beitrag 24.08.22 20:01:44 von
neuester Beitrag 24.08.22 20:01:44 von
Beiträge: 29
ID: 1.292.133
ID: 1.292.133
Aufrufe heute: 0
Gesamt: 3.575
Gesamt: 3.575
Aktive User: 0
ISIN: US6993743029 · WKN: A12EGE · Symbol: N4CN
2,0420
EUR
+1,39 %
+0,0280 EUR
Letzter Kurs 20.09.23 Tradegate
Neuigkeiten
05.10.23 · globenewswire |
21.09.23 · globenewswire |
18.09.23 · globenewswire |
12.09.23 · globenewswire |
07.09.23 · globenewswire |
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,9000 | +59,66 | |
| 0,6000 | +57,48 | |
| 1,9200 | +23,87 | |
| 5,4500 | +19,00 | |
| 6,9300 | +17,46 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 4,3600 | -13,49 | |
| 1,6900 | -14,50 | |
| 2,8150 | -15,21 | |
| 2,2900 | -17,63 | |
| 2,2600 | -30,25 |
Beitrag zu dieser Diskussion schreiben
Achtung, diese Nachricht ist vom vom 04.10.2018, aber Sie fand kaum Beachtung wegen der anderen Zulassungen
Paratek Announces Acceptance of European Marketing Authorization Application for Oral and Intravenous Omadacycline
-- MAA submission for both community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI) --
-- EMA decision expected second half of 2019 --
BOSTON, Oct. 04, 2018 (GLOBE NEWSWIRE) -- Paratek Pharmaceuticals, Inc. (PRTK) today announced the acceptance of the submission to the European Medicines Agency (EMA) of the Marketing Authorization Application (MAA) for omadacycline. Paratek is seeking approval of omadacycline, a modernized tetracycline, for the treatment of community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI). Omadacycline is a once-daily oral and intravenous (IV) broad spectrum antibiotic approved under the tradename NUZYRA™ in the United States.
The MAA is supported by the company’s Phase 3 program for omadacycline, which included three pivotal registration studies: two studies in ABSSSI and one study in CABP. Omadacycline met all required EMA primary endpoints in each study and demonstrated a generally safe and well-tolerated profile.
In the U.S., the FDA approved NUZYRA™ (omadacycline) in October 2018 for the treatment of adults with community-acquired bacterial pneumonia (CABP) and acute skin and skin structure infections (ABSSSI).
About NUZYRA
NUZYRA (omadacycline) is a novel antibiotic with both once-daily intravenous (IV) and oral formulations for the treatment of community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI). A modernized tetracycline, NUZYRA is specifically designed to overcome tetracycline resistance and exhibits activity across a spectrum of bacteria, including Gram-positive, Gram-negative, atypicals, and other drug-resistant strains.
Indications and Usage
NUZYRA™ is a tetracycline class antibacterial indicated for the treatment of adult patients with the following infections caused by susceptible microorganisms:
Community-Acquired Bacterial Pneumonia (CABP) caused by the following: Streptococcus pneumoniae, Staphylococcus aureus (methicillin-susceptible isolates), Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydophila pneumoniae.
Acute Bacterial Skin and Skin Structure Infections (ABSSSI) caused by the following: Staphylococcus aureus (methicillin-susceptible and -resistant isolates), Staphylococcus lugdunensis, Streptococcus pyogenes, Streptococcus anginosus grp. (includes S. anginosus, S. intermedius, and S. constellatus), Enterococcus faecalis, Enterobacter cloacae, and Klebsiella pneumoniae.
Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of NUZYRA and other antibacterial drugs, NUZYRA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
usw.
Quelle
https://finance.yahoo.com/news/paratek-announces-acceptance-…
Paratek Announces Acceptance of European Marketing Authorization Application for Oral and Intravenous Omadacycline
-- MAA submission for both community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI) --
-- EMA decision expected second half of 2019 --
BOSTON, Oct. 04, 2018 (GLOBE NEWSWIRE) -- Paratek Pharmaceuticals, Inc. (PRTK) today announced the acceptance of the submission to the European Medicines Agency (EMA) of the Marketing Authorization Application (MAA) for omadacycline. Paratek is seeking approval of omadacycline, a modernized tetracycline, for the treatment of community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI). Omadacycline is a once-daily oral and intravenous (IV) broad spectrum antibiotic approved under the tradename NUZYRA™ in the United States.
The MAA is supported by the company’s Phase 3 program for omadacycline, which included three pivotal registration studies: two studies in ABSSSI and one study in CABP. Omadacycline met all required EMA primary endpoints in each study and demonstrated a generally safe and well-tolerated profile.
In the U.S., the FDA approved NUZYRA™ (omadacycline) in October 2018 for the treatment of adults with community-acquired bacterial pneumonia (CABP) and acute skin and skin structure infections (ABSSSI).
About NUZYRA
NUZYRA (omadacycline) is a novel antibiotic with both once-daily intravenous (IV) and oral formulations for the treatment of community-acquired bacterial pneumonia (CABP) and acute bacterial skin and skin structure infections (ABSSSI). A modernized tetracycline, NUZYRA is specifically designed to overcome tetracycline resistance and exhibits activity across a spectrum of bacteria, including Gram-positive, Gram-negative, atypicals, and other drug-resistant strains.
Indications and Usage
NUZYRA™ is a tetracycline class antibacterial indicated for the treatment of adult patients with the following infections caused by susceptible microorganisms:
Community-Acquired Bacterial Pneumonia (CABP) caused by the following: Streptococcus pneumoniae, Staphylococcus aureus (methicillin-susceptible isolates), Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydophila pneumoniae.
Acute Bacterial Skin and Skin Structure Infections (ABSSSI) caused by the following: Staphylococcus aureus (methicillin-susceptible and -resistant isolates), Staphylococcus lugdunensis, Streptococcus pyogenes, Streptococcus anginosus grp. (includes S. anginosus, S. intermedius, and S. constellatus), Enterococcus faecalis, Enterobacter cloacae, and Klebsiella pneumoniae.
Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of NUZYRA and other antibacterial drugs, NUZYRA should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria.
usw.
Quelle
https://finance.yahoo.com/news/paratek-announces-acceptance-…
Noch ein paar Folien, die ich ziemlich beeindruckend finde:
Wer sich noch mehr "verzücken" möchte,
siehe unter Quelle: Company corporate presentation
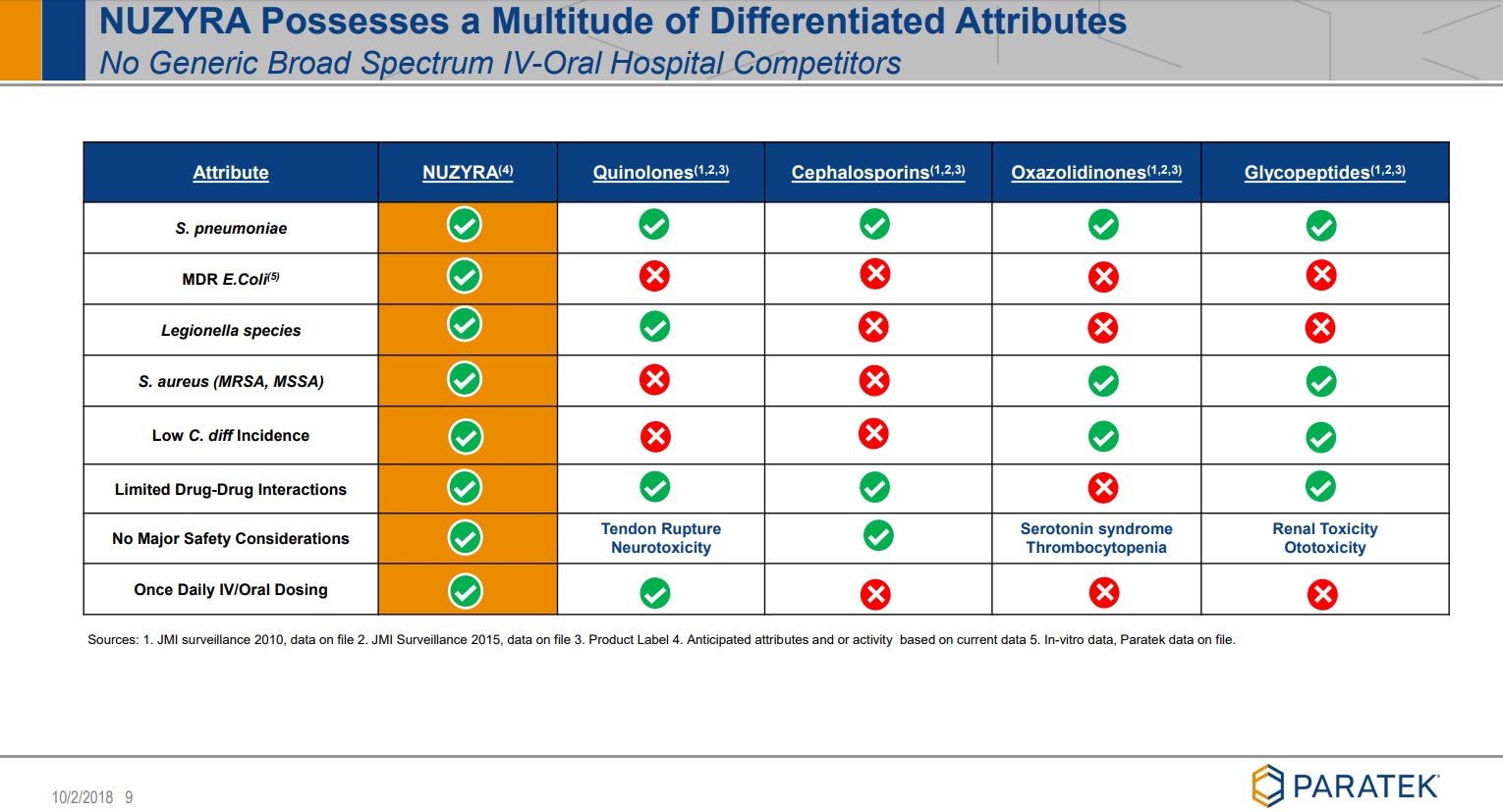
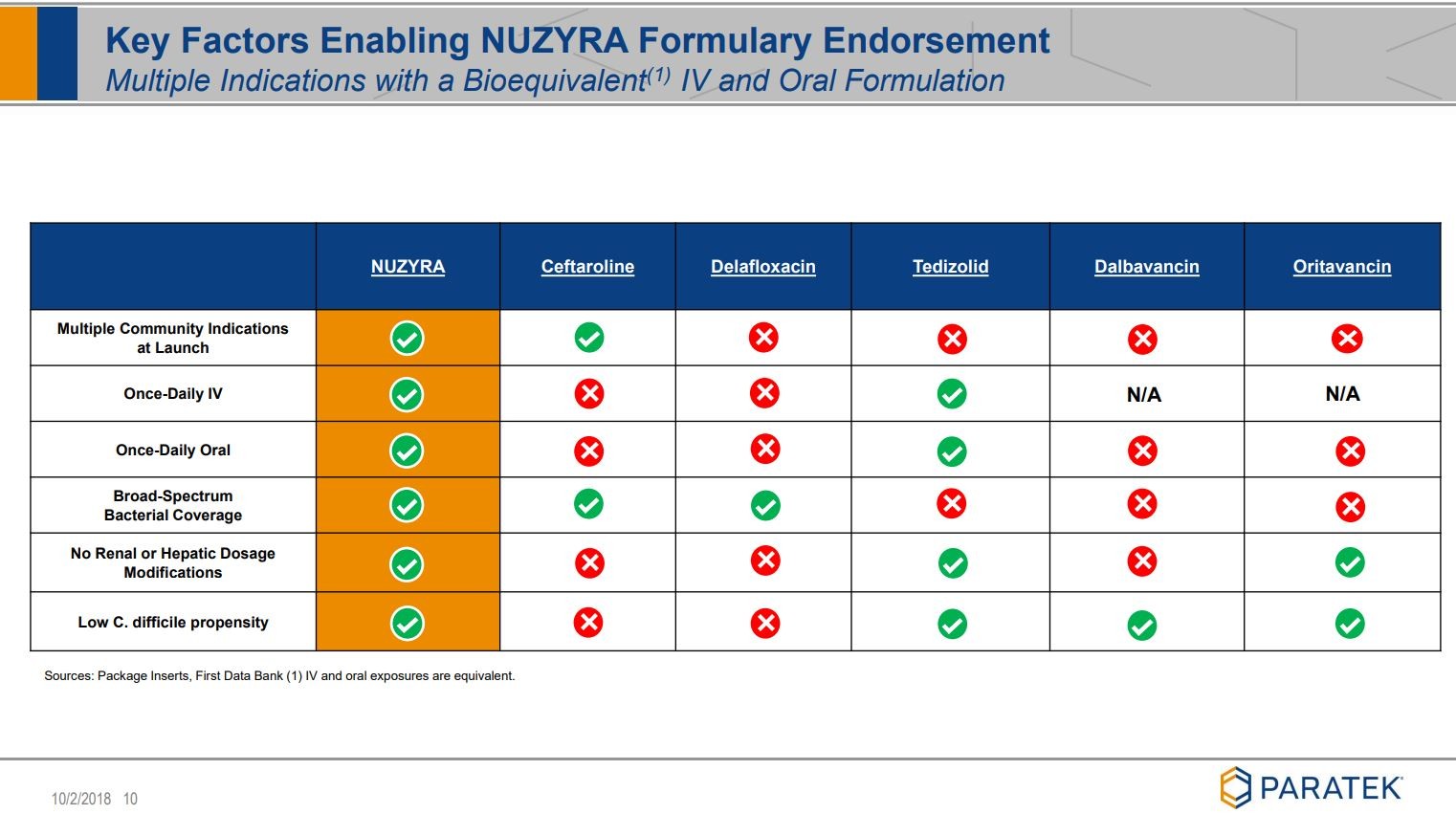
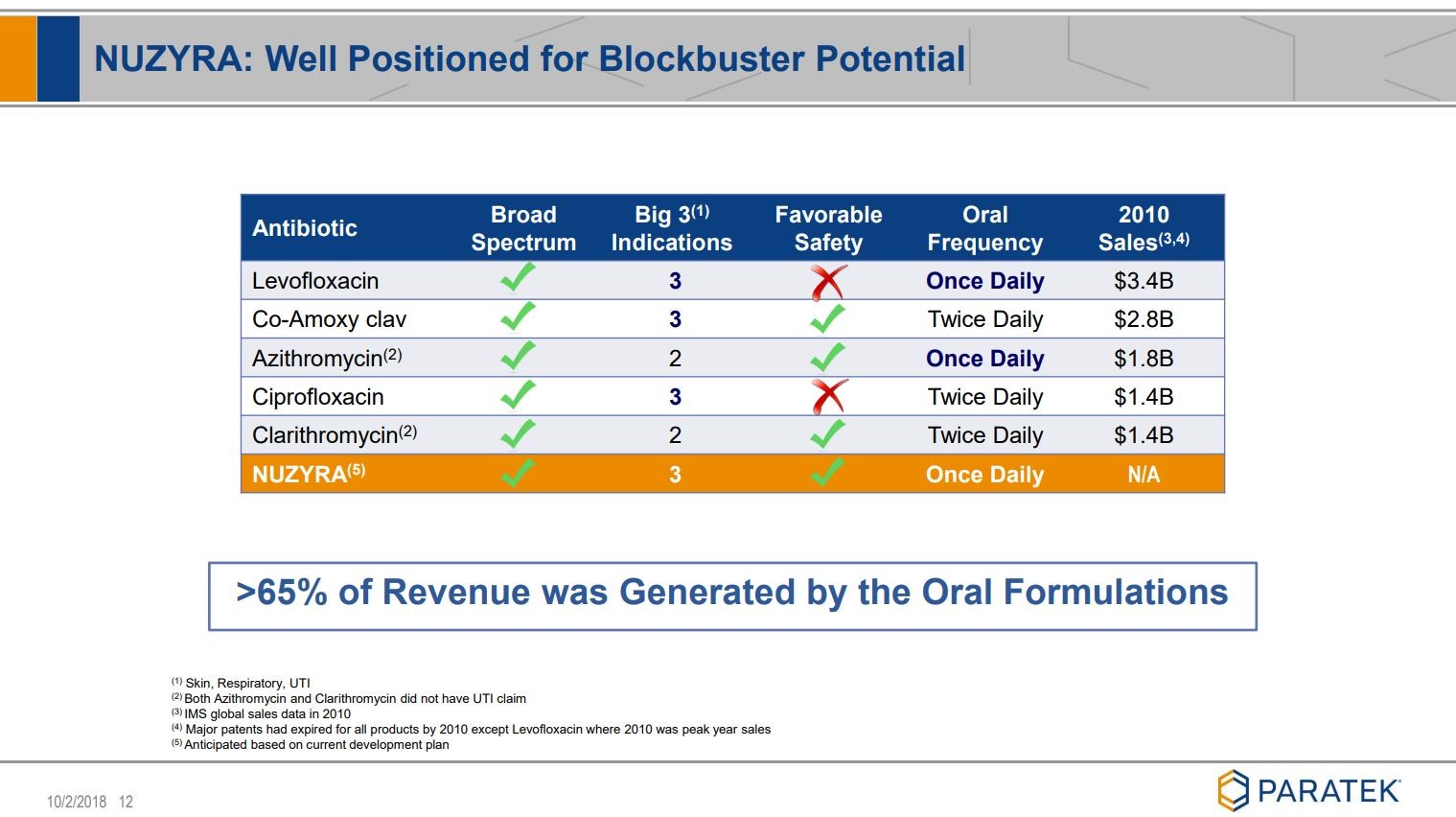
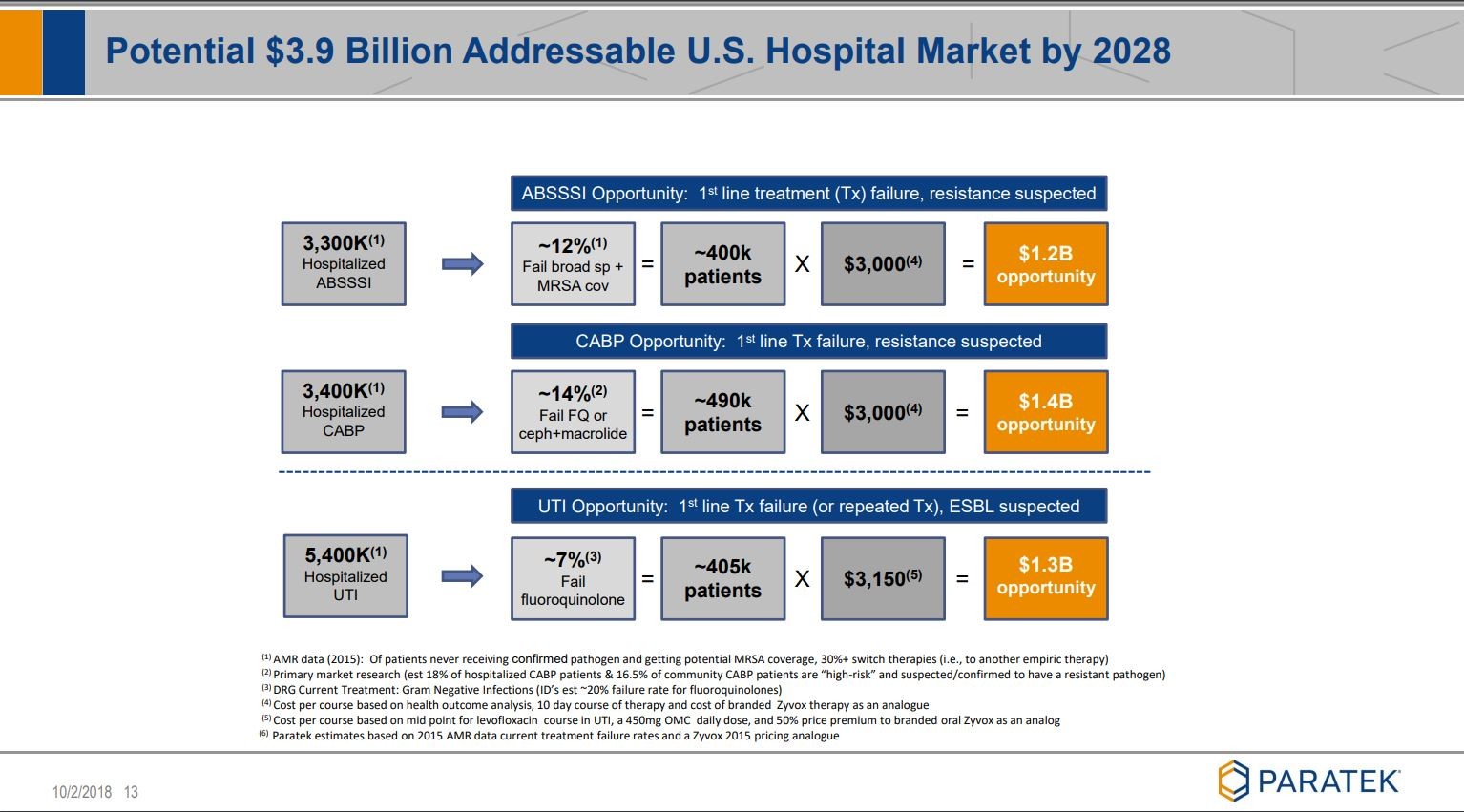
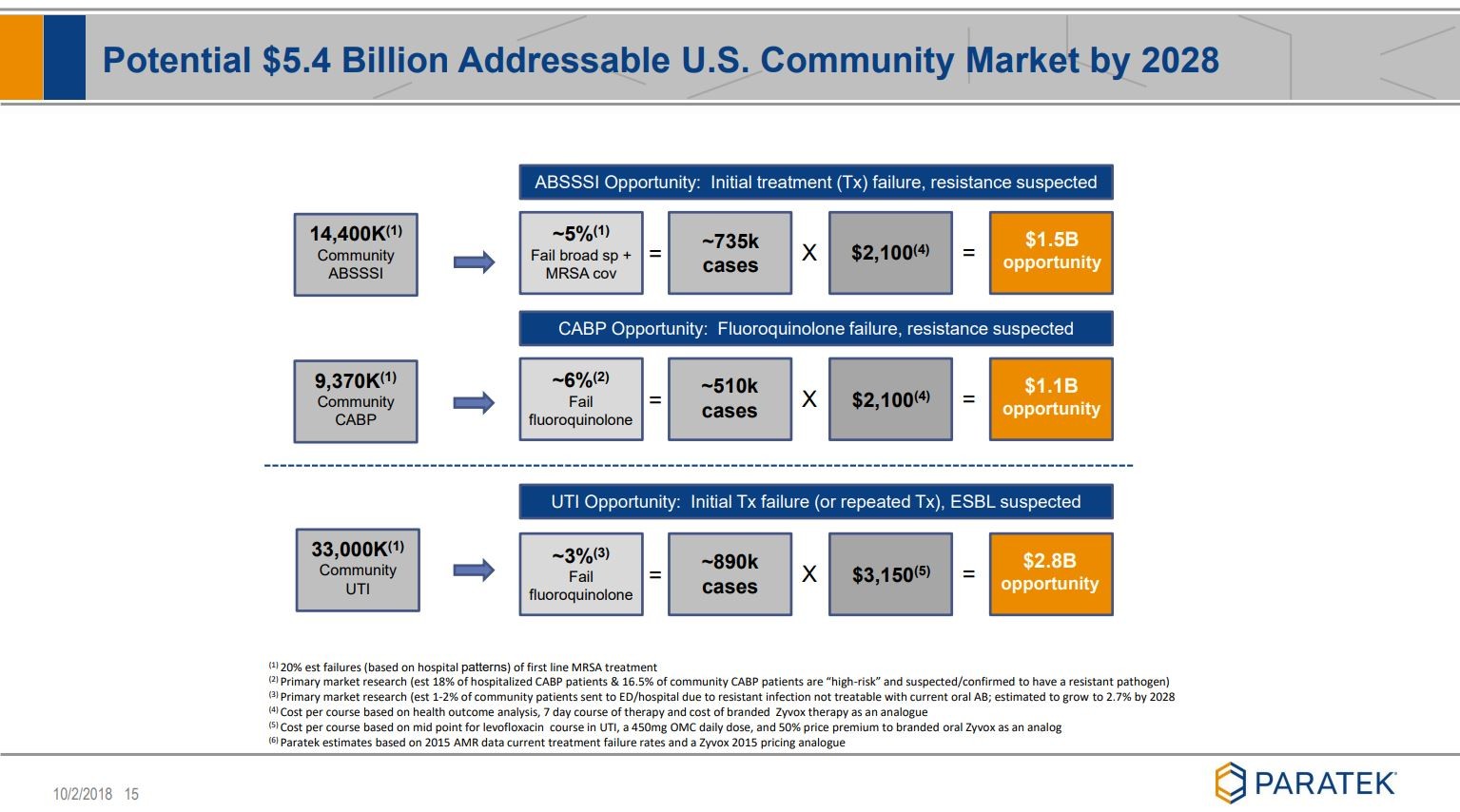

Viel Erfolg mit diesem Wert.
Die Gelegenheit ist günstig.
Alles nur meine bescheidene Meinung und keine Aufforderung zum Kauf.
Bis bald!
Wer sich noch mehr "verzücken" möchte,
siehe unter Quelle: Company corporate presentation






Viel Erfolg mit diesem Wert.
Die Gelegenheit ist günstig.
Alles nur meine bescheidene Meinung und keine Aufforderung zum Kauf.
Bis bald!
Wie versprochen, ein paar links
Quelle:
https://www.smarteranalyst.com/brief/cantor-fitzgerald-think…
Cantor Fitzgerald analyst Louise Chen maintained a Buy rating on Paratek Pharmaceuticals (NASDAQ: PRTK) today and set a price target of $50.
The company’s shares opened today at $8.85, close to its 52-week low of $8.47.
Chen said:
“We rate PRTK’s stock as 12-month price target of $50. We expect upwards earnings revisions for Nuzyra and Seysara sales to levels not reflected in consensus expectations to drive PRTK shares higher. Valuation Summary We use a blend of DCF and multiples (EV/EBITDA) to get to our 12-month price target of $50. The Disclosure Section may be found on pages 3 – 4.Valuation We use a blend of DCF and multiples (EV/EBITDA) to get to our 12-month price target of $50.”
Quelle:
https://m.benzinga.com/article/12451837?utm_referrer=https%3…
FDA Approves Paratek's Bacterial Pneumonia, Skin Infection Treatment
Paratek Pharmaceuticals, Inc. (NASDAQ: PRTK) shares reacted positively following the announcement of a Food and Drug Administration's approved treatment.
What To Know
The company announced FDA approval of Nuzyra, for the treatment of community-acquired bacterial pneumonia (CABP), as well as acute skin infections.
According to company data, Nuzyra is a modernized tetracycline, taken orally, which exhibits activity across several types of bacteria, such as Gram-positive, Gram-negative, atypicals and drug resistant strains. This will be the first antibiotic approved of its kind for nearly 20 years.
“NUZYRA offers clinicians the ability to treat patients with the IV and transition them home to complete treatment with the oral formulation. This potentially helps reduce hospitalizations and the costs associated with hospital stays,” Paratek COO Evan Loh said in a press release.
Why It’s Important
According to the Centers for Disease Control and Prevention, drug-resistant bacteria causes 2 million illnesses and over 20,000 deaths each year in the United States.
“Treating pneumonia and skin infections has become increasingly complex as existing antibiotic therapies sometimes have reduced efficacy as resistance continues to grow. This reality makes it increasingly challenging to provide safe and effective treatments to patients,” said Keith Kaye, Director of Clinical Research at the University of Michigan.
“There continues to be a need for novel antibiotics with both IV and oral formulations, such as NUZYRA, to help physicians stay ahead of the evolving resistance landscape.”
What’s Next
Roughly 2,000 adult patients have received the treatment, and the company plans on making it fully available in the first quarter of 2019. Paratek has developed plans to conduct marketing studies in pediatrics and other areas.
Paratek's stock traded up more than 7 percent to $9.93 per share.
Quelle
https://www.streetwisereports.com/article/2018/10/08/fda-app…
FDA Approves Biopharma's Drug with $1.75 Billion Peak Sales Potential
In an Oct. 3 research note, analyst Ed Arce reported the FDA approved two of Paratek Pharmaceuticals Inc.'s (PRTK:NASDAQ) therapies, Seysara and Nusyra, on Oct. 1 and Oct. 2, respectively.
As expected, Arce noted, Nusyra was approved as treatment for both community-acquired bacterial pneumonia and acute bacterial skin and skin structure infections (ABSSSI).
For both conditions, Nusyra may be given on an intravenous-to-oral basis, a 200 mg intravenous infusion first as a loading dose on day one, followed by 300 mg by mouth a day for seven to 14 days. The single intravenous administration is "an incremental positive" over the two to three days of infusion required in the pivotal trials, Arce pointed out.
For ABSSSI, an all-oral regimen is allowed, consisting of a loading dose of 450 mg once on the first and second days, followed by 300 mg per day. Because the dosing is all by mouth for this indication, Nusyra is more likely to be widely adopted.
In either indication, patients must fast for four hours before and two hours after taking a tablet, which Arce described as "one slight surprise to us."
Paratek intends to announce the price of Nusyra in Q4/18 and commercially launch the therapeutic in Q1/19. H.C. Wainwright estimates that sales of the drug will reach a peak of $1.75 billion.
The other drug, Seysara (saracycline), was approved for the treatment of inflammatory lesions resulting from non-nodular, moderate to severe acne vulgaris in 9-plus year olds, relayed Arce. Almirall, which owns the rights to Seysara, expects to commercially launch it in January 2019 and eventually reach peak annual sales of an estimated $150–200 million.
Paratek is entitled to receive from Almirall royalties on U.S. net sales, in addition to the $12 million approval milestone payment now due.
Arce reiterated his firm's Buy rating and $55 per share price target on Paratek. The target reflects more than fivebagger return potential, as the stock is currently trading at around $9.24 per share.
Quelle:
https://www.smarteranalyst.com/brief/cantor-fitzgerald-think…
Cantor Fitzgerald analyst Louise Chen maintained a Buy rating on Paratek Pharmaceuticals (NASDAQ: PRTK) today and set a price target of $50.
The company’s shares opened today at $8.85, close to its 52-week low of $8.47.
Chen said:
“We rate PRTK’s stock as 12-month price target of $50. We expect upwards earnings revisions for Nuzyra and Seysara sales to levels not reflected in consensus expectations to drive PRTK shares higher. Valuation Summary We use a blend of DCF and multiples (EV/EBITDA) to get to our 12-month price target of $50. The Disclosure Section may be found on pages 3 – 4.Valuation We use a blend of DCF and multiples (EV/EBITDA) to get to our 12-month price target of $50.”
Quelle:
https://m.benzinga.com/article/12451837?utm_referrer=https%3…
FDA Approves Paratek's Bacterial Pneumonia, Skin Infection Treatment
Paratek Pharmaceuticals, Inc. (NASDAQ: PRTK) shares reacted positively following the announcement of a Food and Drug Administration's approved treatment.
What To Know
The company announced FDA approval of Nuzyra, for the treatment of community-acquired bacterial pneumonia (CABP), as well as acute skin infections.
According to company data, Nuzyra is a modernized tetracycline, taken orally, which exhibits activity across several types of bacteria, such as Gram-positive, Gram-negative, atypicals and drug resistant strains. This will be the first antibiotic approved of its kind for nearly 20 years.
“NUZYRA offers clinicians the ability to treat patients with the IV and transition them home to complete treatment with the oral formulation. This potentially helps reduce hospitalizations and the costs associated with hospital stays,” Paratek COO Evan Loh said in a press release.
Why It’s Important
According to the Centers for Disease Control and Prevention, drug-resistant bacteria causes 2 million illnesses and over 20,000 deaths each year in the United States.
“Treating pneumonia and skin infections has become increasingly complex as existing antibiotic therapies sometimes have reduced efficacy as resistance continues to grow. This reality makes it increasingly challenging to provide safe and effective treatments to patients,” said Keith Kaye, Director of Clinical Research at the University of Michigan.
“There continues to be a need for novel antibiotics with both IV and oral formulations, such as NUZYRA, to help physicians stay ahead of the evolving resistance landscape.”
What’s Next
Roughly 2,000 adult patients have received the treatment, and the company plans on making it fully available in the first quarter of 2019. Paratek has developed plans to conduct marketing studies in pediatrics and other areas.
Paratek's stock traded up more than 7 percent to $9.93 per share.
Quelle
https://www.streetwisereports.com/article/2018/10/08/fda-app…
FDA Approves Biopharma's Drug with $1.75 Billion Peak Sales Potential
In an Oct. 3 research note, analyst Ed Arce reported the FDA approved two of Paratek Pharmaceuticals Inc.'s (PRTK:NASDAQ) therapies, Seysara and Nusyra, on Oct. 1 and Oct. 2, respectively.
As expected, Arce noted, Nusyra was approved as treatment for both community-acquired bacterial pneumonia and acute bacterial skin and skin structure infections (ABSSSI).
For both conditions, Nusyra may be given on an intravenous-to-oral basis, a 200 mg intravenous infusion first as a loading dose on day one, followed by 300 mg by mouth a day for seven to 14 days. The single intravenous administration is "an incremental positive" over the two to three days of infusion required in the pivotal trials, Arce pointed out.
For ABSSSI, an all-oral regimen is allowed, consisting of a loading dose of 450 mg once on the first and second days, followed by 300 mg per day. Because the dosing is all by mouth for this indication, Nusyra is more likely to be widely adopted.
In either indication, patients must fast for four hours before and two hours after taking a tablet, which Arce described as "one slight surprise to us."
Paratek intends to announce the price of Nusyra in Q4/18 and commercially launch the therapeutic in Q1/19. H.C. Wainwright estimates that sales of the drug will reach a peak of $1.75 billion.
The other drug, Seysara (saracycline), was approved for the treatment of inflammatory lesions resulting from non-nodular, moderate to severe acne vulgaris in 9-plus year olds, relayed Arce. Almirall, which owns the rights to Seysara, expects to commercially launch it in January 2019 and eventually reach peak annual sales of an estimated $150–200 million.
Paratek is entitled to receive from Almirall royalties on U.S. net sales, in addition to the $12 million approval milestone payment now due.
Arce reiterated his firm's Buy rating and $55 per share price target on Paratek. The target reflects more than fivebagger return potential, as the stock is currently trading at around $9.24 per share.
Antwort auf Beitrag Nr.: 59.162.751 von Cyberhexe am 07.11.18 21:30:26Hallo "Cyberhexe",
ich bin hocherfreut über den "prominenten" Einstieg
Verfolge deine Beiträge schon über 10 Jahre.
Ich hätte damals öfter mal auf deine Hinweise hören sollen,
dann wären mir herbe Verluste und Haarverlust erspart geblieben (GPC Biotech)
Dann auf gutes Gelingen und schöne Zeiten mit dieser Perle.
ich bin hocherfreut über den "prominenten" Einstieg

Verfolge deine Beiträge schon über 10 Jahre.
Ich hätte damals öfter mal auf deine Hinweise hören sollen,
dann wären mir herbe Verluste und Haarverlust erspart geblieben (GPC Biotech)
Dann auf gutes Gelingen und schöne Zeiten mit dieser Perle.
Antwort auf Beitrag Nr.: 59.162.751 von Cyberhexe am 07.11.18 21:30:26wobei der Preis für NUZYRA moderat ist:


Antwort auf Beitrag Nr.: 59.162.211 von amherster am 07.11.18 20:33:09...bin ebenfalls "prominent" eingestiegen.
Die Marktkapitalisierung von Paratek ist mittlerweile richtig lächerlich, obschon das Geschäftsmodell bei den Antibiotikas derzeit in Frage gestellt ist: routinemässig werden die generischen, weil billigen, Antibiotika eingesetzt und die neuen Antibiotika nur dann, wenn die Standardtherapie nicht anschlägt. Damit sind natürlich nicht mehr die ganz grossen Umsätze zu machen, weshalb sich die grossen Pharmafirmen mehrheitlich aus der Antibiotika-Forschung zurückgezogen haben. Ich bin mir jedoch sicher, dass die Gesundheitsbehörden irgendwie nachjustieren. Die Gefahr einer "Epidemie" mit multiresistenten Keimen und keinen geeigneten Therapiemöglichkeiten kann nicht das Ziel sein.
Und PRTK ist mit den 2 kürzlich zugelassenen Medikamenten derat gut aufgestellt, dass zweistellige Kurse sehr wahrscheinlich relativ kurzfristig wieder erzielt werden...spätestens bei der Markteinführung von NUZYRA im Februar 2019.
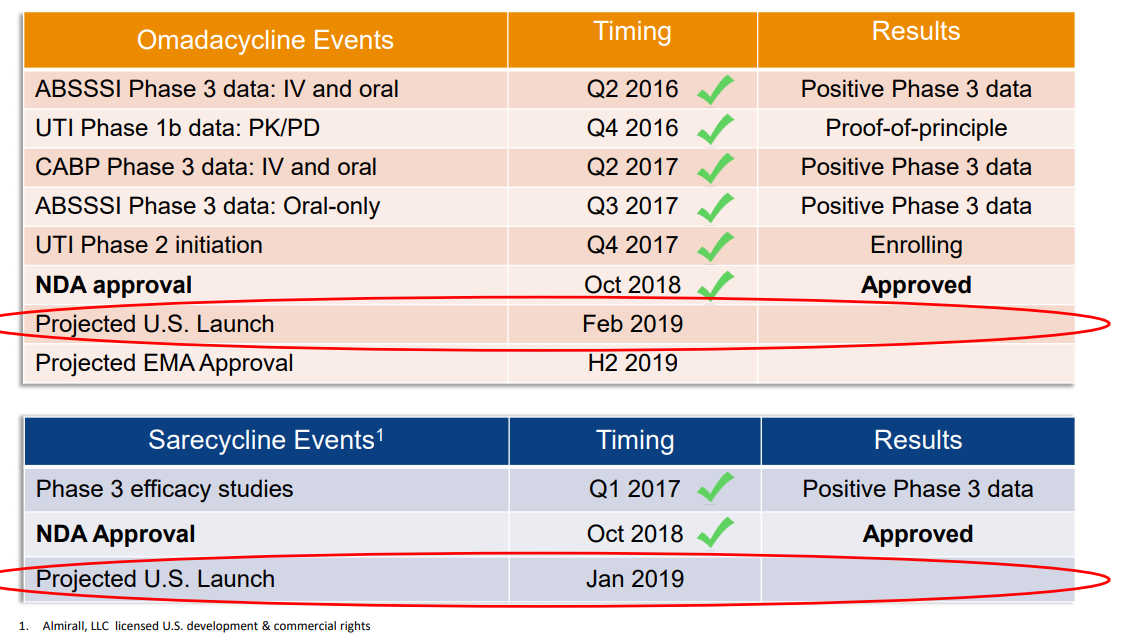
Die Marktkapitalisierung von Paratek ist mittlerweile richtig lächerlich, obschon das Geschäftsmodell bei den Antibiotikas derzeit in Frage gestellt ist: routinemässig werden die generischen, weil billigen, Antibiotika eingesetzt und die neuen Antibiotika nur dann, wenn die Standardtherapie nicht anschlägt. Damit sind natürlich nicht mehr die ganz grossen Umsätze zu machen, weshalb sich die grossen Pharmafirmen mehrheitlich aus der Antibiotika-Forschung zurückgezogen haben. Ich bin mir jedoch sicher, dass die Gesundheitsbehörden irgendwie nachjustieren. Die Gefahr einer "Epidemie" mit multiresistenten Keimen und keinen geeigneten Therapiemöglichkeiten kann nicht das Ziel sein.
Und PRTK ist mit den 2 kürzlich zugelassenen Medikamenten derat gut aufgestellt, dass zweistellige Kurse sehr wahrscheinlich relativ kurzfristig wieder erzielt werden...spätestens bei der Markteinführung von NUZYRA im Februar 2019.

ACHTUNG!
Charts sind Screenshots zur Dokumentation!!!
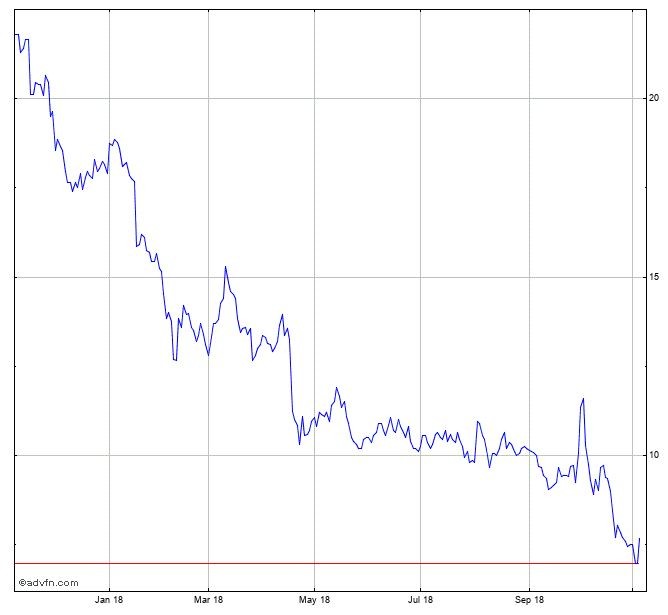
Jahreschart, Stand 7.11.2018
Charts sind Screenshots zur Dokumentation!!!

Jahreschart, Stand 7.11.2018

Intradaychart von heute!
Quelle: IHUB
Paratek Pharmaceuticals, Inc. (PRTK)
Kurs am 07.11.2018 – 7,00 $ (mein Einstieg heute Nachmittag bei 7,35$)
Hier möchte ich einen Wert vorstellen, der meiner Meinung nach in den nächsten Monaten hypen wird.
Wer gern antizyklisch handelt und gern in Werte einsteigt, die vielleicht zur Zeit nicht so „hipp“ sind, der könnte hier fündig werden.
Hier gab es zwei ziemlich heftige Intradayreversals hintereinander, so dass ich glaube, der Boden wurde markiert.
Antibiotika, es wird viel darüber gesprochen, doch niemand traut sich so recht ran an diese „Perle“.
Paratek hat Anfang Oktober gleich mehrere Zulassungen bekommen,
was bei MJ – Werten zu mehreren hundert Prozent Ausschlägen geführt hätte.
Diese Perle wird dagegen seit einigen Monaten und speziell seit der Zulassung gehandelt wie Sauerbier.
Ich bin der Meinung, dass jetzt ein echt guter Zeitpunkt gekommen ist, um einzusteigen.
Alles unter 12€ ist derzeitig ein guter Deal. (mM)
Ein paar links zum Einlesen folgen zeitnah.
Würde mich freuen, wenn ich dadurch den Anstoß zu einer steilen Reise für einige Mitleser / Beteiligte geben konnte.
Viel Erfolg / Glück / Spaß
Ich werde mich nur mit links, Kopien oder Meinungen beteiligen können (und auch das aus Zeitgründen nur begrenzt).
Für eine wissenschaftliche Bemerkung fehlt mir der Horizont.
Aber vielleicht werden sich hier auch Personen anschließen,
welche uns an ihrem Wissen bzw. ihrer fachlichen Meinung teilhaben lassen.
Noch ein paar Daten von heute (20.00 Uhr).
Quelle yahoo
Day's Range - 6.27 - 7.62
52 Week Range - 6.27 - 22.45
Volume - 1,529,543
Avg. Volume - 810,253
Market Cap – 240 Mio
...und wie ich es so schreibe, wandern wir Richtung 7,70 -> cool
Kurs am 07.11.2018 – 7,00 $ (mein Einstieg heute Nachmittag bei 7,35$)
Hier möchte ich einen Wert vorstellen, der meiner Meinung nach in den nächsten Monaten hypen wird.
Wer gern antizyklisch handelt und gern in Werte einsteigt, die vielleicht zur Zeit nicht so „hipp“ sind, der könnte hier fündig werden.
Hier gab es zwei ziemlich heftige Intradayreversals hintereinander, so dass ich glaube, der Boden wurde markiert.
Antibiotika, es wird viel darüber gesprochen, doch niemand traut sich so recht ran an diese „Perle“.
Paratek hat Anfang Oktober gleich mehrere Zulassungen bekommen,
was bei MJ – Werten zu mehreren hundert Prozent Ausschlägen geführt hätte.
Diese Perle wird dagegen seit einigen Monaten und speziell seit der Zulassung gehandelt wie Sauerbier.
Ich bin der Meinung, dass jetzt ein echt guter Zeitpunkt gekommen ist, um einzusteigen.
Alles unter 12€ ist derzeitig ein guter Deal. (mM)
Ein paar links zum Einlesen folgen zeitnah.
Würde mich freuen, wenn ich dadurch den Anstoß zu einer steilen Reise für einige Mitleser / Beteiligte geben konnte.
Viel Erfolg / Glück / Spaß
Ich werde mich nur mit links, Kopien oder Meinungen beteiligen können (und auch das aus Zeitgründen nur begrenzt).
Für eine wissenschaftliche Bemerkung fehlt mir der Horizont.
Aber vielleicht werden sich hier auch Personen anschließen,
welche uns an ihrem Wissen bzw. ihrer fachlichen Meinung teilhaben lassen.
Noch ein paar Daten von heute (20.00 Uhr).
Quelle yahoo
Day's Range - 6.27 - 7.62
52 Week Range - 6.27 - 22.45
Volume - 1,529,543
Avg. Volume - 810,253
Market Cap – 240 Mio
...und wie ich es so schreibe, wandern wir Richtung 7,70 -> cool





















