INTERCEPT (ICPT) mit Potenzial USD 64.46 (27.9.19) (Seite 4)
eröffnet am 28.09.19 23:46:35 von
neuester Beitrag 21.01.23 17:18:40 von
neuester Beitrag 21.01.23 17:18:40 von
Beiträge: 46
ID: 1.312.902
ID: 1.312.902
Aufrufe heute: 0
Gesamt: 5.005
Gesamt: 5.005
Aktive User: 0
ISIN: US45845P1084 · WKN: A1J5U0 · Symbol: ICPT
19,000
USD
0,00 %
0,000 USD
Letzter Kurs 08.11.23 Nasdaq
Neuigkeiten
29.02.24 · globenewswire |
13.11.23 · globenewswire |
10.11.23 · globenewswire |
08.11.23 · globenewswire |
16.10.23 · Business Wire (engl.) |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 6,0000 | +25,00 | |
| 0,6400 | +18,52 | |
| 0,6800 | +16,54 | |
| 0,9060 | +14,83 | |
| 1,0900 | +14,74 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 0,8410 | -17,06 | |
| 9,7200 | -19,60 | |
| 4,0000 | -27,27 | |
| 2,7280 | -29,14 | |
| 14,510 | -32,32 |
Beitrag zu dieser Diskussion schreiben
Antwort auf Beitrag Nr.: 62.101.796 von grayWOLF am 07.12.19 00:52:30stimmt, das war auch das einzige, was ich gefunden habe. Andererseits war es klar, dass ein Kurs, der so steil ansteigt, auch mal einen deutlichen Rücksetzer erleben kann, aber 10 % fand ich schon heftig.
Der Kurs Richtung Süden heute, habe aber nix Negatives gefunden,,,,,https://ca.advfn.com/stock-market/NASDAQ/ICPT/stock-news/812… 

das hat sich jetzt geändert.....aber viele sind nicht unbedingt dabei....
Gute news und 0 Umsatz an deutschen Handelsplätzen.... ....http://ir.interceptpharma.com/news-releases/news-release-det….
....http://ir.interceptpharma.com/news-releases/news-release-det…. 
 ....http://ir.interceptpharma.com/news-releases/news-release-det….
....http://ir.interceptpharma.com/news-releases/news-release-det…. 
Bravo, endlich mal jemand, der eine nachhaltige Pharma-Aktie mit einem wahren Boom-Effekt ins Forum stellt! Bezüglich NASH sind einige am Start, der Amrkt gibt viel her, aber einer muss eben der Erste sein und das wird INTERCEPT! Ich habe ein langes Gesprach mit einem Biotech-Fondmanager geführt, der durchblicken ließ, dass man diesem Wert eine Verdoppelung bis März zutraut, da ist die finale Zulassung noch nicht einmal inbegriffen! Kaufen Kaufen Kaufen....
Antwort auf Beitrag Nr.: 61.591.119 von Cyberhexe am 28.09.19 23:46:35
spätestens am 25. November hat die FDA die formale Annahme des Zulassungsantrages sowie die Entscheidung über "Priority Review" bekanntzugeben.
Dreistelliger Kurs dürfte bald in Sichtweite sein:
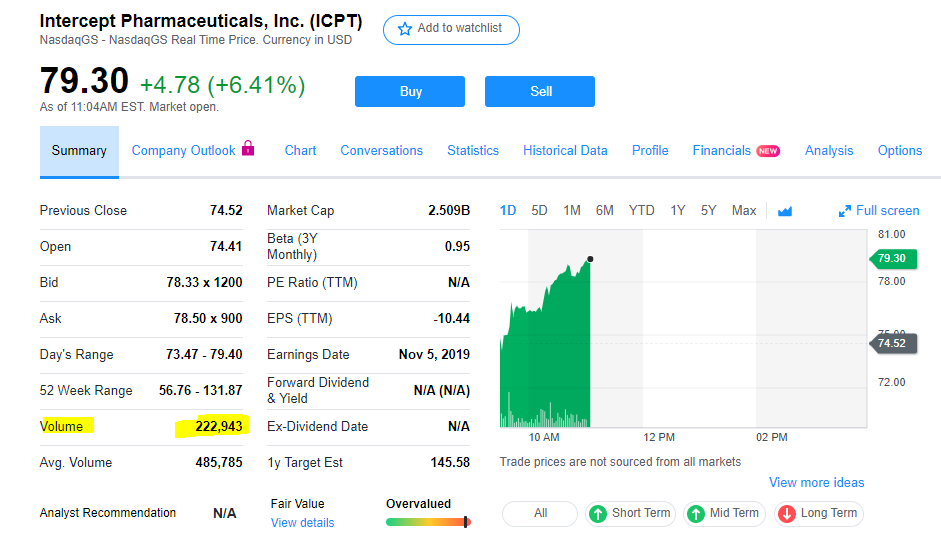
Zitat von Cyberhexe: Intercept hat den Zulassungsantrag (NDA) von Ocaliva zur Behandlung von durch NASH verursachte Fibrose, wie angekündigt im 3q19, bei der FDA eingereicht und kündigt bereits an, im 4q19 den Zulassungsantrag auch bei der EMA einzureichen. Gleichzeitig wurde bei der FDA eine beschleunigte Bearbeitung des Zulassungsantrages (Priority Review) beantragt. Falls dies genehmigt wird, und davon ist auszugehen, könnte eine Zulassungsentscheidung Ende März 2020 realistisch sein.
Bin mit 500 Stück am Freitag eingestiegen.
Sep 27, 2019
Intercept Submits New Drug Application to the U.S. FDA for Obeticholic Acid in Patients with Fibrosis Due to NASH
PDF Version
NDA supported by positive interim analysis results from REGENERATE Phase 3 study demonstrating OCA’s improvement of liver fibrosis without worsening of NASH
ir.interceptpharma.com/news-releases/news-release-details/in…
spätestens am 25. November hat die FDA die formale Annahme des Zulassungsantrages sowie die Entscheidung über "Priority Review" bekanntzugeben.
Dreistelliger Kurs dürfte bald in Sichtweite sein:

Antwort auf Beitrag Nr.: 61.845.066 von Cyberhexe am 05.11.19 20:29:31...von der Quartalspressekonferenz von heute:
- interessanter Hinweis auf das Treffen der "American Association for the study of Liver Diseases" --> AASLD, welches vom 8. bis 12. November in Boston, Massachusetts, stattfinden wird. Mehr als 20 "Abstracts" und "late breaking abstracts" werden angekündigt. Vora allem der Hinweis, dass "...demonstrating OCA's durable therapeutic benefit with no new long-term safety findings in PBC patients on treatment for up to six years"
kein neues Signal bei der Langzeittherapie (5 Jahre !) festgestellt wurde, erhöht die Zulassungswahrscheinlichkeit auch in der Indikation "NASH mit Fibrose".
In addition to the execution of our regulatory filings, we've continued to be laser focused on ensuring commercial readiness for the first-ever NASH launch. With more than 15 years of experience focused on the development of novel therapies to treat progressive non-viral liver diseases, Intercept remains the only company to have demonstrated a therapeutic anti-fibrotic benefit in large placebo-controlled Phase II and III trials with our first-in-class FXR agonist OCA that we believe to be crucial for the effective treatment of patients with advanced fibrosis due to NASH.
As the leader in this space, we've proven time and again, there simply are no shortcuts. Developing effective new treatments for these indications is a marathon, not a sprint, and we remain well positioned for continued success with the anticipated first-approved NASH therapy on the horizon. We are in the fortunate position to be building on our established strong standing within the liver community globally. Based on the foundation we've built and ongoing commercial success of our PBC business worldwide, we have great confidence in our ability to execute a successful first-to-market launch of OCA in NASH, which provided FDA grants us priority review and approval, could be as early as the spring of 2020.
I mentioned PBC and I'm pleased to point out the continued strong momentum in our business globally. We've continued to see steady demand growth versus the prior year quarter, based on solid execution of our commercial organization worldwide. Given our performance to date, we've now increased our 2019 full year net sales guidance for Ocaliva to between $245 million and $250 million.
We believe our established medical and commercial infrastructure supporting the PBC business uniquely positions us for success in NASH. We've developed strong relationships with hepatologists and gastroenterologists, the specialists treating patients with advanced fibrosis due to NASH, many of whom have already gained valuable experience prescribing Ocaliva to PBC patients. As we've previously stated, we are flexing up our existing infrastructure and capabilities, while building on our relationships within the community in preparation for our NASH launch.
Following its anticipated approval, OCA is positioned to become the foundational therapy in patients with advanced fibrosis due to NASH. We continue to have productive interactions with physicians, payers and patient groups, which Jerry will discuss in more detail shortly. These stakeholders all recognize the critical importance of a therapy with a robust anti-fibrotic benefit, underscoring what we believe to be OCA's key advantage.
I now want to pivot and spend some time discussing the upcoming Annual Meeting of the American Association for the study of liver diseases, the AASLD Liver meeting, where we will have a significant presence and more than 20 abstracts being presented in the general and late-breaker sessions. Our team is dedicated to our customers in the hepatology community and we're excited about the posters and presentations that will be showcased at the liver meeting.
Some highlights, on the PBC side, we're presenting the final results from our Phase III poised 5-year open-label phase, demonstrating OCA's durable therapeutic benefit with no new long-term safety findings in PBC patients on treatment for up to six years. On the NASH side, we'll have an oral presentation of the REGENERATE interim analysis results in the expanded intent-to-treat population, which reinforces the consistent benefit that OCA provides across the broader patient population. There are also two important patient-reported outcome or PRO abstracts from REGENERATE, including one demonstrating that patient-reported quality of life scores are substantially below population norms, indicating that NASH with established fibrosis is not an asymptomatic disease. And that effective anti-fibrotic treatment can improve PRO scores.
- interessanter Hinweis auf das Treffen der "American Association for the study of Liver Diseases" --> AASLD, welches vom 8. bis 12. November in Boston, Massachusetts, stattfinden wird. Mehr als 20 "Abstracts" und "late breaking abstracts" werden angekündigt. Vora allem der Hinweis, dass "...demonstrating OCA's durable therapeutic benefit with no new long-term safety findings in PBC patients on treatment for up to six years"
kein neues Signal bei der Langzeittherapie (5 Jahre !) festgestellt wurde, erhöht die Zulassungswahrscheinlichkeit auch in der Indikation "NASH mit Fibrose".
In addition to the execution of our regulatory filings, we've continued to be laser focused on ensuring commercial readiness for the first-ever NASH launch. With more than 15 years of experience focused on the development of novel therapies to treat progressive non-viral liver diseases, Intercept remains the only company to have demonstrated a therapeutic anti-fibrotic benefit in large placebo-controlled Phase II and III trials with our first-in-class FXR agonist OCA that we believe to be crucial for the effective treatment of patients with advanced fibrosis due to NASH.
As the leader in this space, we've proven time and again, there simply are no shortcuts. Developing effective new treatments for these indications is a marathon, not a sprint, and we remain well positioned for continued success with the anticipated first-approved NASH therapy on the horizon. We are in the fortunate position to be building on our established strong standing within the liver community globally. Based on the foundation we've built and ongoing commercial success of our PBC business worldwide, we have great confidence in our ability to execute a successful first-to-market launch of OCA in NASH, which provided FDA grants us priority review and approval, could be as early as the spring of 2020.
I mentioned PBC and I'm pleased to point out the continued strong momentum in our business globally. We've continued to see steady demand growth versus the prior year quarter, based on solid execution of our commercial organization worldwide. Given our performance to date, we've now increased our 2019 full year net sales guidance for Ocaliva to between $245 million and $250 million.
We believe our established medical and commercial infrastructure supporting the PBC business uniquely positions us for success in NASH. We've developed strong relationships with hepatologists and gastroenterologists, the specialists treating patients with advanced fibrosis due to NASH, many of whom have already gained valuable experience prescribing Ocaliva to PBC patients. As we've previously stated, we are flexing up our existing infrastructure and capabilities, while building on our relationships within the community in preparation for our NASH launch.
Following its anticipated approval, OCA is positioned to become the foundational therapy in patients with advanced fibrosis due to NASH. We continue to have productive interactions with physicians, payers and patient groups, which Jerry will discuss in more detail shortly. These stakeholders all recognize the critical importance of a therapy with a robust anti-fibrotic benefit, underscoring what we believe to be OCA's key advantage.
I now want to pivot and spend some time discussing the upcoming Annual Meeting of the American Association for the study of liver diseases, the AASLD Liver meeting, where we will have a significant presence and more than 20 abstracts being presented in the general and late-breaker sessions. Our team is dedicated to our customers in the hepatology community and we're excited about the posters and presentations that will be showcased at the liver meeting.
Some highlights, on the PBC side, we're presenting the final results from our Phase III poised 5-year open-label phase, demonstrating OCA's durable therapeutic benefit with no new long-term safety findings in PBC patients on treatment for up to six years. On the NASH side, we'll have an oral presentation of the REGENERATE interim analysis results in the expanded intent-to-treat population, which reinforces the consistent benefit that OCA provides across the broader patient population. There are also two important patient-reported outcome or PRO abstracts from REGENERATE, including one demonstrating that patient-reported quality of life scores are substantially below population norms, indicating that NASH with established fibrosis is not an asymptomatic disease. And that effective anti-fibrotic treatment can improve PRO scores.
Antwort auf Beitrag Nr.: 61.840.920 von paul81 am 05.11.19 14:31:36Intercept Pharmaceuticals (NASDAQ:ICPT)‘s stock had its “buy” rating reiterated by analysts at Needham & Company LLC in a research report issued to clients and investors on Tuesday, AnalystRatings.com reports. They presently have a $150.00 price target on the biopharmaceutical company’s stock.
...Needham hat das Kursziel von $150 bestätigt - nit too bad!
...Needham hat das Kursziel von $150 bestätigt - nit too bad!
Antwort auf Beitrag Nr.: 61.840.350 von Cyberhexe am 05.11.19 13:19:49nicht gerade beeindruckendes Quartal, Ocaliva net sales im Vergleich zu Q2 rückläufig (65.9 -> 61.5), Geld ist noch genug vorhanden, alles hängt von der Zulassung für NASH nächstes Jahr ab
Antwort auf Beitrag Nr.: 61.770.947 von Cyberhexe am 26.10.19 09:24:27Intercept Pharmaceuticals Reports Third Quarter 2019 Financial Results and Provides Business Update
Worldwide Ocaliva net sales of $61.5 million in the third quarter of 2019 representing 32% growth versus the prior year quarter; increasing full year 2019 worldwide Ocaliva net sales guidance range to between $245 million and $250 million
U.S. NDA for NASH submitted; EU MAA submission for NASH planned for fourth quarter of 2019
New NASH and PBC clinical data to be presented at The Liver Meeting® 2019
Conference call scheduled for 8:30 a.m. ET today
http://ir.interceptpharma.com/news-releases/news-release-det…
Worldwide Ocaliva net sales of $61.5 million in the third quarter of 2019 representing 32% growth versus the prior year quarter; increasing full year 2019 worldwide Ocaliva net sales guidance range to between $245 million and $250 million
U.S. NDA for NASH submitted; EU MAA submission for NASH planned for fourth quarter of 2019
New NASH and PBC clinical data to be presented at The Liver Meeting® 2019
Conference call scheduled for 8:30 a.m. ET today
http://ir.interceptpharma.com/news-releases/news-release-det…
INTERCEPT (ICPT) mit Potenzial USD 64.46 (27.9.19)





















