Trinity biotech eine Aktie mit Phantsie - 500 Beiträge pro Seite (Seite 2)
eröffnet am 25.10.01 08:11:29 von
neuester Beitrag 30.07.15 12:37:45 von
neuester Beitrag 30.07.15 12:37:45 von
Beiträge: 899
ID: 493.776
ID: 493.776
Aufrufe heute: 0
Gesamt: 79.844
Gesamt: 79.844
Aktive User: 0
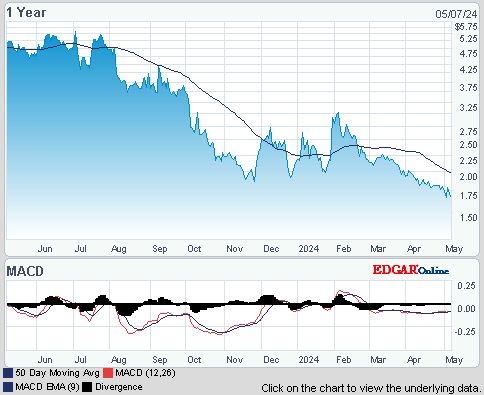
ISIN: US8964385046 · WKN: A404JF · Symbol: TRIB
1,6800
USD
-4,00 %
-0,0700 USD
Letzter Kurs 23:20:00 NYSE
Neuigkeiten
15.02.24 · globenewswire |
13.02.24 · globenewswire |
08.02.24 · globenewswire |
07.02.24 · globenewswire |
31.01.24 · globenewswire |
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 3,3000 | +70,98 | |
| 1,7850 | +42,80 | |
| 1,6800 | +32,28 | |
| 1,0700 | +31,13 | |
| 1,7500 | +27,27 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 23,390 | -16,34 | |
| 0,7500 | -16,67 | |
| 0,5305 | -17,51 | |
| 3,5000 | -26,24 | |
| 11,500 | -29,23 |
Hallo Mant!
Schön zu hören das es noch mehr Interessierte gibt.
Ich kann auch nur Sachen reinstellen die ich irgendwo lese. Was anderes ist nicht glaubwürdig, auch wenn es stimmt.
@kaiholla
Ich muß neidlos anerkennen, das Du mit Deinen 2€ richtig lagst.
Gruss
vaka
Schön zu hören das es noch mehr Interessierte gibt.
Ich kann auch nur Sachen reinstellen die ich irgendwo lese. Was anderes ist nicht glaubwürdig, auch wenn es stimmt.
@kaiholla
Ich muß neidlos anerkennen, das Du mit Deinen 2€ richtig lagst.
Gruss
vaka
wer ist denn kaiholla? Kann keinen Beitrag finden.
kaiholla war auch mal investiert.
Aus yahoo:
open letter to the trib board
by: w_onecyprus
Long-Term Sentiment: Hold 12/11/04 08:45 am
Msg: 52955 of 52960
Dear Sirs,
Firtsly let me wish you and Trib a merry xmas and a happy new year.Although the spiritual part is at the end of the year highlighted one cannot ignore that bonus time has arrived ,and I am in case of our company sure that the proposals to which you most likely have to nick will be generous ,however as a long time shareholder I am of the opinion and certainly a lot more investors that your senior mangement deserves a kick in the ass rather then any bonus.The reasons are :a total ignorance for the interest of the owners of this company,financial mismanagement they have been unable to cash in on inventories,accounts receivables is way out of control and on the money in the bank they have not even obtained a decent return instead they are still paying excessive interest charges for some outstanding loan.In addition internal control and ethics should not allow any board or management member to indulge in real estate deals whereby the company picks up the tab.The further dilution of our shares by the most stupid arrangement of all,a shareprice over 6 dollars and at the same time selling at 4,25 tops it all.The result has been a share price well under three dollars whilst your direct competitor who does not make money has a value of 2,5 time ours.Commendable is our product line and the workers who have been at work,for our management no bonus whatsoever unless they get this share over 10 dollars within six months.
psThe readers of this board may send a letter on the same lines to the directors.
Re: open letter to the trib board
by: stklord01 12/11/04 06:19 pm
Msg: 52958 of 52960
No PR needed.
If TRIB continues to grow top line and bottom line numbers share price will follow.
Aus yahoo:
open letter to the trib board
by: w_onecyprus
Long-Term Sentiment: Hold 12/11/04 08:45 am
Msg: 52955 of 52960
Dear Sirs,
Firtsly let me wish you and Trib a merry xmas and a happy new year.Although the spiritual part is at the end of the year highlighted one cannot ignore that bonus time has arrived ,and I am in case of our company sure that the proposals to which you most likely have to nick will be generous ,however as a long time shareholder I am of the opinion and certainly a lot more investors that your senior mangement deserves a kick in the ass rather then any bonus.The reasons are :a total ignorance for the interest of the owners of this company,financial mismanagement they have been unable to cash in on inventories,accounts receivables is way out of control and on the money in the bank they have not even obtained a decent return instead they are still paying excessive interest charges for some outstanding loan.In addition internal control and ethics should not allow any board or management member to indulge in real estate deals whereby the company picks up the tab.The further dilution of our shares by the most stupid arrangement of all,a shareprice over 6 dollars and at the same time selling at 4,25 tops it all.The result has been a share price well under three dollars whilst your direct competitor who does not make money has a value of 2,5 time ours.Commendable is our product line and the workers who have been at work,for our management no bonus whatsoever unless they get this share over 10 dollars within six months.
psThe readers of this board may send a letter on the same lines to the directors.
Re: open letter to the trib board
by: stklord01 12/11/04 06:19 pm
Msg: 52958 of 52960
No PR needed.
If TRIB continues to grow top line and bottom line numbers share price will follow.
@all
Bin schon lange bei TRIB investiert. Seit Oktober gibt es nicht viel Neues. Vielleicht Ende Januar wenn Zahlen kommen. Müßte besser ausfallen als bisher. Der schwache Dollar ist nicht besonders günstig für TRIB es sei denn die verkaufen was das Zeug hält. Bevor ich nichts konkretes weiß poste ich auch nicht also abwarten und Tee trinken oder vielmehr irish coffee.


PS. im Winterhalbjahr laufen die Biotechs immer besser vielleicht auch TRIB.!!!
Bin schon lange bei TRIB investiert. Seit Oktober gibt es nicht viel Neues. Vielleicht Ende Januar wenn Zahlen kommen. Müßte besser ausfallen als bisher. Der schwache Dollar ist nicht besonders günstig für TRIB es sei denn die verkaufen was das Zeug hält. Bevor ich nichts konkretes weiß poste ich auch nicht also abwarten und Tee trinken oder vielmehr irish coffee.



PS. im Winterhalbjahr laufen die Biotechs immer besser vielleicht auch TRIB.!!!
www.trinitybiotech.com
Q4 2004 Trinity Biotech Earnings Release
3rd week February, 2005 (Date to be announced)
Q4 2004 Trinity Biotech Earnings Release
3rd week February, 2005 (Date to be announced)
EL DORADO HILLS, Calif.--(BUSINESS WIRE)--Feb. 1, 2005--J.M. Dutton & Associates continues coverage of Trinity Biotech plc (Nasdaq:TRIB - News), lowering its rating to Buy and a price target of $4.25. The 10-page report by J.M. Dutton senior analyst Stephen Handley is available at www.jmdutton.com, as well as from First Call, Bloomberg, Zacks, Reuters, and other leading financial portals.
Trinity Biotech is a solid and well managed diagnostic products company, and we continue to believe that the stock offers good value at current levels. Nevertheless, we are reducing our rating from Strong Buy to Buy, concurrent with a lowering of our 2004 fully diluted EPS estimate from $0.132 to $0.08, and an expectation that the rebound in 2005 is likely to be less than we previously assumed, although still substantial. The year 2004 was a period of transition, related to the costly expansion of the Company`s U.S. salesforce and a changing relationship with its U.S. distributor (actual results should be reported about mid February). In addition, equity financing early in the year was dilutive and will remain so until revenues increase more significantly from recent and future acquisitions, as well as from the Company`s expanded sales and marketing efforts. There are several factors noted in the report that have caused us to temper our near-term expectations. The uninspiring recent results reflect neither deteriorating industry conditions nor major new competition. Instead, this situation has resulted from management`s decision to fund significant investment spending and to incur dilutive financing in order to position the Company for significant long-term growth and profitability. Therefore, we have stated that the biggest risk in the stock, in our opinion, relates to the uncertain speed with which the Company can return to satisfactory profits, for turnarounds typically take longer than anticipated. Our lowering of near-term expectations in effect demonstrates this frequent pattern.
About Dutton & Associates
Dutton & Associates is one of the largest independent investment research firms in the U.S. Its 28 senior analysts are primarily CFAs, and have expertise in many industries. Dutton & Associates provides continuing analyst coverage of over 85 enrolled companies, and its research, estimates, and ratings are carried in all the major databases serving institutions and online investors.
The cost of enrollment in our one-year continuing research program is US $33,000 prepaid for 4 Research Reports, typically published quarterly, and requisite Research Notes. Dutton & Associates received $46,000 from the Company for 8 Research Reports with coverage commencing on 1/27/2003. The Firm does not accept any equity compensation. Our principals and analysts are prohibited from owning or trading in securities of covered companies. The views expressed in this research report accurately reflect the analyst`s personal views about the subject securities or issuer. Neither the analyst`s compensation nor the compensation received by us is in any way related to the specific ratings or views contained in this research report or note. Please read full disclosures and analyst background at www.jmdutton.com before investing.
Trinity Biotech is a solid and well managed diagnostic products company, and we continue to believe that the stock offers good value at current levels. Nevertheless, we are reducing our rating from Strong Buy to Buy, concurrent with a lowering of our 2004 fully diluted EPS estimate from $0.132 to $0.08, and an expectation that the rebound in 2005 is likely to be less than we previously assumed, although still substantial. The year 2004 was a period of transition, related to the costly expansion of the Company`s U.S. salesforce and a changing relationship with its U.S. distributor (actual results should be reported about mid February). In addition, equity financing early in the year was dilutive and will remain so until revenues increase more significantly from recent and future acquisitions, as well as from the Company`s expanded sales and marketing efforts. There are several factors noted in the report that have caused us to temper our near-term expectations. The uninspiring recent results reflect neither deteriorating industry conditions nor major new competition. Instead, this situation has resulted from management`s decision to fund significant investment spending and to incur dilutive financing in order to position the Company for significant long-term growth and profitability. Therefore, we have stated that the biggest risk in the stock, in our opinion, relates to the uncertain speed with which the Company can return to satisfactory profits, for turnarounds typically take longer than anticipated. Our lowering of near-term expectations in effect demonstrates this frequent pattern.
About Dutton & Associates
Dutton & Associates is one of the largest independent investment research firms in the U.S. Its 28 senior analysts are primarily CFAs, and have expertise in many industries. Dutton & Associates provides continuing analyst coverage of over 85 enrolled companies, and its research, estimates, and ratings are carried in all the major databases serving institutions and online investors.
The cost of enrollment in our one-year continuing research program is US $33,000 prepaid for 4 Research Reports, typically published quarterly, and requisite Research Notes. Dutton & Associates received $46,000 from the Company for 8 Research Reports with coverage commencing on 1/27/2003. The Firm does not accept any equity compensation. Our principals and analysts are prohibited from owning or trading in securities of covered companies. The views expressed in this research report accurately reflect the analyst`s personal views about the subject securities or issuer. Neither the analyst`s compensation nor the compensation received by us is in any way related to the specific ratings or views contained in this research report or note. Please read full disclosures and analyst background at www.jmdutton.com before investing.
Biotechs sind gerade am wieder entdeckt werden. WOllen wir mal hoffen dass auch Trinity davon profitiert und endlich mal wieder steigt.
beim letzten Quartalsbericht ist die Aktie von 3,00 auf 2,50 eingebrochen. In dieser Woche werden die Zahlen zum 4 Quartal 2004 veröffentlicht. Da der US-D schwach war, wird wohl das Ergebnis nicgt blendend ausfallen. Natürlich hoffe ich auf eine positive Überraschung, so dass wird die 3,00 US-D mal nach oben durchbrechen, aber die Tendenz ist leider immer noch abwärts gerichtet.
sieht doch gut aus ,oder

gruß maha
Ich habe gelesen die Q4 Zahlen sollen am Dienstag, den 22. kommen.
http://www.trinitybiotech.com/EN/index.asp
Gruss
vaka
http://www.trinitybiotech.com/EN/index.asp
Gruss
vaka
Trinity Biotech Announces 2004 Results
Revenues Increase 22% DUBLIN, Ireland - PRNewswire-FirstCall - February 22
DUBLIN, Ireland, February 22 /PRNewswire-FirstCall/ -- Trinity Biotech plc today announced results for the quarter and year ended December 31, 2004.
Annual revenues increased 22% from US$65.68 million in 2003 to US$79.94 million in 2004. Q4 revenues, compared to the same period last year, have increased by 42%. Revenues by key product area were as follows :
2003 2004 %
US$000 US$000 Increase
Infectious Disease (excl 30,678 31,638 3%
rapids)
Coagulation 24,435 26,772 10%
Rapids 4,449 9,807 120%
Other 6,113 11,727 92%
Total 65,675 79,944 22%
Revenues by geographical location were as follows :
2003 2004 %
US$000 US$000 Increase
USA 36,299 41,380 14%
Europe 19,983 22,654 13%
Asia / Africa 9,393 15,910 69%
Total 65,675 79,944 22%
Gross profit in the quarter amounted to US$10.93 million, representing a gross margin of 49.6%. The annual gross margin of 50.4% compares to 49.9% in 2003. SG&A expenses have remained relatively constant quarter on quarter at approximately US$7.4 million. The increase in SG&A costs from US$16.69 million in 2003 to US$27.30 million in the current year is primarily attributable to the increased salesforce in the USA.
The net effect of the above factors was a profit after tax of US$1.75 million for the quarter and an EPS of 3.10 US cents. The annual profit after tax was US$5.17 million and annual EPS was 9.37 US cents.
Commenting on the results, Rory Nealon, Chief Financial Officer, said "2004 was a very good year for Trinity in that we increased our sales by 22% to US$80 million and despite increasing our SG&A costs, primarily in the USA, from US$16.7 million to US$27.3 million (an increase which will pay dividends in the future) we still achieved EBITDA of US$10.1 million and a profit after tax of US$5.2 million. The key objectives for our expanded salesforce in the USA are to grow sales of our Unigold rapid HIV test, to significantly increase our market share in the coagulation market, to recover business from our US distributor and to expand our presence in the infectious disease market. To this end the provision of Adaltis instrumentation provides Trinity for the first time with an instrumentation platform which permits us to offer an integrated reagent and instrumentation package for our infectious disease customers. During the year we have also successfully integrated the Fitzgerald operations into the Trinity group and this is reflected in an increase in our Other Sales from US$6.1 million to US$11.7 million."
Ronan O`Caoimh, CEO commented "The strategic goal of Trinity is to attain revenues of at least US$250 million by 2009. Trinity has developed into a geographically diverse diagnostic company with a broad instrumentation and reagent product range in infectious disease, coagulation and point of care testing. We also have an enviable distribution network with direct sales in the USA, Germany and the UK and a wide distributor network in seventy eight countries. The ongoing growth of our business will continue to be focused on our infectious disease, coagulation and rapid product ranges. We will actively seek earnings accretive acquisitions and will focus on delivering improved returns from our existing business."
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in 78 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: http://www.trinitybiotech.com/.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Revenues Increase 22% DUBLIN, Ireland - PRNewswire-FirstCall - February 22
DUBLIN, Ireland, February 22 /PRNewswire-FirstCall/ -- Trinity Biotech plc today announced results for the quarter and year ended December 31, 2004.
Annual revenues increased 22% from US$65.68 million in 2003 to US$79.94 million in 2004. Q4 revenues, compared to the same period last year, have increased by 42%. Revenues by key product area were as follows :
2003 2004 %
US$000 US$000 Increase
Infectious Disease (excl 30,678 31,638 3%
rapids)
Coagulation 24,435 26,772 10%
Rapids 4,449 9,807 120%
Other 6,113 11,727 92%
Total 65,675 79,944 22%
Revenues by geographical location were as follows :
2003 2004 %
US$000 US$000 Increase
USA 36,299 41,380 14%
Europe 19,983 22,654 13%
Asia / Africa 9,393 15,910 69%
Total 65,675 79,944 22%
Gross profit in the quarter amounted to US$10.93 million, representing a gross margin of 49.6%. The annual gross margin of 50.4% compares to 49.9% in 2003. SG&A expenses have remained relatively constant quarter on quarter at approximately US$7.4 million. The increase in SG&A costs from US$16.69 million in 2003 to US$27.30 million in the current year is primarily attributable to the increased salesforce in the USA.
The net effect of the above factors was a profit after tax of US$1.75 million for the quarter and an EPS of 3.10 US cents. The annual profit after tax was US$5.17 million and annual EPS was 9.37 US cents.
Commenting on the results, Rory Nealon, Chief Financial Officer, said "2004 was a very good year for Trinity in that we increased our sales by 22% to US$80 million and despite increasing our SG&A costs, primarily in the USA, from US$16.7 million to US$27.3 million (an increase which will pay dividends in the future) we still achieved EBITDA of US$10.1 million and a profit after tax of US$5.2 million. The key objectives for our expanded salesforce in the USA are to grow sales of our Unigold rapid HIV test, to significantly increase our market share in the coagulation market, to recover business from our US distributor and to expand our presence in the infectious disease market. To this end the provision of Adaltis instrumentation provides Trinity for the first time with an instrumentation platform which permits us to offer an integrated reagent and instrumentation package for our infectious disease customers. During the year we have also successfully integrated the Fitzgerald operations into the Trinity group and this is reflected in an increase in our Other Sales from US$6.1 million to US$11.7 million."
Ronan O`Caoimh, CEO commented "The strategic goal of Trinity is to attain revenues of at least US$250 million by 2009. Trinity has developed into a geographically diverse diagnostic company with a broad instrumentation and reagent product range in infectious disease, coagulation and point of care testing. We also have an enviable distribution network with direct sales in the USA, Germany and the UK and a wide distributor network in seventy eight countries. The ongoing growth of our business will continue to be focused on our infectious disease, coagulation and rapid product ranges. We will actively seek earnings accretive acquisitions and will focus on delivering improved returns from our existing business."
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in 78 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: http://www.trinitybiotech.com/.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except Three Three Three Twelve Twelve
share data) Months Months Months Months Months
Ended Ended Ended Ended Ended
December September December December December
31, 30, 31, 31 31
2004 2004 2003 2004 2003
Revenues 22,048 21,448 15,483 79,944 65,675
Cost of sales (11,119) (10,435) (7,299) (39,688) (32,877)
Gross profit 10,929 11,013 8,184 40,256 32,798
Research &
development
expenses (946) (1,342) (941) (4,641) (5,210)
S,G&A expenses (7,421) (7,101) (4,342) (27,304) (16,690)
Amortisation of
goodwill (816) (814) 13 (2,570) (1,229)
Operating profit 1,746 1,756 2,914 5,741 9,669
Interest
receivable 66 55 116 302 174
Interest payable (207) (184) (241) (824) (793)
Profit on
ordinary
activities before
tax 1,605 1,627 2,789 5,219 9,050
HiberGen
write-off 0 0 (967) 0 (1,067)
Profit on
ordinary
activities before
tax 1,605 1,627 1,822 5,219 7,983
Tax on profit on
ordinary
activities 140 (123) (1,282) (53) (2,186)
Profit after tax 1,745 1,504 540 5,166 5,797
Earnings per
share (US cents) 3.10 2.68 1.21 9.37 13.45
Diluted earnings
per share (US
cents) 2.90 2.54 1.25 8.89 12.14
Weighted average
no. of shares
used in computing
earnings per
share 56,273,201 56,090,236 44,456,089 55,132,024 43,093,146
Prepared in accordance with Irish generally accepted accounting principles.
Trinity Biotech plc
Consolidated Balance Sheet
December 31, 2004 December 31, 2003
US$000`s US$000`s
CURRENT ASSETS
Cash at bank 22,287 20,563
Accounts receivable and prepayments 17,140 14,463
Inventories 37,519 30,555
76,946 65,581
FIXED ASSETS
Tangible assets 15,942 13,660
Intangible assets 57,870 38,851
Financial assets 0 0
73,812 52,511
TOTAL ASSETS 150,758 118,092
LIABILITIES
Accounts payable & accrued expenses 14,496 19,151
Convertible debentures 15,819 13,037
Long term liabilities 4,305 5,643
34,620 37,831
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 764 658
Class "B" ordinary shares 12 12
Share premium account 120,444 87,596
Revenue and other reserves (5,082) (8,005)
116,138 80,261
TOTAL LIABILITIES & SHAREHOLDERS`
EQUITY 150,758 118,092
Prepared in accordance with Irish generally accepted accounting principles.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except Three Three Three Twelve Twelve
share data) Months Months Months Months Months
Ended Ended Ended Ended Ended
December September December December December
31, 30, 31, 31 31
2004 2004 2003 2004 2003
Revenues 22,048 21,448 15,483 79,944 65,675
Cost of sales (11,119) (10,435) (7,299) (39,688) (32,877)
Gross profit 10,929 11,013 8,184 40,256 32,798
Research &
development
expenses (946) (1,342) (941) (4,641) (5,210)
S,G&A expenses (7,421) (7,101) (4,342) (27,304) (16,690)
Amortisation of
goodwill (816) (814) 13 (2,570) (1,229)
Operating profit 1,746 1,756 2,914 5,741 9,669
Interest
receivable 66 55 116 302 174
Interest payable (207) (184) (241) (824) (793)
Profit on
ordinary
activities before
tax 1,605 1,627 2,789 5,219 9,050
HiberGen
write-off 0 0 (967) 0 (1,067)
Profit on
ordinary
activities before
tax 1,605 1,627 1,822 5,219 7,983
Tax on profit on
ordinary
activities 140 (123) (1,282) (53) (2,186)
Profit after tax 1,745 1,504 540 5,166 5,797
Earnings per
share (US cents) 3.10 2.68 1.21 9.37 13.45
Diluted earnings
per share (US
cents) 2.90 2.54 1.25 8.89 12.14
Weighted average
no. of shares
used in computing
earnings per
share 56,273,201 56,090,236 44,456,089 55,132,024 43,093,146
Prepared in accordance with Irish generally accepted accounting principles.
Trinity Biotech plc
Consolidated Balance Sheet
December 31, 2004 December 31, 2003
US$000`s US$000`s
CURRENT ASSETS
Cash at bank 22,287 20,563
Accounts receivable and prepayments 17,140 14,463
Inventories 37,519 30,555
76,946 65,581
FIXED ASSETS
Tangible assets 15,942 13,660
Intangible assets 57,870 38,851
Financial assets 0 0
73,812 52,511
TOTAL ASSETS 150,758 118,092
LIABILITIES
Accounts payable & accrued expenses 14,496 19,151
Convertible debentures 15,819 13,037
Long term liabilities 4,305 5,643
34,620 37,831
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 764 658
Class "B" ordinary shares 12 12
Share premium account 120,444 87,596
Revenue and other reserves (5,082) (8,005)
116,138 80,261
TOTAL LIABILITIES & SHAREHOLDERS`
EQUITY 150,758 118,092
Prepared in accordance with Irish generally accepted accounting principles.
Trinity Biotech plc
das Ergebnis ist das. Zum Glück keine negative Überraschung. Die Umsatzsteigerung im IV Quartal ist gut, aber der Gewinn stagniert immer noch.
http://biz.yahoo.com/prnews/050222/uktu018_1.html
Die wichtigsten Facts:
Annual revenues increased 22% from US$65.68 million in 2003 to US$79.94 million in 2004. The annual profit after tax was US$5.17 million and annual EPS was 9.37 US cents.
Q4 revenues, compared to the same period last year, have increased by 42%. Revenues by key product area were as follows. The net effect of the above factors was a profit after tax of US$1.75 million for the quarter and an EPS of 3.10 US cents
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except Three Three Three Twelve Twelve
share data) Months Months Months Months Months
Ended Ended Ended Ended Ended
December September December December December
31, 30, 31, 31 31
2004 2004 2003 2004 2003
Revenues 22,048 21,448 15,483 79,944 65,675
Cost of sales (11,119) (10,435) (7,299) (39,688) (32,877)
Gross profit 10,929 11,013 8,184 40,256 32,798
Research &
development
expenses (946) (1,342) (941) (4,641) (5,210)
S,G&A expenses (7,421) (7,101) (4,342) (27,304) (16,690)
Amortisation of
goodwill (816) (814) 13 (2,570) (1,229)
Operating profit 1,746 1,756 2,914 5,741 9,669
Interest
receivable 66 55 116 302 174
Interest payable (207) (184) (241) (824) (793)
Profit on
ordinary
activities before
tax 1,605 1,627 2,789 5,219 9,050
HiberGen
write-off 0 0 (967) 0 (1,067)
Profit on
ordinary
activities before
tax 1,605 1,627 1,822 5,219 7,983
Tax on profit on
ordinary
activities 140 (123) (1,282) (53) (2,186)
Profit after tax 1,745 1,504 540 5,166 5,797
Earnings per
share (US cents) 3.10 2.68 1.21 9.37 13.45
Diluted earnings
per share (US
cents) 2.90 2.54 1.25 8.89 12.14
http://biz.yahoo.com/prnews/050222/uktu018_1.html
Die wichtigsten Facts:
Annual revenues increased 22% from US$65.68 million in 2003 to US$79.94 million in 2004. The annual profit after tax was US$5.17 million and annual EPS was 9.37 US cents.
Q4 revenues, compared to the same period last year, have increased by 42%. Revenues by key product area were as follows. The net effect of the above factors was a profit after tax of US$1.75 million for the quarter and an EPS of 3.10 US cents
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except Three Three Three Twelve Twelve
share data) Months Months Months Months Months
Ended Ended Ended Ended Ended
December September December December December
31, 30, 31, 31 31
2004 2004 2003 2004 2003
Revenues 22,048 21,448 15,483 79,944 65,675
Cost of sales (11,119) (10,435) (7,299) (39,688) (32,877)
Gross profit 10,929 11,013 8,184 40,256 32,798
Research &
development
expenses (946) (1,342) (941) (4,641) (5,210)
S,G&A expenses (7,421) (7,101) (4,342) (27,304) (16,690)
Amortisation of
goodwill (816) (814) 13 (2,570) (1,229)
Operating profit 1,746 1,756 2,914 5,741 9,669
Interest
receivable 66 55 116 302 174
Interest payable (207) (184) (241) (824) (793)
Profit on
ordinary
activities before
tax 1,605 1,627 2,789 5,219 9,050
HiberGen
write-off 0 0 (967) 0 (1,067)
Profit on
ordinary
activities before
tax 1,605 1,627 1,822 5,219 7,983
Tax on profit on
ordinary
activities 140 (123) (1,282) (53) (2,186)
Profit after tax 1,745 1,504 540 5,166 5,797
Earnings per
share (US cents) 3.10 2.68 1.21 9.37 13.45
Diluted earnings
per share (US
cents) 2.90 2.54 1.25 8.89 12.14
Habt Ihr keine Meinung nach den Zahlen?
Würde mich schon interessieren was Ihr darüber denkt.
Wie sieht Eure Einschätzung aus?
Wenn ich Zeit finde werde ich am Wochenende mal schreiben was ich die nächsten Jahre erwarte.
Gruss
vaka
Würde mich schon interessieren was Ihr darüber denkt.
Wie sieht Eure Einschätzung aus?
Wenn ich Zeit finde werde ich am Wochenende mal schreiben was ich die nächsten Jahre erwarte.
Gruss
vaka
für das Jahr 2005 habe ich eine Analystenschätzung gelesen Gewinn 0,12 US-D und Kursziel 3,00 US-D. Das Umsatzwachstum ist gut, aber solange die Gewinne bei 0,03 Cent je Quartal stagnieren, wird auch der Kurs nicht steigen. Ich bin jetzt mal Optimist und versuche folgende Erwartung:
Umsatz 90 Mio
Gewinn 7,5 Mio
Gewinn ca. 0,12-0,15
Es müßte ein Aktien-Rückkauf beschlossen werden, damit weitere Übernahme daraus erfolgen können. Dann würden auch die Gewinne je Aktie im gleichen Maße oder schneller wachsen. Daher ist die Aktie im Bereich 3,00 fair bewertet und bei 4,50 US-D spekulativ gut bewertet.
für 2006
Umsatz 100 Mio
Gewinn 9 Mio
Gewinn ca. 0,16-0,20
Dann könnte spekulativ ein Kurs um 6,00 möglich sein. Meine Schätzung ist bestimmt sehr vorsichtig, denn in der Meldung heißt es:
Ronan O`Caoimh, CEO commented " The strategic goal of Trinity is to attain revenues of at least US$250 million by 2009.
Wenn ich die Ergebnisse von 80 Mio Umsatz ausgehend gleichmäßig verteile bis 2009, dann würde das folgende Umsatzentwicklung bedeuten:
pro Jahr 34 Mio Zuwachs
2005 Mio 114
2006 Mio 148
2007 Mio 182
2008 Mio 216
2009 Mio 250
aus organischen Wachstum kann das nicht erzeugt werden. Da man den genauen Zeitpunkt von Übernahmen nicht ermessen kann, wird es sicher keinen gleichbleibenden Zuwachs, wie oben berechnet, geben. Wenn Trib nur nur in diese Nähe der Schätzungen kommt, dann würde es schon bald neue Höchststände geben. Hoffentlich steigt mal der Dollar. Das würde dem Gewinn ordentlich auf die Sprünge helfen.
Umsatz 90 Mio
Gewinn 7,5 Mio
Gewinn ca. 0,12-0,15
Es müßte ein Aktien-Rückkauf beschlossen werden, damit weitere Übernahme daraus erfolgen können. Dann würden auch die Gewinne je Aktie im gleichen Maße oder schneller wachsen. Daher ist die Aktie im Bereich 3,00 fair bewertet und bei 4,50 US-D spekulativ gut bewertet.
für 2006
Umsatz 100 Mio
Gewinn 9 Mio
Gewinn ca. 0,16-0,20
Dann könnte spekulativ ein Kurs um 6,00 möglich sein. Meine Schätzung ist bestimmt sehr vorsichtig, denn in der Meldung heißt es:
Ronan O`Caoimh, CEO commented " The strategic goal of Trinity is to attain revenues of at least US$250 million by 2009.
Wenn ich die Ergebnisse von 80 Mio Umsatz ausgehend gleichmäßig verteile bis 2009, dann würde das folgende Umsatzentwicklung bedeuten:
pro Jahr 34 Mio Zuwachs
2005 Mio 114
2006 Mio 148
2007 Mio 182
2008 Mio 216
2009 Mio 250
aus organischen Wachstum kann das nicht erzeugt werden. Da man den genauen Zeitpunkt von Übernahmen nicht ermessen kann, wird es sicher keinen gleichbleibenden Zuwachs, wie oben berechnet, geben. Wenn Trib nur nur in diese Nähe der Schätzungen kommt, dann würde es schon bald neue Höchststände geben. Hoffentlich steigt mal der Dollar. Das würde dem Gewinn ordentlich auf die Sprünge helfen.
Hallo Dangerous
Könnte #515 fast so unterschreiben, ich sehe das ähnlich.
Bei nasdaq.com liegen die Analystenschätzungen für 2005 bei
0,16 und für 2006 bei 0,27 EPS.
12 Month Price Target Range liegt bei 4,62 USD.
Die 0,27 EPS halte ich für wenig wahrscheinlich.
Allerdings hat das Management eine Menge Druck bekommen um die Gewinnsituation zu verbessern. Vielleicht ist da doch eine Überraschung drin.
Das Umsatzziel von 250 Mio USD bis 2009 kann nur durch Übernahmen erreicht werden. Aus organischen Wachstum könnten bis 2009 wahrscheinlich nicht mehr wie ca. 150 Mio. erreicht werden. Deswegen ist in der Planung laut Aussage vom Management eine ( oder mehrere ) große Aquisition.
Ein Aktienrückkaufprogramm kann ich mir bei den sturen Iren nicht vorstellen. Bei denen geht es nur um cash, momentan 22 Mio. auf der Bank.
Die Frage ist wann folgt eine größere Übernahme und der Umsatzsprung, bald oder erst in 2-3 Jahren? Wenn man das wüßte könnte man den Aktienkurs präziser hochrechnen.
Ich sehe Trinity nicht als Zock, sondern wirklich langfristig die nächsten Jahre.
Wenn wir bis 2009 einen Aktienkurs von 8 Dollar haben sind das von heute 200%. Mehr kann ich nicht erwarten von einer soliden Firma. Wenn die 250 Mio USD erreicht werden und die Gewinne bewegen sich im vernünftigen Rahmen hoffe ich auf einen Aktienkurs höher als 10 USD.
Für mich also unter 3 USD klare Kaufkurse.
Gruss
vaka
Könnte #515 fast so unterschreiben, ich sehe das ähnlich.
Bei nasdaq.com liegen die Analystenschätzungen für 2005 bei
0,16 und für 2006 bei 0,27 EPS.
12 Month Price Target Range liegt bei 4,62 USD.
Die 0,27 EPS halte ich für wenig wahrscheinlich.
Allerdings hat das Management eine Menge Druck bekommen um die Gewinnsituation zu verbessern. Vielleicht ist da doch eine Überraschung drin.
Das Umsatzziel von 250 Mio USD bis 2009 kann nur durch Übernahmen erreicht werden. Aus organischen Wachstum könnten bis 2009 wahrscheinlich nicht mehr wie ca. 150 Mio. erreicht werden. Deswegen ist in der Planung laut Aussage vom Management eine ( oder mehrere ) große Aquisition.
Ein Aktienrückkaufprogramm kann ich mir bei den sturen Iren nicht vorstellen. Bei denen geht es nur um cash, momentan 22 Mio. auf der Bank.
Die Frage ist wann folgt eine größere Übernahme und der Umsatzsprung, bald oder erst in 2-3 Jahren? Wenn man das wüßte könnte man den Aktienkurs präziser hochrechnen.
Ich sehe Trinity nicht als Zock, sondern wirklich langfristig die nächsten Jahre.
Wenn wir bis 2009 einen Aktienkurs von 8 Dollar haben sind das von heute 200%. Mehr kann ich nicht erwarten von einer soliden Firma. Wenn die 250 Mio USD erreicht werden und die Gewinne bewegen sich im vernünftigen Rahmen hoffe ich auf einen Aktienkurs höher als 10 USD.
Für mich also unter 3 USD klare Kaufkurse.
Gruss
vaka
Hallo Vaka da die Kurse täglich hin und her pendeln, wissen wohl viele Anlager nicht, wie das Ergebnis zu bewerten ist. Richtigen Aufschluß werden wir wohl erst mit den Zahlen zum I. Quartal 2005 bekommen. Bleibt nur abzuwarten und zu hoffen, dass der Kurs nicht unter 2,50 abkippt. Vielen Dank für deine Antwort. Ich kann deine Meinung nur unterstreichen.
Ungewöhnlich hohes Volumen die letzten Tage an der Nasdaq,
teilweise große Blöcke von 35k und 75 k.
Hat jemand von Euch irgendwas erfahren?
gruss
vaka
teilweise große Blöcke von 35k und 75 k.
Hat jemand von Euch irgendwas erfahren?
gruss
vaka
Trinity Biotech Completes Acquisition of Research Diagnostics Inc
DUBLIN, Ireland - PRNewswire-FirstCall - March 21
DUBLIN, Ireland, March 21 /PRNewswire-FirstCall/ -- Trinity Biotech plc today announced that it has completed the acquisition of Research Diagnostics Inc. ("RDI"), of Flanders, New Jersey.
Trinity Biotech has acquired the business of RDI for US$4.2 million in cash. RDI provides a comprehensive range of immunodiagnostic products to research facilities, pharmaceutical companies, reference laboratories, diagnostic manufacturers, and universities worldwide. The Company`s revenue and profit performance has shown consistent growth with sales last year in excess of US$5 million. The acquisition is expected to be accretive in the current financial year.
The range of products provided by RDI is similar to that provided by Fitzgeralds, a company which Trinity acquired in April 2004. Specifically the range includes monoclonal and polyclonal antibodies, antigens, proteins, enzymes and immunochemicals employed in the areas of cancer, cardiac, fertility and infectious disease diagnosis. The operations of RDI will be integrated with those of Fitzgeralds in the coming months and the combined entity will operate on a stand alone basis within the Trinity group.
Ronan O`Caoimh, Trinity Biotech CEO, commented "We are delighted to be deploying our cash resources to this earnings accretive acquisition. RDI is a natural fit with our existing operations in Fitzgerald in that it is particularly strong in supplying product to research facilities worldwide whereas Fitzgerald`s focus is on diagnostic manufacturers. There is minimal overlap in the products supplied by both companies. We are confident that the combination of both RDI and Fitzgerald will strengthen Trinity`s position in this segment of the life sciences market with combined turnover in excess of US$11 million."
Edward Fitzgerald, Chairman Fitzgerald Industries International, commented "The acquisition of RDI by Trinity will provide Fitzgeralds with access to an alternative customer base and exciting new products for supply to existing Fitzgerald customers. We are very excited at the opportunities this will present to continue the growth profile of our business and to position Fitzgerald and RDI as leading suppliers to the life sciences market."
Trinity Biotech develops, acquires manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own salesforces and a network of international distributors and strategic partners. For further information please see the company`s website: http://www.trinitybiotech.com/.
DUBLIN, Ireland - PRNewswire-FirstCall - March 21
DUBLIN, Ireland, March 21 /PRNewswire-FirstCall/ -- Trinity Biotech plc today announced that it has completed the acquisition of Research Diagnostics Inc. ("RDI"), of Flanders, New Jersey.
Trinity Biotech has acquired the business of RDI for US$4.2 million in cash. RDI provides a comprehensive range of immunodiagnostic products to research facilities, pharmaceutical companies, reference laboratories, diagnostic manufacturers, and universities worldwide. The Company`s revenue and profit performance has shown consistent growth with sales last year in excess of US$5 million. The acquisition is expected to be accretive in the current financial year.
The range of products provided by RDI is similar to that provided by Fitzgeralds, a company which Trinity acquired in April 2004. Specifically the range includes monoclonal and polyclonal antibodies, antigens, proteins, enzymes and immunochemicals employed in the areas of cancer, cardiac, fertility and infectious disease diagnosis. The operations of RDI will be integrated with those of Fitzgeralds in the coming months and the combined entity will operate on a stand alone basis within the Trinity group.
Ronan O`Caoimh, Trinity Biotech CEO, commented "We are delighted to be deploying our cash resources to this earnings accretive acquisition. RDI is a natural fit with our existing operations in Fitzgerald in that it is particularly strong in supplying product to research facilities worldwide whereas Fitzgerald`s focus is on diagnostic manufacturers. There is minimal overlap in the products supplied by both companies. We are confident that the combination of both RDI and Fitzgerald will strengthen Trinity`s position in this segment of the life sciences market with combined turnover in excess of US$11 million."
Edward Fitzgerald, Chairman Fitzgerald Industries International, commented "The acquisition of RDI by Trinity will provide Fitzgeralds with access to an alternative customer base and exciting new products for supply to existing Fitzgerald customers. We are very excited at the opportunities this will present to continue the growth profile of our business and to position Fitzgerald and RDI as leading suppliers to the life sciences market."
Trinity Biotech develops, acquires manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own salesforces and a network of international distributors and strategic partners. For further information please see the company`s website: http://www.trinitybiotech.com/.
komisch sind immer die deutlich anziehenden Kurse nachbörslich. Im Börsenhandel wird ein Amstieg zur Eröffnung bei ca. 2,80-2,70 nach unten abverkauft. Obwohl hier übernommende Aktien abstoßen und damit das nicht so deutlich wird, werden die Pakete außerbörslich verschoben?
Hallo!
Ich möchte bezüglich Trinity nur allgemein in den Raum stellen, wie es mit einer Übernahme aussieht?
Eigentlich verwundert es mich, dass sich nocht kein `Grosser` für Trinity interessiert, da ich denke, dass Trinity wohl für so manchen Multi eine positive Ergänzung wäre.
Ein positive Ergänzung wohl nicht zu einem ausgesprochenem Schnäppchenpreis, doch wurden bereits deutlich höhere Kurse bei Übernahmen bezahlt.
Gruss.
Ich möchte bezüglich Trinity nur allgemein in den Raum stellen, wie es mit einer Übernahme aussieht?
Eigentlich verwundert es mich, dass sich nocht kein `Grosser` für Trinity interessiert, da ich denke, dass Trinity wohl für so manchen Multi eine positive Ergänzung wäre.
Ein positive Ergänzung wohl nicht zu einem ausgesprochenem Schnäppchenpreis, doch wurden bereits deutlich höhere Kurse bei Übernahmen bezahlt.
Gruss.
Hallo Mant,
ich denke dafür ist trinity noch zu klein. Die nächsten Jahre wird trib noch ein paar Firmen übernehmen. Wer weiß wie das in ein paar Jahren aussieht..
Q1 2005 Trinity Biotech Earnings Release
28 April 2005 (tentative)
http://www.trinitybiotech.com/EN/index.asp
vaka
ich denke dafür ist trinity noch zu klein. Die nächsten Jahre wird trib noch ein paar Firmen übernehmen. Wer weiß wie das in ein paar Jahren aussieht..
Q1 2005 Trinity Biotech Earnings Release
28 April 2005 (tentative)
http://www.trinitybiotech.com/EN/index.asp
vaka
...das ist hier wirklich ein Drama...wenn man den Langzeitchart ansieht, dann könnte es glatt bis 1 USD runtergehen?!? 
Habe heute entnervt aufgegeben und warte auf einen besseren Einstieg

Habe heute entnervt aufgegeben und warte auf einen besseren Einstieg

ja spinn i? Heute schon bei 1,91 USD. 

Ich habe keine Ahnung warum der Kurs so fällt.
Im Yahoo Board sind auch alle ahnungslos, es werden nur die üblichen Spekulationen angestellt.
Ich habe vor 2 Wochen noch Aktien nachgekauft, mit weiteren Käufen werde ich aber erstmal abwarten, die Charttechnik sieht nicht gerade gut aus. Kennt sich jemand damit aus?
Auf jedenfall haben wir heute mit 2.02 USD den Buchwert erreicht. Und 22 Mio auf der Bank
2002 hatten wir das schonmal. Es sollte irgendwann wieder drehen.
Die Q1 Zahlen sollen am 28.4 kommen, die sollten eigentlich besser aussehen.
Gruss
vaka
Balance Sheet
Total Cash (mrq): 22.29M
Total Cash Per Share (mrq): 0.48
Total Debt (mrq)²: 15.82M
Total Debt/Equity (mrq): 0.136
Current Ratio (mrq): 5.308
Book Value Per Share (mrq): 2.02
Im Yahoo Board sind auch alle ahnungslos, es werden nur die üblichen Spekulationen angestellt.
Ich habe vor 2 Wochen noch Aktien nachgekauft, mit weiteren Käufen werde ich aber erstmal abwarten, die Charttechnik sieht nicht gerade gut aus. Kennt sich jemand damit aus?
Auf jedenfall haben wir heute mit 2.02 USD den Buchwert erreicht. Und 22 Mio auf der Bank

2002 hatten wir das schonmal. Es sollte irgendwann wieder drehen.
Die Q1 Zahlen sollen am 28.4 kommen, die sollten eigentlich besser aussehen.
Gruss
vaka
Balance Sheet
Total Cash (mrq): 22.29M
Total Cash Per Share (mrq): 0.48
Total Debt (mrq)²: 15.82M
Total Debt/Equity (mrq): 0.136
Current Ratio (mrq): 5.308
Book Value Per Share (mrq): 2.02
Hat da vielleicht einer etwas zuviel Insiderwissen,
oder warum wird der Kurs so geprügelt????
 - charttechnisch sieht es wirklich nicht gut aus und es gibt bisher keinerlei Nachrichten, Vermutungen???
- charttechnisch sieht es wirklich nicht gut aus und es gibt bisher keinerlei Nachrichten, Vermutungen???
Würde mich freuen hier etwas über die Gründe des Absturzes zu erfahren,
Danke
oder warum wird der Kurs so geprügelt????

 - charttechnisch sieht es wirklich nicht gut aus und es gibt bisher keinerlei Nachrichten, Vermutungen???
- charttechnisch sieht es wirklich nicht gut aus und es gibt bisher keinerlei Nachrichten, Vermutungen???Würde mich freuen hier etwas über die Gründe des Absturzes zu erfahren,
Danke
da werden wahrscheinlich die Q-Zahlen enttüschend sein und die Insider verkaufen vorher. Am 28.04 wissen wir mehr.
Ich weiß nicht ob Ihr das mitverfolgt, aber in den letzten Wochen wird zum Schluß immer ins BID verkauft um den Kurs zu drücken. Ist schon fast so wie an der otc.
Ich glaube eigentlich nicht an schlechte Q Zahlen, der Kursrückgang könnte auch andere Gründe haben.
Ich habe nochmal die letzten Umsätze aufgeführt.
2.14 200 16:00:02
2.19 200 15:59:03
2.18 100 15:59:01
2.18 300 15:59:01
2.16 1600 15:57:06
2.18 14452 15:56:41
2.18 700 15:56:37
2.18 200 15:56:36
2.18 100 15:56:36
2.18 700 15:56:18
2.18 100 15:56:18
2.16 2000 15:56:16
2.18 1100 15:56:11
2.18 100 15:56:07
2.18 100 15:56:05
2.18 100 15:55:45
2.18 1500 15:54:53
2.18 300 15:54:53
2.18 100 15:54:53
2.18 1800 15:54:53
2.18 100 15:54:26
2.17 500 15:53:31
2.17 1000 15:53:31
2.17 100 15:53:31
2.17 1000 15:53:31
2.16 148 15:53:29
2.16 100 15:53:29
2.16 100 15:53:25
2.17 700 15:52:38
2.16 1000 15:52:38
Ich glaube eigentlich nicht an schlechte Q Zahlen, der Kursrückgang könnte auch andere Gründe haben.
Ich habe nochmal die letzten Umsätze aufgeführt.
2.14 200 16:00:02
2.19 200 15:59:03
2.18 100 15:59:01
2.18 300 15:59:01
2.16 1600 15:57:06
2.18 14452 15:56:41
2.18 700 15:56:37
2.18 200 15:56:36
2.18 100 15:56:36
2.18 700 15:56:18
2.18 100 15:56:18
2.16 2000 15:56:16
2.18 1100 15:56:11
2.18 100 15:56:07
2.18 100 15:56:05
2.18 100 15:55:45
2.18 1500 15:54:53
2.18 300 15:54:53
2.18 100 15:54:53
2.18 1800 15:54:53
2.18 100 15:54:26
2.17 500 15:53:31
2.17 1000 15:53:31
2.17 100 15:53:31
2.17 1000 15:53:31
2.16 148 15:53:29
2.16 100 15:53:29
2.16 100 15:53:25
2.17 700 15:52:38
2.16 1000 15:52:38
Trinity Biotech Announces Quarter 1 Results
Revenues Increase 29% DUBLIN, Ireland - PRNewswire-FirstCall - April 28
DUBLIN, Ireland, April 28 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ:TRIB) today announced results for the quarter ended March 31, 2005.
Revenues in the quarter increased 29.4% to US$21.2 million compared to US$16.4 million in the same period last year. Revenues by key product area were as follows :
Q1 2004 Q1 2005 % Increase
US$000 US$000
Infectious Disease 5,978 7,570 26.6
Coagulation 6,704 7,325 9.3
POC 1,821 3,026 66.2
Other 1,906 3,306 73.5
Total 16,409 21,227 29.4
Revenues by geographical location were as follows :
Q1 2004 Q1 2005 % Increase
US$000 US$000
USA 7,926 10,812 36.4
Europe 5,408 6,017 11.3
Asia / Africa 3,075 4,398 43.0
Total 16,409 21,227 29.4
Gross profit amounted to US$10.4 million, representing a gross margin of 49.0%. This compares to a gross profit of US$8.0 million and a gross margin of 48.9% in the same period last year.
R&D expenses have increased from US$1.2 million in Quarter 1 last year to US$1.3 million in the current quarter. The increase in SG&A costs from US$5.7 million in Quarter 1 last year to US$7.2 million in the current year is primarily attributable to the increased salesforce in the USA and increased costs associated with the recent Adaltis and Fitzgerald acquisitions which were acquired since Quarter 1 last year. However SG&A expenses have remained relatively constant quarter on quarter at US$7.2 million.
The net effect of the above factors was an operating profit of US$1.44 million for the quarter as compared to US$1.1 million in the same period last year and an EPS of 1.8 US cents.
Commenting on the results, Rory Nealon, Chief Financial Officer, said "Our quarterly revenues are showing strong growth in all segments and we are particularly pleased with the 66% organic growth in Point of Care which is almost entirely composed of sales of HIV. Overall the increase in our sales would have been greater except for the fact that we are experiencing an unusually quiet flu season.
Our business in the US has increased by 36% over the same period last year due to a combination of increased revenues generated by our expanded salesforce and the impact of the our acquisitions last year."
Ronan O`Caoimh, CEO commented, "At the end of the quarter we completed the acquisition of Research Diagnostics Inc ("RDI"). RDI will be integrated into the operations of Fitzgerald in the coming months and we anticipate that the operation will be earnings positive post integration. RDI is a natural fit with our existing operations in Fitzgerald in that it is particularly strong in supplying product to research facilities worldwide whereas Fitzgerald`s focus is on diagnostic manufacturers.
We are pleased with the organic growth exhibited in all four business divisions. Unigold HIV sales continue to grow both in the US and Africa while instrument placements in both coagulation and infectious disease are gathering momentum. These factors will drive growth in the coming quarters.
The Company today filed a US$75 million shelf registration statement with the SEC. The Company has current cash balances of US$17.2 million and is not in due diligence on any potential acquisition. We therefore have no current intention of issuing equity. However, we have filed the shelf registration statement in order that, in the event of issuing equity, we will issue registered as opposed to unregistered shares - thereby reducing the requirement to issue shares at a discount to the market price at the time. As stated in the past Trinity`s goal is to achieve revenues of US$250 million by 2009 through a combination of organic growth and acquisitions. This shelf registration equips us better to achieve that goal."
Revenues Increase 29% DUBLIN, Ireland - PRNewswire-FirstCall - April 28
DUBLIN, Ireland, April 28 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ:TRIB) today announced results for the quarter ended March 31, 2005.
Revenues in the quarter increased 29.4% to US$21.2 million compared to US$16.4 million in the same period last year. Revenues by key product area were as follows :
Q1 2004 Q1 2005 % Increase
US$000 US$000
Infectious Disease 5,978 7,570 26.6
Coagulation 6,704 7,325 9.3
POC 1,821 3,026 66.2
Other 1,906 3,306 73.5
Total 16,409 21,227 29.4
Revenues by geographical location were as follows :
Q1 2004 Q1 2005 % Increase
US$000 US$000
USA 7,926 10,812 36.4
Europe 5,408 6,017 11.3
Asia / Africa 3,075 4,398 43.0
Total 16,409 21,227 29.4
Gross profit amounted to US$10.4 million, representing a gross margin of 49.0%. This compares to a gross profit of US$8.0 million and a gross margin of 48.9% in the same period last year.
R&D expenses have increased from US$1.2 million in Quarter 1 last year to US$1.3 million in the current quarter. The increase in SG&A costs from US$5.7 million in Quarter 1 last year to US$7.2 million in the current year is primarily attributable to the increased salesforce in the USA and increased costs associated with the recent Adaltis and Fitzgerald acquisitions which were acquired since Quarter 1 last year. However SG&A expenses have remained relatively constant quarter on quarter at US$7.2 million.
The net effect of the above factors was an operating profit of US$1.44 million for the quarter as compared to US$1.1 million in the same period last year and an EPS of 1.8 US cents.
Commenting on the results, Rory Nealon, Chief Financial Officer, said "Our quarterly revenues are showing strong growth in all segments and we are particularly pleased with the 66% organic growth in Point of Care which is almost entirely composed of sales of HIV. Overall the increase in our sales would have been greater except for the fact that we are experiencing an unusually quiet flu season.
Our business in the US has increased by 36% over the same period last year due to a combination of increased revenues generated by our expanded salesforce and the impact of the our acquisitions last year."
Ronan O`Caoimh, CEO commented, "At the end of the quarter we completed the acquisition of Research Diagnostics Inc ("RDI"). RDI will be integrated into the operations of Fitzgerald in the coming months and we anticipate that the operation will be earnings positive post integration. RDI is a natural fit with our existing operations in Fitzgerald in that it is particularly strong in supplying product to research facilities worldwide whereas Fitzgerald`s focus is on diagnostic manufacturers.
We are pleased with the organic growth exhibited in all four business divisions. Unigold HIV sales continue to grow both in the US and Africa while instrument placements in both coagulation and infectious disease are gathering momentum. These factors will drive growth in the coming quarters.
The Company today filed a US$75 million shelf registration statement with the SEC. The Company has current cash balances of US$17.2 million and is not in due diligence on any potential acquisition. We therefore have no current intention of issuing equity. However, we have filed the shelf registration statement in order that, in the event of issuing equity, we will issue registered as opposed to unregistered shares - thereby reducing the requirement to issue shares at a discount to the market price at the time. As stated in the past Trinity`s goal is to achieve revenues of US$250 million by 2009 through a combination of organic growth and acquisitions. This shelf registration equips us better to achieve that goal."
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except share data) Three Months Three Months
Ended Ended
March 31, March 31,
2005 2004
Unaudited Unaudited
Revenues 21,227 16,409
Cost of sales (10,823) (8,372)
Gross profit 10,404 8,037
Research & development expenses (1,320) (1,177)
S,G&A expenses (7,212) (5,705)
Amortisation of intangibles (434) (47)
Operating profit 1,438 1,108
Share option expense (212) (47)
Interest receivable 113 116
Interest payable (238) (207)
Profit on ordinary activities before tax 1,101 970
Tax on profit on ordinary activities (98) 32
Profit after tax 1,003 1,002
Earnings per share (US cents) 1.8 1.9
Diluted earnings per share (US cents) 1.8 1.8
Weighted average no. of shares used in
computing earnings per share 56,954,314 52,560,726
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
Consolidated Balance Sheet
March 31, 2005 December 31, 2004
US$000`s US$000`s
Unaudited Unaudited
CURRENT ASSETS
Cash and cash equivalents 17,177 22,287
Accounts receivable and prepayments 15,263 13,588
Inventories 36,874 37,519
Current tax 697 697
70,011 74,091
NON CURRENT ASSETS
Tangible assets 16,106 15,942
Intangible assets 67,470 62,884
Deferred tax asset 2,302 2,402
Other assets 1,271 1,330
87,149 82,558
TOTAL ASSETS 157,160 156,649
CURRENT LIABILITIES
Short-term financial liabilities 10,825 11,087
Accounts payable & accrued expenses 8,916 8,665
Current tax 644 542
20,385 20,294
NON CURRENT LIABILITIES
Long-term financial liabilities 10,728 12,923
Deferred tax liability and other 3,426 3,558
taxes
14,154 16,481
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 772 764
Class "B" ordinary shares 12 12
Share premium account 122,513 120,444
Currency adjustment (4,349) (3,975)
Revenue and other reserves 3,673 2,629
122,621 119,874
TOTAL LIABILITIES & SHAREHOLDERS` EQUITY 157,160 156,649
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except share data) Three Months Three Months
Ended Ended
March 31, March 31,
2005 2004
Unaudited Unaudited
Revenues 21,227 16,409
Cost of sales (10,823) (8,372)
Gross profit 10,404 8,037
Research & development expenses (1,320) (1,177)
S,G&A expenses (7,212) (5,705)
Amortisation of intangibles (434) (47)
Operating profit 1,438 1,108
Share option expense (212) (47)
Interest receivable 113 116
Interest payable (238) (207)
Profit on ordinary activities before tax 1,101 970
Tax on profit on ordinary activities (98) 32
Profit after tax 1,003 1,002
Earnings per share (US cents) 1.8 1.9
Diluted earnings per share (US cents) 1.8 1.8
Weighted average no. of shares used in
computing earnings per share 56,954,314 52,560,726
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
Consolidated Balance Sheet
March 31, 2005 December 31, 2004
US$000`s US$000`s
Unaudited Unaudited
CURRENT ASSETS
Cash and cash equivalents 17,177 22,287
Accounts receivable and prepayments 15,263 13,588
Inventories 36,874 37,519
Current tax 697 697
70,011 74,091
NON CURRENT ASSETS
Tangible assets 16,106 15,942
Intangible assets 67,470 62,884
Deferred tax asset 2,302 2,402
Other assets 1,271 1,330
87,149 82,558
TOTAL ASSETS 157,160 156,649
CURRENT LIABILITIES
Short-term financial liabilities 10,825 11,087
Accounts payable & accrued expenses 8,916 8,665
Current tax 644 542
20,385 20,294
NON CURRENT LIABILITIES
Long-term financial liabilities 10,728 12,923
Deferred tax liability and other 3,426 3,558
taxes
14,154 16,481
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 772 764
Class "B" ordinary shares 12 12
Share premium account 122,513 120,444
Currency adjustment (4,349) (3,975)
Revenue and other reserves 3,673 2,629
122,621 119,874
TOTAL LIABILITIES & SHAREHOLDERS` EQUITY 157,160 156,649
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
hmmmm..."However, we have filed the shelf registration statement in order that, in the event of issuing equity, we will issue registered as opposed to unregistered shares - thereby reducing the requirement to issue shares at a discount to the market price at the time".... was meinen sie bloß damit? Klingt nicht so sehr vertrauenwürdig...weiß jemand mehr?
wieder das gleiche Bild. Umsätze steigen deutlich, aber die Gewinne stagnieren. Wer füllt sich da die Taschen? Vorstandsgehälter? Die Gewinnsituation ist frustrierend. So wird die Aktie nie steigen und die 2,00 Dollar die bisher charttechnisch nie eine Rolle gespielt haben, werden wohl auch bald fallen! Bei einem KGV von 10 also Kurs 1,20 US-D wird wohl der Boden sein?
[posting]16.482.294 von ehrenwort am 28.04.05 18:37:25[/posting]Lest mal im Yahoo Board, da kann man vieles erfahren.
Geht wieder hoch her heute.
z.b folgendes von ruff
gruss vaka
Re: Shelf Registration
by: ruffneck28 04/28/05 07:03 am
Msg: 55504 of 55609
It depends ....
we already know the goal is 250 million revenues and they can t reach it by organic growth only. So further financing will be needed in the future and theirfore the shelf reg.
If they do additional PP s leaving the money at bank unused for a long time again we have dilution of course.
If the money is invested in an accretive aquisition at the same time again it depends on the price revenues and profit of the aquired company.
Actually the money of the last PP is still not invested completely and they have additional bank facilities they can use for a possible bigger aquisition while I am not aware of such a deal and it seems is getting more and more difficult for them to work out the bigger the to be aquired companies are.
Ruff
Posted as a reply to: Msg 55501 by claypooj
Geht wieder hoch her heute.
z.b folgendes von ruff
gruss vaka
Re: Shelf Registration
by: ruffneck28 04/28/05 07:03 am
Msg: 55504 of 55609
It depends ....
we already know the goal is 250 million revenues and they can t reach it by organic growth only. So further financing will be needed in the future and theirfore the shelf reg.
If they do additional PP s leaving the money at bank unused for a long time again we have dilution of course.
If the money is invested in an accretive aquisition at the same time again it depends on the price revenues and profit of the aquired company.
Actually the money of the last PP is still not invested completely and they have additional bank facilities they can use for a possible bigger aquisition while I am not aware of such a deal and it seems is getting more and more difficult for them to work out the bigger the to be aquired companies are.
Ruff
Posted as a reply to: Msg 55501 by claypooj
SL vom Feinsten haben die Amis heute abend ausgelöst. Konnte mir welche zu 1,64 $ sichern 

@ honorar
...ob Du das nicht mal bereuen wirst
...ob Du das nicht mal bereuen wirst

fundamental ist trib ne perle. darum mache ich mir diesbezüglich keine sorgen. die kann man getrost über einen längeren zeitraum liegen lassen. ich habe hier schon einmal eine lange reise in den norden mitgemacht und gehe davon aus, dass diese reise alsbald wieder beginnt.
für alle, die in realtime kostenlos in usa verfolgen wollen hier mal ein sehr interessanter link:
http://d-traderz.com/
für alle, die in realtime kostenlos in usa verfolgen wollen hier mal ein sehr interessanter link:
http://d-traderz.com/
Fundamental ist Trinity wirklich ne Perle, aber wo ist der Boden für den Aktienkurs?
Wir hatten das 2002 auch, da habe ich welche für 1,05 bekommen.
Value Per Share (mrq): 2.063
http://finance.yahoo.com/q/ks?s=TRIB Book
Wir hatten das 2002 auch, da habe ich welche für 1,05 bekommen.
Value Per Share (mrq): 2.063
http://finance.yahoo.com/q/ks?s=TRIB Book
Habr mir am Freitag nochmals welche bei 1,60 $ zugelegt. MACD ziemlich weit unten. Ich erwarte baldige Gegenreaktion.
Proposed Change to ADR Ratio and Application to List on NASDAQ National Market
DUBLIN, Ireland, June 1 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it is filing a notification of an ADR ratio change with the Securities & Exchange Commission (SEC) as part of a series of initiatives to improve its institutional investor relations programme and to enhance value for shareholders. These initiatives include an adjustment of the ratio of American Depositary Receipts (ADRs) to Ordinary Shares and an application to list on the NASDAQ National Market. Trinity is currently listed on the NASDAQ Small Cap Market and the Irish Stock Exchange.
The notification being filed with the SEC today describes Trinity`s application to adjust its ADR ratio (the ratio of ADRs to underlying Ordinary Shares) from 1 ADR: 1 Ordinary Share to 1 ADR: 4 Ordinary Shares. The purpose of the adjusted ratio of ADRs is a technical increase in Trinity`s share price (with a corresponding reduction in ADRs in issue) which will allow Trinity achieve a listing on the NASDAQ National Market.
Trinity currently meets all of the stringent requirements for NASDAQ National Market listing status with the exception of the minimum bid price requirement. As part of the application process, Trinity is adjusting the ADR ratio in order to meet the minimum bid price requirement of US$5. Trinity expects the ratio change to take effect tomorrow and a listing on the NASDAQ National Market to commence on Tuesday June 7th, 2005.
Ronan O`Caoimh, Trinity Biotech Chief Executive Officer, commented "We are pleased that Trinity is upgrading its listing to the NASDAQ National Market. We believe that this upgrade, coupled with our institutional investor relations programme will positively impact our market value. In particular we believe that it will contribute to increased visibility among institutional investors and the sell-side community and that it will significantly increase the pool of institutional investors willing to invest in the stock."
About Trinity Biotech plc
Trinity Biotech develops, acquires manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own sales forces and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
SOURCE Trinity Biotech plc
-0- 06/01/2005
/CONTACT: Trinity Biotech plc, Rory Nealon, +353-1-2769800, E-mail:
rory.nealon@trinitybiotech.com/
(TRIB)
CO: Trinity Biotech plc
ST: Republic of Ireland
IN: BIO HEA MTC
SU: RTG
UK
-- UKW003 --
1416 06/01/200506:00 EDThttp://www.prnewswire.com
DUBLIN, Ireland, June 1 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it is filing a notification of an ADR ratio change with the Securities & Exchange Commission (SEC) as part of a series of initiatives to improve its institutional investor relations programme and to enhance value for shareholders. These initiatives include an adjustment of the ratio of American Depositary Receipts (ADRs) to Ordinary Shares and an application to list on the NASDAQ National Market. Trinity is currently listed on the NASDAQ Small Cap Market and the Irish Stock Exchange.
The notification being filed with the SEC today describes Trinity`s application to adjust its ADR ratio (the ratio of ADRs to underlying Ordinary Shares) from 1 ADR: 1 Ordinary Share to 1 ADR: 4 Ordinary Shares. The purpose of the adjusted ratio of ADRs is a technical increase in Trinity`s share price (with a corresponding reduction in ADRs in issue) which will allow Trinity achieve a listing on the NASDAQ National Market.
Trinity currently meets all of the stringent requirements for NASDAQ National Market listing status with the exception of the minimum bid price requirement. As part of the application process, Trinity is adjusting the ADR ratio in order to meet the minimum bid price requirement of US$5. Trinity expects the ratio change to take effect tomorrow and a listing on the NASDAQ National Market to commence on Tuesday June 7th, 2005.
Ronan O`Caoimh, Trinity Biotech Chief Executive Officer, commented "We are pleased that Trinity is upgrading its listing to the NASDAQ National Market. We believe that this upgrade, coupled with our institutional investor relations programme will positively impact our market value. In particular we believe that it will contribute to increased visibility among institutional investors and the sell-side community and that it will significantly increase the pool of institutional investors willing to invest in the stock."
About Trinity Biotech plc
Trinity Biotech develops, acquires manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own sales forces and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
SOURCE Trinity Biotech plc
-0- 06/01/2005
/CONTACT: Trinity Biotech plc, Rory Nealon, +353-1-2769800, E-mail:
rory.nealon@trinitybiotech.com/
(TRIB)
CO: Trinity Biotech plc
ST: Republic of Ireland
IN: BIO HEA MTC
SU: RTG
UK
-- UKW003 --
1416 06/01/200506:00 EDThttp://www.prnewswire.com
Guten Morgen,
im Board ist es aber still geworden, ich habe auch Trinity und wir alle verfolgten diesen beständigen Sinkflug begeistert, doch was ist jetzt los???
Listing in den USA, meine Verkauforder wurde von der Bank sofort gestrichen, was passiert hier und wie lange können wir in Deutschland nicht handeln???
HELP - HELP - HELP
im Board ist es aber still geworden, ich habe auch Trinity und wir alle verfolgten diesen beständigen Sinkflug begeistert, doch was ist jetzt los???
Listing in den USA, meine Verkauforder wurde von der Bank sofort gestrichen, was passiert hier und wie lange können wir in Deutschland nicht handeln???
HELP - HELP - HELP
es wurde eine Veränderung bei den in den USA und Dt. notierten ADR vorgenommen. Das Verhältnis zum Kurs in Great Britain wurde geändert. Dadurch ändert sich nicht viel. Die Anzahl der Aktien verringert sich und der Kurs steigt im gleichen Zuge (ähnlich einem Reverse-Split). Hintergrund ist sicher, daß viele weltweite Aktienfonds nur in Aktien investieren oder Analysten die Aktien beobachten und bewerten ab einem Kurs von 5,00 US-D. Dass wurde damit erreicht. Hier die Meldung:
DUBLIN, Ireland, June 1 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ: TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it is filing a notification of an ADR ratio change with the Securities & Exchange Commission (SEC) as part of a series of initiatives to improve its institutional investor relations programme and to enhance value for shareholders. These initiatives include an adjustment of the ratio of American Depositary Receipts (ADRs) to Ordinary Shares and an application to list on the NASDAQ National Market. Trinity is currently listed on the NASDAQ Small Cap Market and the Irish Stock Exchange.
ADVERTISEMENT
The notification being filed with the SEC today describes Trinity`s application to adjust its ADR ratio (the ratio of ADRs to underlying Ordinary Shares) from 1 ADR: 1 Ordinary Share to 1 ADR: 4 Ordinary Shares. The purpose of the adjusted ratio of ADRs is a technical increase in Trinity`s share price (with a corresponding reduction in ADRs in issue) which will allow Trinity achieve a listing on the NASDAQ National Market.
Trinity currently meets all of the stringent requirements for NASDAQ National Market listing status with the exception of the minimum bid price requirement. As part of the application process, Trinity is adjusting the ADR ratio in order to meet the minimum bid price requirement of US$5. Trinity expects the ratio change to take effect tomorrow and a listing on the NASDAQ National Market to commence on Tuesday June 7th, 2005.
Ronan O`Caoimh, Trinity Biotech Chief Executive Officer, commented "We are pleased that Trinity is upgrading its listing to the NASDAQ National Market. We believe that this upgrade, coupled with our institutional investor relations programme will positively impact our market value. In particular we believe that it will contribute to increased visibility among institutional investors and the sell-side community and that it will significantly increase the pool of institutional investors willing to invest in the stock."
in den USA hat jetzt die Aktie den Stock-Code:
TRIBD
http://finance.yahoo.com/q?s=TRIBD
in Dt. findest du die Aktie neu unter:
A0ET3S
oder ISIN
US8964383066
Der Verkaufsstop wird von deiner Bank gesetzt, da die Aktien jetzt im verhältnis umgetauscht werden. Für 4 bisherige Aktien hast du dann eine neue Aktie. Ziel ist sicher auch vom Nasdaq Small Market wieder in die Nasdaq zu kommen, um mehr Umsätze in die Aktie zu bekommen.
DUBLIN, Ireland, June 1 /PRNewswire-FirstCall/ -- Trinity Biotech plc (NASDAQ: TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it is filing a notification of an ADR ratio change with the Securities & Exchange Commission (SEC) as part of a series of initiatives to improve its institutional investor relations programme and to enhance value for shareholders. These initiatives include an adjustment of the ratio of American Depositary Receipts (ADRs) to Ordinary Shares and an application to list on the NASDAQ National Market. Trinity is currently listed on the NASDAQ Small Cap Market and the Irish Stock Exchange.
ADVERTISEMENT
The notification being filed with the SEC today describes Trinity`s application to adjust its ADR ratio (the ratio of ADRs to underlying Ordinary Shares) from 1 ADR: 1 Ordinary Share to 1 ADR: 4 Ordinary Shares. The purpose of the adjusted ratio of ADRs is a technical increase in Trinity`s share price (with a corresponding reduction in ADRs in issue) which will allow Trinity achieve a listing on the NASDAQ National Market.
Trinity currently meets all of the stringent requirements for NASDAQ National Market listing status with the exception of the minimum bid price requirement. As part of the application process, Trinity is adjusting the ADR ratio in order to meet the minimum bid price requirement of US$5. Trinity expects the ratio change to take effect tomorrow and a listing on the NASDAQ National Market to commence on Tuesday June 7th, 2005.
Ronan O`Caoimh, Trinity Biotech Chief Executive Officer, commented "We are pleased that Trinity is upgrading its listing to the NASDAQ National Market. We believe that this upgrade, coupled with our institutional investor relations programme will positively impact our market value. In particular we believe that it will contribute to increased visibility among institutional investors and the sell-side community and that it will significantly increase the pool of institutional investors willing to invest in the stock."
in den USA hat jetzt die Aktie den Stock-Code:
TRIBD
http://finance.yahoo.com/q?s=TRIBD
in Dt. findest du die Aktie neu unter:
A0ET3S
oder ISIN
US8964383066
Der Verkaufsstop wird von deiner Bank gesetzt, da die Aktien jetzt im verhältnis umgetauscht werden. Für 4 bisherige Aktien hast du dann eine neue Aktie. Ziel ist sicher auch vom Nasdaq Small Market wieder in die Nasdaq zu kommen, um mehr Umsätze in die Aktie zu bekommen.
news
CHESTER, N.J., June 21 /PRNewswire/ -- Today, GraficaGroup officially announced that Trinity Biotech USA has named the Chester-based company as its new Agency of Record. After reviewing several agencies in the first quarter of 2005, Trinity selected Grafica to help develop and communicate a new brand position to support an overall marketing-communications campaign. Grafica will provide advertising, media, trade-show support, and interactive services for Trinity, as the company continues its growth and expansion within multiple clinical diagnostic markets, including hemostasis, immunodiagnostics, specialty chemistry, and rapid HIV testing.
ADVERTISEMENT
"After establishing our U.S. headquarters in New Jersey this year, we sought an agency that could help position the Trinity brand and tell the Trinity story to our key markets," said Philippe Gadal, Ph.D., U.S. CEO, Trinity Biotech. "Grafica proved they possess the requisite creative talent and branding experience that Trinity needs to effectively promote our products, technology, and service, as we continue growing business and market share in the United States."
Grafica began its relationship with Trinity by developing a new brand position, print advertisement, and tagline: Diagnostics Driven by Life.® The campaign starts in June in several publications targeting clinical diagnostic professionals and public health officials. The new Trinity campaign conveys how Trinity understands the pressures of life in the laboratory, while communicating the well-known brands in the company`s portfolio, which includes AMAX Destiny(TM) (coagulation), NEXGEN Four (infectious disease), and the Uni-Gold(TM) Recombigen® rapid HIV test.
"Trinity`s products greatly improve the way diagnostic laboratories function, and its Uni-Gold product is the fastest and simplest test for detecting HIV infection," said Debra Taeschler, President and CEO, GraficaGroup. "We at Grafica look forward to integrating the brand position we developed for Trinity across multiple media, including the Web, which we believe will help Trinity assume market leadership in hemostasis, infectious disease, clinical chemistry, and rapid HIV testing."
About Trinity Biotech USA
With its U.S. Corporate Headquarters based in Berkeley Heights, New Jersey, Trinity Biotech Inc. is a wholly owned subsidiary of Trinity Biotech plc (Nasdaq: TRIB - News). With World Headquarters in Bray, Ireland, Trinity Biotech plc was established in 1992. Through an acquisition-growth strategy, Trinity has acquired companies with expertise in the development and commercialization of both instrumentation and reagents primarily servicing the infectious disease, hemostasis, specialty chemistry, and HIV diagnostics markets. Additional information about Trinity is available on the company Web site at http://www.trinitybiotech.com.
About GraficaGroup
GraficaGroup is a full-service, fully integrated advertising agency led by professionals from Madison Avenue who continue to craft brand -- and relationship-building campaigns from just off of Main Street in Chester, New Jersey. Grafica (http://www.grafica.com) provides both traditional and online marketing campaigns, including broadcast, print, out of home, direct mail, point of purchase, email, and Web-site design and development. Grafica also offers media planning and buying, search-engine optimization, database management, and public relations. Clients include AT&T, Cathedral Healthcare System, First Morris Bank and Trust, Horizon Blue Cross Blue Shield of New Jersey, Jackson Hewitt Tax Service, the New Jersey Board of Public Utilities, the New Jersey Office of the Attorney General, PSE&G, Singer Nelson Charlmers, Synchronoss Technologies, and Wells Fargo Home Mortgage
CHESTER, N.J., June 21 /PRNewswire/ -- Today, GraficaGroup officially announced that Trinity Biotech USA has named the Chester-based company as its new Agency of Record. After reviewing several agencies in the first quarter of 2005, Trinity selected Grafica to help develop and communicate a new brand position to support an overall marketing-communications campaign. Grafica will provide advertising, media, trade-show support, and interactive services for Trinity, as the company continues its growth and expansion within multiple clinical diagnostic markets, including hemostasis, immunodiagnostics, specialty chemistry, and rapid HIV testing.
ADVERTISEMENT
"After establishing our U.S. headquarters in New Jersey this year, we sought an agency that could help position the Trinity brand and tell the Trinity story to our key markets," said Philippe Gadal, Ph.D., U.S. CEO, Trinity Biotech. "Grafica proved they possess the requisite creative talent and branding experience that Trinity needs to effectively promote our products, technology, and service, as we continue growing business and market share in the United States."
Grafica began its relationship with Trinity by developing a new brand position, print advertisement, and tagline: Diagnostics Driven by Life.® The campaign starts in June in several publications targeting clinical diagnostic professionals and public health officials. The new Trinity campaign conveys how Trinity understands the pressures of life in the laboratory, while communicating the well-known brands in the company`s portfolio, which includes AMAX Destiny(TM) (coagulation), NEXGEN Four (infectious disease), and the Uni-Gold(TM) Recombigen® rapid HIV test.
"Trinity`s products greatly improve the way diagnostic laboratories function, and its Uni-Gold product is the fastest and simplest test for detecting HIV infection," said Debra Taeschler, President and CEO, GraficaGroup. "We at Grafica look forward to integrating the brand position we developed for Trinity across multiple media, including the Web, which we believe will help Trinity assume market leadership in hemostasis, infectious disease, clinical chemistry, and rapid HIV testing."
About Trinity Biotech USA
With its U.S. Corporate Headquarters based in Berkeley Heights, New Jersey, Trinity Biotech Inc. is a wholly owned subsidiary of Trinity Biotech plc (Nasdaq: TRIB - News). With World Headquarters in Bray, Ireland, Trinity Biotech plc was established in 1992. Through an acquisition-growth strategy, Trinity has acquired companies with expertise in the development and commercialization of both instrumentation and reagents primarily servicing the infectious disease, hemostasis, specialty chemistry, and HIV diagnostics markets. Additional information about Trinity is available on the company Web site at http://www.trinitybiotech.com.
About GraficaGroup
GraficaGroup is a full-service, fully integrated advertising agency led by professionals from Madison Avenue who continue to craft brand -- and relationship-building campaigns from just off of Main Street in Chester, New Jersey. Grafica (http://www.grafica.com) provides both traditional and online marketing campaigns, including broadcast, print, out of home, direct mail, point of purchase, email, and Web-site design and development. Grafica also offers media planning and buying, search-engine optimization, database management, and public relations. Clients include AT&T, Cathedral Healthcare System, First Morris Bank and Trust, Horizon Blue Cross Blue Shield of New Jersey, Jackson Hewitt Tax Service, the New Jersey Board of Public Utilities, the New Jersey Office of the Attorney General, PSE&G, Singer Nelson Charlmers, Synchronoss Technologies, and Wells Fargo Home Mortgage
DUBLIN, Ireland, June 27 /PRNewswire-FirstCall/ -- In recognition of National HIV Testing Day, Trinity Biotech plc (NASDAQ: TRIBD, ISE: TRIB.I) announced today that it will make a donation to the value of US$1m of their Uni-Gold(TM) Recombigen® HIV Rapid tests to community based organizations (CBO`s) and public health facilities in the US in an effort to expand the availability of rapid HIV testing programmes.
ADVERTISEMENT
The company will distribute the Uni-Gold Rapid HIV tests over the next six months to qualifying CBO`s and public health facilities. Applications should be made through Trinity`s corporate website: (www.trinitybiotech.com).
Trinity`s Uni-Gold Recombigen HIV test is the only rapid test available in the United States which is both CLIA waived for venous and fingerstick whole blood use, and has FDA approval for use with serum, plasma, and whole blood. Similarly, it is the only CLIA waived product which can provide a result in as quickly as 10 minutes. The Uni-Gold HIV product has a demonstrated sensitivity and specificity of 100% and 99.7% respectively.
The CLIA waived status of the product makes it suitable for use in many publicly sponsored outreach settings, such as STD and HIV clinics, where workers and volunteers can perform the test without formal medical training. More information in regard to the Uni-Gold Recombigen HIV test can be found at www.unigoldhiv.com.
"We are delighted to help increase the number of Rapid HIV tests being performed in the United States through this donation of Uni-Gold Recombigen HIV Rapid tests, said Ronan O`Caoimh, Trinity Biotech CEO. "We are particularly pleased to do so on National HIV Testing Day and we hope that this donation will help the ongoing fight against HIV/AIDS. This donation is accessible to community based groups and public health facilities of all sizes and applications should be made though our website. An objective review panel consisting of members from NAPWA (National Association of People with AIDS) and NASTAD (National Alliance of State and Territorial AIDS Directors) will evaluate all completed applications, and a decision will be made on a needs based analysis."
As part of this donation, Trinity has worked with NAPWA to provide several community based organizations a total of 15,000 Uni-Gold Recombigen HIV tests which will be used for testing on National HIV Testing Day. These organizations include the Metropolitan Atlanta Outreach Association of Georgia and the Minority AIDS Project of Los Angeles California.
National HIV Testing Day (NHTD) is an annual campaign organized by NAPWA to encourage at-risk individuals to receive voluntary HIV counseling and testing. NAPWA distributes campaign kits to community groups and health departments of all sizes to help create NHTD campaigns and events targeting their local communities.
Trinity Biotech develops, acquires, manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com.
ADVERTISEMENT
The company will distribute the Uni-Gold Rapid HIV tests over the next six months to qualifying CBO`s and public health facilities. Applications should be made through Trinity`s corporate website: (www.trinitybiotech.com).
Trinity`s Uni-Gold Recombigen HIV test is the only rapid test available in the United States which is both CLIA waived for venous and fingerstick whole blood use, and has FDA approval for use with serum, plasma, and whole blood. Similarly, it is the only CLIA waived product which can provide a result in as quickly as 10 minutes. The Uni-Gold HIV product has a demonstrated sensitivity and specificity of 100% and 99.7% respectively.
The CLIA waived status of the product makes it suitable for use in many publicly sponsored outreach settings, such as STD and HIV clinics, where workers and volunteers can perform the test without formal medical training. More information in regard to the Uni-Gold Recombigen HIV test can be found at www.unigoldhiv.com.
"We are delighted to help increase the number of Rapid HIV tests being performed in the United States through this donation of Uni-Gold Recombigen HIV Rapid tests, said Ronan O`Caoimh, Trinity Biotech CEO. "We are particularly pleased to do so on National HIV Testing Day and we hope that this donation will help the ongoing fight against HIV/AIDS. This donation is accessible to community based groups and public health facilities of all sizes and applications should be made though our website. An objective review panel consisting of members from NAPWA (National Association of People with AIDS) and NASTAD (National Alliance of State and Territorial AIDS Directors) will evaluate all completed applications, and a decision will be made on a needs based analysis."
As part of this donation, Trinity has worked with NAPWA to provide several community based organizations a total of 15,000 Uni-Gold Recombigen HIV tests which will be used for testing on National HIV Testing Day. These organizations include the Metropolitan Atlanta Outreach Association of Georgia and the Minority AIDS Project of Los Angeles California.
National HIV Testing Day (NHTD) is an annual campaign organized by NAPWA to encourage at-risk individuals to receive voluntary HIV counseling and testing. NAPWA distributes campaign kits to community groups and health departments of all sizes to help create NHTD campaigns and events targeting their local communities.
Trinity Biotech develops, acquires, manufactures and markets diagnostic products for the clinical laboratory and point-of-care segments of the diagnostic market. The broad line of over 500 test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity is also a significant provider of raw materials to the life science industry. Trinity Biotech sells worldwide in 78 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com.
2005 Second Quarter Results
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2005 second quarter results on Thursday, 28 July, 2005.
The Trinity Biotech management team will discuss second quarter financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
The call is being webcast and can be accessed at www.vcall.com
Further details are available from K Capital Source on +353 1 631 5500 or TRIB@kcapitalsource.com
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2005 second quarter results on Thursday, 28 July, 2005.
The Trinity Biotech management team will discuss second quarter financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
The call is being webcast and can be accessed at www.vcall.com
Further details are available from K Capital Source on +353 1 631 5500 or TRIB@kcapitalsource.com
!
Dieser Beitrag wurde vom System automatisch gesperrt. Bei Fragen wenden Sie sich bitte an feedback@wallstreet-online.de!
Dieser Beitrag wurde vom System automatisch gesperrt. Bei Fragen wenden Sie sich bitte an feedback@wallstreet-online.de
Press Release Source: Trinity Biotech
Trinity Biotech Completes Acquisition of Primus Corporation
Thursday July 21, 10:12 am ET
DUBLIN, IRELAND--(MARKET WIRE)--Jul 21, 2005 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has completed the acquisition of Primus Corporation, a leader in the field of in vitro diagnostic testing for haemoglobin A1c and haemoglobin variants.
ADVERTISEMENT
Trinity Biotech has acquired Primus for US$12 million, consisting of a cash consideration of US$9 million and a one-year promissory note of US$3 million. The shareholders of Primus will also be entitled to an additional consideration of up to US$4.6 million depending on the growth of the business during the remainder of 2005. The transaction is being funded entirely with bank borrowings, thereby leaving Trinity`s cash balances available for future potential acquisitions.
Primus, a Kansas City, Missouri based corporation advised by Crosstree Capital Partners, was founded in 1989 and employs 36 people in the manufacture of glycohaemoglobin instruments and reagents to serve customers from physicians` offices to the largest reference laboratories worldwide. Trinity is committed to further developing the business from the Kansas City location.
Primus has demonstrated consistent sales and profit growth. Sales increased from $3.6 million for the year 2001 to $7.9 million for the year 2004, a compound annual growth rate of 30%. The acquisition will be earnings positive for Trinity Biotech in 2005. As part of the transaction, Primus CEO and founder, Tom Reidy, will remain with the Company and continue to direct the operations of Primus within the Trinity Group.
Ronan O`Caoimh, Trinity Biotech CEO, commented, "We are delighted to acquire Primus which has a strong track record of growth and a unique patented technology in the field of A1c testing. In particular, Primus has a strong position in the US market for variant A1c testing with an estimated market share of 35%. Trinity`s distribution network through direct sales and worldwide distributors can further expand the Primus sales effort and ultimately result in a significant increase in the number of instruments placed above the current level of approximately three hundred. We are also excited about the new Primus Rapid Gel product which is currently awaiting FDA approval which we expect will be granted in early 2006. The Rapid Gel product will compete in the US$100 million point-of-care A1c market and is based on the Company`s unique boronate affinity methodology. We are confident that Primus will make a significant contribution to Trinity`s growth in future years."
Tom Reidy, CEO and founder of Primus, commented, "The acquisition of Primus by Trinity will provide the company with access to the wider distribution base and technical support infrastructure of Trinity`s direct sales operations in the USA, Germany and the UK and to its worldwide distributor network. We are very excited at the opportunities this will present to continue the growth profile of our business and to better serve our customers."
Trinity will provide additional information regarding the acquisition during its second quarter earnings conference call on Thursday 28th July at 11.00am EST.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Contact:
Contact:
Trinity Biotech plc
Rory Nealon
(353)-1-2769800
E-mail: Email Contact
Trinity Biotech Completes Acquisition of Primus Corporation
Thursday July 21, 10:12 am ET
DUBLIN, IRELAND--(MARKET WIRE)--Jul 21, 2005 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has completed the acquisition of Primus Corporation, a leader in the field of in vitro diagnostic testing for haemoglobin A1c and haemoglobin variants.
ADVERTISEMENT
Trinity Biotech has acquired Primus for US$12 million, consisting of a cash consideration of US$9 million and a one-year promissory note of US$3 million. The shareholders of Primus will also be entitled to an additional consideration of up to US$4.6 million depending on the growth of the business during the remainder of 2005. The transaction is being funded entirely with bank borrowings, thereby leaving Trinity`s cash balances available for future potential acquisitions.
Primus, a Kansas City, Missouri based corporation advised by Crosstree Capital Partners, was founded in 1989 and employs 36 people in the manufacture of glycohaemoglobin instruments and reagents to serve customers from physicians` offices to the largest reference laboratories worldwide. Trinity is committed to further developing the business from the Kansas City location.
Primus has demonstrated consistent sales and profit growth. Sales increased from $3.6 million for the year 2001 to $7.9 million for the year 2004, a compound annual growth rate of 30%. The acquisition will be earnings positive for Trinity Biotech in 2005. As part of the transaction, Primus CEO and founder, Tom Reidy, will remain with the Company and continue to direct the operations of Primus within the Trinity Group.
Ronan O`Caoimh, Trinity Biotech CEO, commented, "We are delighted to acquire Primus which has a strong track record of growth and a unique patented technology in the field of A1c testing. In particular, Primus has a strong position in the US market for variant A1c testing with an estimated market share of 35%. Trinity`s distribution network through direct sales and worldwide distributors can further expand the Primus sales effort and ultimately result in a significant increase in the number of instruments placed above the current level of approximately three hundred. We are also excited about the new Primus Rapid Gel product which is currently awaiting FDA approval which we expect will be granted in early 2006. The Rapid Gel product will compete in the US$100 million point-of-care A1c market and is based on the Company`s unique boronate affinity methodology. We are confident that Primus will make a significant contribution to Trinity`s growth in future years."
Tom Reidy, CEO and founder of Primus, commented, "The acquisition of Primus by Trinity will provide the company with access to the wider distribution base and technical support infrastructure of Trinity`s direct sales operations in the USA, Germany and the UK and to its worldwide distributor network. We are very excited at the opportunities this will present to continue the growth profile of our business and to better serve our customers."
Trinity will provide additional information regarding the acquisition during its second quarter earnings conference call on Thursday 28th July at 11.00am EST.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the company`s website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Contact:
Contact:
Trinity Biotech plc
Rory Nealon
(353)-1-2769800
E-mail: Email Contact
immer das gleiche Umsätze steigen leicht, Gewinne sinken! Werden die Übernahmen aus dem Cash bezahlt oder durch Ausgabe von Aktien?
DUBLIN, IRELAND--(MARKET WIRE)--Jul 28, 2005 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2005.
Revenues in the quarter increased 13.2% to US$22.7 million compared to US$20.0 million in the same period last year. Revenues for the year to date by key product area were as follows :
H1 2004 H1 2005 % Increase
US$000 US$000
Infectious Disease 13,919 14,909 7.1
Coagulation 13,603 14,399 5.9
POC 4,275 6,318 47.8
Other 4,661 8,313 78.3
Total 36,458 43,939 20.5
Revenues by geographical location were as follows :
H1 2004 H1 2005 % Increase
US$000 US$000
USA 18,119 21,808 20.4
Europe 11,302 11,894 5.2
Asia / Africa 7,037 10,237 45.5
Total 36,458 43,939 20.5
ADVERTISEMENT
Gross profit for the quarter amounted to US$11.0 million, representing a gross margin of 48.2%. This compares to a gross profit of US$10.3 million in the same period last year.
R&D expenses have increased from US$1.2 million in Quarter 2 last year to US$1.3 million in the current quarter. The increase in Administrative costs from US$7.2 million in Quarter 2 last year to US$8.0 million in the current year is primarily attributable to the increased salesforce in the USA and the recent RDI acquisition.
The net effect of the above factors was an operating profit of US$1.67 million for the quarter as compared to US$1.44 million in the most recent quarter. Operating profit of US$1.67 million equates to an earnings per ADR for the quarter of 8.4 US cents (earnings per Ordinary Share of 2.1 cents).
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "Quarter 2 has been a quarter of continuing progress for Trinity. Our revenues have grown significantly, we reported increased operating profits over the prior quarter and we have recently completed the acquisition of Primus. Our quarterly revenues have improved by 13% on the same period last year and by 7% on the most recent quarter. We are particularly pleased with the recent increases in our Point of Care business which grew by 9% on Quarter 1 and by the $1.5 million increase in our Fitzgerald business during the quarter, half of which is attributable to the recent RDI acquisition."
Ronan O`Caoimh, CEO, commented, "We are particularly pleased with the ongoing success of our HIV point-of-care business in both Africa and the US markets which has grown by 48% in the first half of the year as compared to the same period last year. During the quarter, following the consolidation of our ADR unit, we successfully completed our transition from the Nasdaq Small Cap to the Nasdaq National Market.
"We recently completed the acquisition of Primus Corporation, a leader in the field of in vitro diagnostic testing for haemoglobin A1c and haemoglobin variants. Primus has demonstrated average annual growth rates of 30% in recent years primarily due to its patented HPLC boronate affinity testing which is the only truly interference-free A1c test on the market today. We are also excited about the new Primus Rapid Gel product which is currently awaiting FDA approval which we expect will be granted in early 2006. The acquisition has been funded entirely by bank debt which will enhance our financial flexibility and our ability to deliver further shareholder value."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except
share data) Three Months Three Months Six Months Six Months
Ended Ended Ended Ended
June 30, June 30, June 30, June 30,
2005 2004 2005 2004
(Unaudited) (Unaudited) (Unaudited) (Unaudited)
Revenues 22,712 20,049 43,939 36,458
Cost of goods sold (11,754) (9,761) (22,577) (18,133)
-------- ------- -------- --------
Gross profit 10,958 10,288 21,362 18,325
Research &
development expenses (1,302) (1,175) (2,622) (2,353)
Administrative
expenses (7,986) (7,212) (15,632) (12,963)
Operating profit 1,670 1,901 3,108 3,009
Share option expense (201) (54) (413) (97)
Interest receivable 117 64 230 180
Interest payable (222) (225) (460) (432)
----- ----- ----- -----
Profit on
ordinary activities
before tax 1,364 1,686 2,465 2,660
Tax on profit on ordinary
activities (146) 124 (244) 156
Profit after tax 1,218 1,810 2,221 2,816
Earnings per ADR 8.4 13.2 15.5 20.8
Earnings per Ordinary
Share (US$ cents) 2.1 3.3 3.9 5.2
Diluted earnings
per ADR 8.4 12.0 15.5 19.6
Diluted earnings per
Ordinary Share
(US$ cents) 2.1 3.0 3.9 4.9
DUBLIN, IRELAND--(MARKET WIRE)--Jul 28, 2005 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2005.
Revenues in the quarter increased 13.2% to US$22.7 million compared to US$20.0 million in the same period last year. Revenues for the year to date by key product area were as follows :
H1 2004 H1 2005 % Increase
US$000 US$000
Infectious Disease 13,919 14,909 7.1
Coagulation 13,603 14,399 5.9
POC 4,275 6,318 47.8
Other 4,661 8,313 78.3
Total 36,458 43,939 20.5
Revenues by geographical location were as follows :
H1 2004 H1 2005 % Increase
US$000 US$000
USA 18,119 21,808 20.4
Europe 11,302 11,894 5.2
Asia / Africa 7,037 10,237 45.5
Total 36,458 43,939 20.5
ADVERTISEMENT
Gross profit for the quarter amounted to US$11.0 million, representing a gross margin of 48.2%. This compares to a gross profit of US$10.3 million in the same period last year.
R&D expenses have increased from US$1.2 million in Quarter 2 last year to US$1.3 million in the current quarter. The increase in Administrative costs from US$7.2 million in Quarter 2 last year to US$8.0 million in the current year is primarily attributable to the increased salesforce in the USA and the recent RDI acquisition.
The net effect of the above factors was an operating profit of US$1.67 million for the quarter as compared to US$1.44 million in the most recent quarter. Operating profit of US$1.67 million equates to an earnings per ADR for the quarter of 8.4 US cents (earnings per Ordinary Share of 2.1 cents).
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "Quarter 2 has been a quarter of continuing progress for Trinity. Our revenues have grown significantly, we reported increased operating profits over the prior quarter and we have recently completed the acquisition of Primus. Our quarterly revenues have improved by 13% on the same period last year and by 7% on the most recent quarter. We are particularly pleased with the recent increases in our Point of Care business which grew by 9% on Quarter 1 and by the $1.5 million increase in our Fitzgerald business during the quarter, half of which is attributable to the recent RDI acquisition."
Ronan O`Caoimh, CEO, commented, "We are particularly pleased with the ongoing success of our HIV point-of-care business in both Africa and the US markets which has grown by 48% in the first half of the year as compared to the same period last year. During the quarter, following the consolidation of our ADR unit, we successfully completed our transition from the Nasdaq Small Cap to the Nasdaq National Market.
"We recently completed the acquisition of Primus Corporation, a leader in the field of in vitro diagnostic testing for haemoglobin A1c and haemoglobin variants. Primus has demonstrated average annual growth rates of 30% in recent years primarily due to its patented HPLC boronate affinity testing which is the only truly interference-free A1c test on the market today. We are also excited about the new Primus Rapid Gel product which is currently awaiting FDA approval which we expect will be granted in early 2006. The acquisition has been funded entirely by bank debt which will enhance our financial flexibility and our ability to deliver further shareholder value."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except
share data) Three Months Three Months Six Months Six Months
Ended Ended Ended Ended
June 30, June 30, June 30, June 30,
2005 2004 2005 2004
(Unaudited) (Unaudited) (Unaudited) (Unaudited)
Revenues 22,712 20,049 43,939 36,458
Cost of goods sold (11,754) (9,761) (22,577) (18,133)
-------- ------- -------- --------
Gross profit 10,958 10,288 21,362 18,325
Research &
development expenses (1,302) (1,175) (2,622) (2,353)
Administrative
expenses (7,986) (7,212) (15,632) (12,963)
Operating profit 1,670 1,901 3,108 3,009
Share option expense (201) (54) (413) (97)
Interest receivable 117 64 230 180
Interest payable (222) (225) (460) (432)
----- ----- ----- -----
Profit on
ordinary activities
before tax 1,364 1,686 2,465 2,660
Tax on profit on ordinary
activities (146) 124 (244) 156
Profit after tax 1,218 1,810 2,221 2,816
Earnings per ADR 8.4 13.2 15.5 20.8
Earnings per Ordinary
Share (US$ cents) 2.1 3.3 3.9 5.2
Diluted earnings
per ADR 8.4 12.0 15.5 19.6
Diluted earnings per
Ordinary Share
(US$ cents) 2.1 3.0 3.9 4.9
Die Firma hat schon immer eine schlechte Aktionärspolitik gemacht. Keine Dvidende gezahlt und neue Aktien ohne die Ausgabe von Bezugsrechten auf den MArkt geworfen. Ich rechne nicht damit, dass sich das ändert. Das Verhalten ist im übrigen typsich für kleine Firmen, die die Börse lediglich dazu benutzen Geld zu holen und ansonsten undurchsichtig bleiben und durch die Interessen der Eigner dominiert werden. Mit Standardwerte oder Midcaps läßt sich besser Geld verdienen. Rühmliche Ausnahme zur Zeit "Cybio".
[posting]17.542.764 von kaiholla am 13.08.05 13:18:17[/posting]Richtig!
Auch Geld verdienen ist woanders zur Zeit leichter.
Aber trib wird in eine Umsatzgröße wachsen wo das nicht mehr funktioniert.
Die haben in den letzten Monaten Druck bekommen das Geschäft transparenter darzustellen und die Gewinne zu steigern.
Das Thema ist nicht neu.
Aber geht mal auf die HP von trinity und schaut Euch an wieviele Firmen seit der Gründung gekauft wurden.
Das die Integration einer Firma nicht einfach ist sollte klar sein.
Jedes Jahr ein oder zwei Firmen noch schwieriger.
Das Management denkt hier ganz klar langfristig.
Ich sehe auch einige Entscheidungen der guys aus Bray sehr kritisch, denke aber das die das Wohl der Firma als 1. im Auge haben und nicht nur an Ihre Optionen denken.
PS: Buchwert ist aktuell bei 8,8$
Total Cash (mrq): 17.95M
Total Cash Per Share (mrq): 1.291
Total Debt (mrq): 19.34M
Total Debt/Equity (mrq): 0.152
Current Ratio (mrq): 3.401
Book Value Per Share (mrq): 8.801
Gruss
vaka
Auch Geld verdienen ist woanders zur Zeit leichter.
Aber trib wird in eine Umsatzgröße wachsen wo das nicht mehr funktioniert.
Die haben in den letzten Monaten Druck bekommen das Geschäft transparenter darzustellen und die Gewinne zu steigern.
Das Thema ist nicht neu.
Aber geht mal auf die HP von trinity und schaut Euch an wieviele Firmen seit der Gründung gekauft wurden.
Das die Integration einer Firma nicht einfach ist sollte klar sein.
Jedes Jahr ein oder zwei Firmen noch schwieriger.
Das Management denkt hier ganz klar langfristig.
Ich sehe auch einige Entscheidungen der guys aus Bray sehr kritisch, denke aber das die das Wohl der Firma als 1. im Auge haben und nicht nur an Ihre Optionen denken.
PS: Buchwert ist aktuell bei 8,8$

Total Cash (mrq): 17.95M
Total Cash Per Share (mrq): 1.291
Total Debt (mrq): 19.34M
Total Debt/Equity (mrq): 0.152
Current Ratio (mrq): 3.401
Book Value Per Share (mrq): 8.801
Gruss
vaka
EL DORADO HILLS, Calif.--(BUSINESS WIRE)--Aug. 19, 2005--Dutton Associates updates coverage of Trinity Biotech (NASDAQ: TRIB - News) maintaining a Strong Speculative Buy rating and a 12- 18 month $12 price target. The 20-page report by Dutton senior analyst Steve Handley is available at www.jmdutton.com as well as from First Call, Bloomberg, Zacks, Reuters, and other leading financial portals.
ADVERTISEMENT
Trinity Biotech plc, develops, acquires, manufactures, and markets diagnostic products for the point-of-care (POC) and clinical laboratory segments of the diagnostic market. The broad line of over 500 different tests is used to detect infectious diseases, sexually transmitted diseases (STD), blood coagulation disorders and autoimmune diseases. Through its own salesforce as well as a network of international distributors and strategic partners, it sells products in 80 countries. During the first six months of 2005, The Americas contributed 50% of total sales, 27% were obtained from Europe, and the remaining 23% were generated from other areas. While Trinity accounts for a very small share of the rather fragmented $25 billion worldwide market for diagnostic reagents, consumables and instrumentation, we believe that this small share should be viewed as an opportunity to grow rapidly from a small base, aided by an aggressive acquisition program. Trinity`s earnings have been disappointing for several quarters, and this is the main reason that the stock has declined significantly from a 2003 high. In addition to being penalized by the decline of the U.S. dollar vs. the Euro, reported earnings have been hurt by several internal factors. In our view, one reason investors have become increasingly intolerant of recent poor earnings is that management`s actions and statements appear to place the highest priority on expanding sales, and seemingly there is a lack of urgency about achieving satisfactory profitability. This concern, which we do not believe is well founded, has probably been heightened by the following reasons we discuss. It remains our belief that often the most rewarding time to purchase a company`s stock is during a temporary period of depressed earnings, while many investors are focusing on current results rather than on longer term prospects. Our judgment about the current timeliness of Trinity`s stock is based on two considerations: the Company`s depressed valuation (with a market capitalization of slightly less than one times 2005E sales) and our judgment that earnings pressures are subsiding.
ADVERTISEMENT
Trinity Biotech plc, develops, acquires, manufactures, and markets diagnostic products for the point-of-care (POC) and clinical laboratory segments of the diagnostic market. The broad line of over 500 different tests is used to detect infectious diseases, sexually transmitted diseases (STD), blood coagulation disorders and autoimmune diseases. Through its own salesforce as well as a network of international distributors and strategic partners, it sells products in 80 countries. During the first six months of 2005, The Americas contributed 50% of total sales, 27% were obtained from Europe, and the remaining 23% were generated from other areas. While Trinity accounts for a very small share of the rather fragmented $25 billion worldwide market for diagnostic reagents, consumables and instrumentation, we believe that this small share should be viewed as an opportunity to grow rapidly from a small base, aided by an aggressive acquisition program. Trinity`s earnings have been disappointing for several quarters, and this is the main reason that the stock has declined significantly from a 2003 high. In addition to being penalized by the decline of the U.S. dollar vs. the Euro, reported earnings have been hurt by several internal factors. In our view, one reason investors have become increasingly intolerant of recent poor earnings is that management`s actions and statements appear to place the highest priority on expanding sales, and seemingly there is a lack of urgency about achieving satisfactory profitability. This concern, which we do not believe is well founded, has probably been heightened by the following reasons we discuss. It remains our belief that often the most rewarding time to purchase a company`s stock is during a temporary period of depressed earnings, while many investors are focusing on current results rather than on longer term prospects. Our judgment about the current timeliness of Trinity`s stock is based on two considerations: the Company`s depressed valuation (with a market capitalization of slightly less than one times 2005E sales) and our judgment that earnings pressures are subsiding.
der Cahrt ist falsch, da der Reverse-Split nicht berücksichtigt wurde. Hier mal den richtige Chart ansehen
http://finance.yahoo.com/q/bc?s=TRIB&t=2y&l=on&z=m&q=l&c=
http://finance.yahoo.com/q/bc?s=TRIB&t=2y&l=on&z=m&q=l&c=
Auch nich schlecht... 
DUBLIN, IRELAND, Sep 07, 2005 (MARKET WIRE via COMTEX) -- Trinity Biotech plc (TRIB) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that members of its board of directors acquired a total of 4,501,675 Ordinary Shares in the company by purchasing TRIB stock on the open market and exercising share options.
The Company reported that Chairman and CEO Ronan O` Caoimh purchased 1,750,800 shares in the open market and exercised 1,070,000 options, President Brendan Farrell purchased 10,000 shares and exercised 526,875 options, COO Jim Walsh purchased 184,000 shares and exercised 750,000 options, CFO Rory Nealon exercised 200,000 options and non-executive director Denis Burger purchased 10,000 shares.
Ronan O`Caoimh, Trinity Biotech CEO, commented, "These transactions reflect the confidence and belief that the senior executives and Board of the Company have in the future of Trinity Biotech."
Trinity Biotech also announces that Brendan Farrell, President, will be speaking at the Roth Capital Partners New York conference today at 8am E.S.T. The event will be webcast and can be accessed on www.wsw.com/webcast/roth6/trib/
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners.
Trinity Biotech trades its Ordinary Shares on the Irish Stock Exchange and ADR`s on Nasdaq. Each ADR represents four Ordinary Shares. For further information please see the company`s website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Contact :
Trinity Biotech plc
Rory Nealon
(353)-1-2769800
E-mail: Contact via http://www.marketwire.com/mw/emailprcntct?id=C5B43A50AEB8AD5…
SOURCE: Trinity Biotech
Copyright 2005 Market Wire, All rights reserved. ********************************************************************** As of Saturday, 09-03-2005 23:59, the latest Comtex SmarTrend(SM) Alert, an automated pattern recognition system, indicated an UPTREND on 05-26-2005 for TRIB @ $1.88. (C) 2005 Comtex News Network, Inc. All rights reserved.

DUBLIN, IRELAND, Sep 07, 2005 (MARKET WIRE via COMTEX) -- Trinity Biotech plc (TRIB) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that members of its board of directors acquired a total of 4,501,675 Ordinary Shares in the company by purchasing TRIB stock on the open market and exercising share options.
The Company reported that Chairman and CEO Ronan O` Caoimh purchased 1,750,800 shares in the open market and exercised 1,070,000 options, President Brendan Farrell purchased 10,000 shares and exercised 526,875 options, COO Jim Walsh purchased 184,000 shares and exercised 750,000 options, CFO Rory Nealon exercised 200,000 options and non-executive director Denis Burger purchased 10,000 shares.
Ronan O`Caoimh, Trinity Biotech CEO, commented, "These transactions reflect the confidence and belief that the senior executives and Board of the Company have in the future of Trinity Biotech."
Trinity Biotech also announces that Brendan Farrell, President, will be speaking at the Roth Capital Partners New York conference today at 8am E.S.T. The event will be webcast and can be accessed on www.wsw.com/webcast/roth6/trib/
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners.
Trinity Biotech trades its Ordinary Shares on the Irish Stock Exchange and ADR`s on Nasdaq. Each ADR represents four Ordinary Shares. For further information please see the company`s website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Contact :
Trinity Biotech plc
Rory Nealon
(353)-1-2769800
E-mail: Contact via http://www.marketwire.com/mw/emailprcntct?id=C5B43A50AEB8AD5…
SOURCE: Trinity Biotech
Copyright 2005 Market Wire, All rights reserved. ********************************************************************** As of Saturday, 09-03-2005 23:59, the latest Comtex SmarTrend(SM) Alert, an automated pattern recognition system, indicated an UPTREND on 05-26-2005 for TRIB @ $1.88. (C) 2005 Comtex News Network, Inc. All rights reserved.
Jeden Tag hohes Volumen und über 5%
aktuell über 7%.
Liegt das an der PR vom 7.9 ?
Geht mir jetzt etwas zu schnell nach oben.
aktuell über 7%.
Liegt das an der PR vom 7.9 ?
Geht mir jetzt etwas zu schnell nach oben.
IR Calender
Roth Capital Conference, New York 7 Sept 2005
Davy Conference, Dublin 18 Oct 2005
Q3 2005 Trinity Biotech Earnings Release
27 Oct 2005*
Rodman Techvest Conference, New York 7 Nov 2005
Nasdaq Opening, World Aids Day, New York 1 Dec 2005
Q4 2005 Trinity Biotech Earnings Release
21 Feb 2006*
Roth Capital Conference, New York 7 Sept 2005
Davy Conference, Dublin 18 Oct 2005
Q3 2005 Trinity Biotech Earnings Release
27 Oct 2005*
Rodman Techvest Conference, New York 7 Nov 2005
Nasdaq Opening, World Aids Day, New York 1 Dec 2005
Q4 2005 Trinity Biotech Earnings Release
21 Feb 2006*
na hoffentlich mal steigende Umsätze und steigende Gewinne! Damit diese tote Aktie wieder zum Leben erweckt wird.
October 27, 2005 - 9:20 AM EDT Print Font Down Font Up Charts
TRIB 7.05 -0.219
Today 5d 1m 3m 1y 5y 10y
Trinity Biotech Announces Quarter 3 Results
Revenues Increase 25%
Trinity Biotech plc (NASDAQ: TRIB) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended September 30, 2005.
Revenues in the quarter increased 25.1% to US$26.9 million compared to US$21.5 million in the same period last year. Revenues for the year to date by key product area were as follows :
2004 2005
Q1 - Q3 Q1 - Q3
US$000 US$000 % Increase
Infectious Disease 25,442 33,130 30.2
Coagulation 19,911 21,490 7.9
POC 7,221 9,467 31.1
Clinical Chemistry 5,361 6,731 25.6
Total 57,935 70,818 22.2
(Note that the category of "Other" has been split with "Fitzgerald"
included in Infectious Disease and Clinical Chemistry combined with the
recent Primus acquisition)
Revenues by geographical location were as follows :
2004 2005
Q1 - Q3 Q1 - Q3
US$000 US$000 % Increase
USA 29,521 36,200 22.6
Europe 16,847 18,339 8.9
Asia / Africa 11,567 16,279 40.7
Total 57,935 70,818 22.2
Gross profit for the quarter amounted to US$13.2 million, representing a gross margin of 48.9%. This compares to a gross profit of US$11.0 million in the same period last year and represents an increase on the Quarter 2 margin of 48.2%.
R&D expenses have increased from US$1.3 million in Quarter 3 last year to US$1.6 million in the current quarter. The increase in Administrative costs from US$7.6 million in Quarter 2 last year to US$9.4 million in the current year is primarily attributable to the recent Primus and RDI acquisitions.
The net effect of the above factors was an operating profit of US$2.15 million and a profit after tax of US$1.6 million for the quarter as compared to US$1.2 million in quarter 2. This equates to an earnings per ADR for the quarter of 10.8 US cents (earnings per Ordinary Share of 2.7 US cents).
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "Quarter 3 has been a quarter of continuing progress for Trinity. Our revenues have grown by 18% on Quarter 2 and our operating margins have increased to 8.0% as compared to 6.8% at the start of the year. We have also funded our recent Primus acquisition entirely by bank debt which will enhance our financial flexibility and our ability to deliver further shareholder value.
"We are especially pleased with the growth across all of our key product areas, each of which is outperforming the market growth rates. In particular, our infectious disease business has benefited strongly from large sales of flu antibodies in the current quarter."
Ronan O`Caoimh, CEO, commented, "Primus, which we acquired on July 21 has experienced very strong instrument placements during the quarter. We are particularly excited about the new Primus Rapid Gel product which is currently awaiting FDA approval. We expect that this approval will be granted in Quarter 2, 2006 and will enable the Company to compete in the US$200 million point of care HbA1c market.
"Our revenues have increased US$4.16 million from US$22.71 million last quarter to US$26.88 million this quarter. Approximately 50% of this increase is due to our acquisition of Primus with the other 50% reflecting an excellent 8% organic growth over the three month period. With operating profits of US$2.15 million in the quarter we have returned to the levels of profit achieved by the Company prior to the huge investment in our sales and marketing effort in the US during 2004. Given the growth being experienced in all product areas we are confident of continuing this trend in profitability in coming quarters.
"In September the directors acquired a total of 4,501,675 Ordinary Shares (1,125,419 ADR`s) in the Company through a combination of 1,954,800 Ordinary Shares purchased on the open market and 2,546,875 share options exercised. The Directors believe that these transactions reflect the confidence and belief that the senior executives and Board of the Company have in the future of Trinity Biotech."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except
share data) Three Months Three Months Nine Months Nine Months
Ended Ended Ended Ended
September 30, September 30, September 30, September 30,
2005 2004 2005 2004
(Unaudited) (Unaudited) (Unaudited) (Unaudited)
Revenues 26,878 21,477 70,818 57,935
Cost of goods sold (13,726) (10,435) (36,304) (28,568)
---------- ---------- ---------- ----------
Gross profit 13,152 11,042 34,514 29,367
Research & development
expenses (1,610) (1,342) (4,232) (3,695)
Administrative
expenses (9,394) (7,574) (25,026) (20,537)
---------- ---------- ---------- ----------
Operating profit 2,148 2,126 5,256 5,135
Share option expense (195) (109) (607) (206)
Interest receivable 70 55 300 235
Interest payable (270) (184) (731) (616)
---------- ---------- ---------- ----------
Profit on ordinary
activities before tax 1,753 1,888 4,218 4,548
Tax on profit on
ordinary activities (148) (180) (392) (24)
---------- ---------- ---------- ----------
Profit after tax
1,605 1,708 3,826 4,524
Earnings per ADR 10.8 12 26.4 33.2
Earnings per Ordinary
Share (US$ cents) 2.7 3 6.6 8.3
Diluted earnings per
ADR 10.4 10.8 25.6 30
Diluted earnings per 2.6 2.7 6.4 7.5
Ordinary Share
(US$ cents)
Weighted average no.
of Ordinary Shares
used in computing
earnings per
Ordinary Share 59,402,312 56,090,236 58,032,535 54,748,831
Prepared in accordance with International Financial Reporting Standards
Trinity Biotech plc
Consolidated Balance Sheet
September 30, December 31,
2005 2004
US$000`s US$000`s
Unaudited Unaudited
CURRENT ASSETS
Cash and cash equivalents 19,286 22,287
Accounts receivable and prepayments 19,608 13,705
Inventories 38,629 37,519
Current tax 138 815
------- -------
77,661 74,326
NON CURRENT ASSETS
Tangible assets 19,339 15,942
Intangible assets 77,681 62,884
Financial assets 382 -
Deferred tax asset 2,421 2,338
Other assets 943 1,330
------- -------
100,766 82,494
TOTAL ASSETS 178,427 156,820
CURRENT LIABILITIES
Short-term financial liabilities 11,854 11,087
Accounts payable & accrued expenses 15,805 8,631
Current tax 596 792
------- -------
28,255 20,510
NON CURRENT LIABILITIES
Long-term financial liabilities 14,084 12,923
Deferred tax liability and other taxes 4,074 3,552
Other liabilities 115 35
------- -------
18,273 16,510
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 806 764
Class "B" ordinary shares 12 12
Share premium account 127,063 120,444
Revenue and other reserves 4,018 (1,420)
------- -------
131,899 119,800
TOTAL LIABILITIES & SHAREHOLDERS`EQUITY 178,427 156,820
Prepared in accordance with International Financial Reporting Standards
Source: Market Wire (October 27, 2005 - 9:20 AM EDT)
News by QuoteMedia
www.quotemedia.com
TRIB 7.05 -0.219
Today 5d 1m 3m 1y 5y 10y
Trinity Biotech Announces Quarter 3 Results
Revenues Increase 25%
Trinity Biotech plc (NASDAQ: TRIB) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended September 30, 2005.
Revenues in the quarter increased 25.1% to US$26.9 million compared to US$21.5 million in the same period last year. Revenues for the year to date by key product area were as follows :
2004 2005
Q1 - Q3 Q1 - Q3
US$000 US$000 % Increase
Infectious Disease 25,442 33,130 30.2
Coagulation 19,911 21,490 7.9
POC 7,221 9,467 31.1
Clinical Chemistry 5,361 6,731 25.6
Total 57,935 70,818 22.2
(Note that the category of "Other" has been split with "Fitzgerald"
included in Infectious Disease and Clinical Chemistry combined with the
recent Primus acquisition)
Revenues by geographical location were as follows :
2004 2005
Q1 - Q3 Q1 - Q3
US$000 US$000 % Increase
USA 29,521 36,200 22.6
Europe 16,847 18,339 8.9
Asia / Africa 11,567 16,279 40.7
Total 57,935 70,818 22.2
Gross profit for the quarter amounted to US$13.2 million, representing a gross margin of 48.9%. This compares to a gross profit of US$11.0 million in the same period last year and represents an increase on the Quarter 2 margin of 48.2%.
R&D expenses have increased from US$1.3 million in Quarter 3 last year to US$1.6 million in the current quarter. The increase in Administrative costs from US$7.6 million in Quarter 2 last year to US$9.4 million in the current year is primarily attributable to the recent Primus and RDI acquisitions.
The net effect of the above factors was an operating profit of US$2.15 million and a profit after tax of US$1.6 million for the quarter as compared to US$1.2 million in quarter 2. This equates to an earnings per ADR for the quarter of 10.8 US cents (earnings per Ordinary Share of 2.7 US cents).
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "Quarter 3 has been a quarter of continuing progress for Trinity. Our revenues have grown by 18% on Quarter 2 and our operating margins have increased to 8.0% as compared to 6.8% at the start of the year. We have also funded our recent Primus acquisition entirely by bank debt which will enhance our financial flexibility and our ability to deliver further shareholder value.
"We are especially pleased with the growth across all of our key product areas, each of which is outperforming the market growth rates. In particular, our infectious disease business has benefited strongly from large sales of flu antibodies in the current quarter."
Ronan O`Caoimh, CEO, commented, "Primus, which we acquired on July 21 has experienced very strong instrument placements during the quarter. We are particularly excited about the new Primus Rapid Gel product which is currently awaiting FDA approval. We expect that this approval will be granted in Quarter 2, 2006 and will enable the Company to compete in the US$200 million point of care HbA1c market.
"Our revenues have increased US$4.16 million from US$22.71 million last quarter to US$26.88 million this quarter. Approximately 50% of this increase is due to our acquisition of Primus with the other 50% reflecting an excellent 8% organic growth over the three month period. With operating profits of US$2.15 million in the quarter we have returned to the levels of profit achieved by the Company prior to the huge investment in our sales and marketing effort in the US during 2004. Given the growth being experienced in all product areas we are confident of continuing this trend in profitability in coming quarters.
"In September the directors acquired a total of 4,501,675 Ordinary Shares (1,125,419 ADR`s) in the Company through a combination of 1,954,800 Ordinary Shares purchased on the open market and 2,546,875 share options exercised. The Directors believe that these transactions reflect the confidence and belief that the senior executives and Board of the Company have in the future of Trinity Biotech."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company`s periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company`s website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Statements of Income
(US$000`s except
share data) Three Months Three Months Nine Months Nine Months
Ended Ended Ended Ended
September 30, September 30, September 30, September 30,
2005 2004 2005 2004
(Unaudited) (Unaudited) (Unaudited) (Unaudited)
Revenues 26,878 21,477 70,818 57,935
Cost of goods sold (13,726) (10,435) (36,304) (28,568)
---------- ---------- ---------- ----------
Gross profit 13,152 11,042 34,514 29,367
Research & development
expenses (1,610) (1,342) (4,232) (3,695)
Administrative
expenses (9,394) (7,574) (25,026) (20,537)
---------- ---------- ---------- ----------
Operating profit 2,148 2,126 5,256 5,135
Share option expense (195) (109) (607) (206)
Interest receivable 70 55 300 235
Interest payable (270) (184) (731) (616)
---------- ---------- ---------- ----------
Profit on ordinary
activities before tax 1,753 1,888 4,218 4,548
Tax on profit on
ordinary activities (148) (180) (392) (24)
---------- ---------- ---------- ----------
Profit after tax
1,605 1,708 3,826 4,524
Earnings per ADR 10.8 12 26.4 33.2
Earnings per Ordinary
Share (US$ cents) 2.7 3 6.6 8.3
Diluted earnings per
ADR 10.4 10.8 25.6 30
Diluted earnings per 2.6 2.7 6.4 7.5
Ordinary Share
(US$ cents)
Weighted average no.
of Ordinary Shares
used in computing
earnings per
Ordinary Share 59,402,312 56,090,236 58,032,535 54,748,831
Prepared in accordance with International Financial Reporting Standards
Trinity Biotech plc
Consolidated Balance Sheet
September 30, December 31,
2005 2004
US$000`s US$000`s
Unaudited Unaudited
CURRENT ASSETS
Cash and cash equivalents 19,286 22,287
Accounts receivable and prepayments 19,608 13,705
Inventories 38,629 37,519
Current tax 138 815
------- -------
77,661 74,326
NON CURRENT ASSETS
Tangible assets 19,339 15,942
Intangible assets 77,681 62,884
Financial assets 382 -
Deferred tax asset 2,421 2,338
Other assets 943 1,330
------- -------
100,766 82,494
TOTAL ASSETS 178,427 156,820
CURRENT LIABILITIES
Short-term financial liabilities 11,854 11,087
Accounts payable & accrued expenses 15,805 8,631
Current tax 596 792
------- -------
28,255 20,510
NON CURRENT LIABILITIES
Long-term financial liabilities 14,084 12,923
Deferred tax liability and other taxes 4,074 3,552
Other liabilities 115 35
------- -------
18,273 16,510
SHAREHOLDERS` EQUITY
Called up share capital
Class "A" ordinary shares 806 764
Class "B" ordinary shares 12 12
Share premium account 127,063 120,444
Revenue and other reserves 4,018 (1,420)
------- -------
131,899 119,800
TOTAL LIABILITIES & SHAREHOLDERS`EQUITY 178,427 156,820
Prepared in accordance with International Financial Reporting Standards
Source: Market Wire (October 27, 2005 - 9:20 AM EDT)
News by QuoteMedia
www.quotemedia.com
Trinity Biotech Presenting at Rodman & Renshaw Techvest Healthcare Conference
Trinity Biotech plc (NASDAQ: TRIB), (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced Brendan Farrell, President, will be speaking at the Rodman & Renshaw Techvest 7th Annual Healthcare Conference in New York City at 8am on November 8, 2005. The event will be webcast and can be accessed on http://wsw.com/webcast/rrshq7/trib/
Trinity Biotech plc (NASDAQ: TRIB), (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced Brendan Farrell, President, will be speaking at the Rodman & Renshaw Techvest 7th Annual Healthcare Conference in New York City at 8am on November 8, 2005. The event will be webcast and can be accessed on http://wsw.com/webcast/rrshq7/trib/
Wer Infos will liest im yahoo board
Medica summary
by: ruffneck28 11/16/05 09:08 am
Msg: 59445 of 59507
Medica Report 16-11-2005
Hi there, today I talked to Mr. Farrell for about an hour. As always here my summary for ya.
First of all Trinity s story is an Earnigs-Story ! The goal is to reach a level of 12 percent operating margin again. Actually we are at 8 percent. 11 % is above average of the top 10 players and again the clear goal is 12 percent by Q4 2006.
Personal take : If revenues inrease and S,G & A keep stable this should bring us to an EPS of 4 cent as the low end
U.S. sales force produce about 15 million $ costs per year. Actually they are working break even.
Mr. Farrell spends 1 week a month travelling to the U.S. speaking to investors and telling the story.
Trinity will not announce contracts unless they are very very big. They are winning and loosing contracts all over the world each day.
By the way Orasure stopped being very open with releases about contracts and numbers. It seems they go the Trib way 
Trinity has 3 distributors in China. There is NO HIV market over there because competitors have very cheap LOW QUALITY tests. There is a huge market for coag and infectious desease products.
They are very pleased with the Primus aquisition and the Gel hopefully gets approval Q2 2006. The market for testing A1C we can target is about 200 million $.
There is a guy on the board complaining about his „visit“ of RDI. When you arrived at the building, the familiy-company was already transfered and the guy who opened and was rude was no Trinity employee.
Actually 3 ongoing discussions for aquisitions. Two smaller and one bigger one. Two of them in an advanced stadium. If possible payment with cash and/or bank credit.
Institutional ownership difficult to say because of the ADR s , but IR company K Capital worked it from about 12 % to about 20 %. Add the 14% owned by management.
Management also shareholders and interested in inreasing shareprice.
Considering a 2nd facility in Bray just across the street. 1st facility running on full steam.
Valuation of a company in the diagnostic business depends on growth rates and products.
Based on that Trinity should be worth between 1.5 and 2 times revenues. ==> UNDERVALUED
Hope this helps
Ruffneck
Medica summary
by: ruffneck28 11/16/05 09:08 am
Msg: 59445 of 59507
Medica Report 16-11-2005
Hi there, today I talked to Mr. Farrell for about an hour. As always here my summary for ya.
First of all Trinity s story is an Earnigs-Story ! The goal is to reach a level of 12 percent operating margin again. Actually we are at 8 percent. 11 % is above average of the top 10 players and again the clear goal is 12 percent by Q4 2006.
Personal take : If revenues inrease and S,G & A keep stable this should bring us to an EPS of 4 cent as the low end
U.S. sales force produce about 15 million $ costs per year. Actually they are working break even.
Mr. Farrell spends 1 week a month travelling to the U.S. speaking to investors and telling the story.
Trinity will not announce contracts unless they are very very big. They are winning and loosing contracts all over the world each day.
By the way Orasure stopped being very open with releases about contracts and numbers. It seems they go the Trib way

Trinity has 3 distributors in China. There is NO HIV market over there because competitors have very cheap LOW QUALITY tests. There is a huge market for coag and infectious desease products.
They are very pleased with the Primus aquisition and the Gel hopefully gets approval Q2 2006. The market for testing A1C we can target is about 200 million $.
There is a guy on the board complaining about his „visit“ of RDI. When you arrived at the building, the familiy-company was already transfered and the guy who opened and was rude was no Trinity employee.
Actually 3 ongoing discussions for aquisitions. Two smaller and one bigger one. Two of them in an advanced stadium. If possible payment with cash and/or bank credit.
Institutional ownership difficult to say because of the ADR s , but IR company K Capital worked it from about 12 % to about 20 %. Add the 14% owned by management.
Management also shareholders and interested in inreasing shareprice.
Considering a 2nd facility in Bray just across the street. 1st facility running on full steam.
Valuation of a company in the diagnostic business depends on growth rates and products.
Based on that Trinity should be worth between 1.5 and 2 times revenues. ==> UNDERVALUED
Hope this helps
Ruffneck
Ich als begründer dieses Treads habe mich vor fast 2 Jahren von dieser Aktie verabschiedet.
Wie sich heute im nachhinein zeit. Bin ich zur richtigen Zeit ausgestiegen !
Hier nun ein Aktientipp von mir:
BAd Toys Holding A0ETLS
Vielleicht auch was für euch. Ist zwar nicht Biotech aber die Firma macht Gewinne und wird derzeit mit einem KGV von 6,25 für 2005 und 3,75 für 2006 bewertet !
Am 12.1.2006 gibt es hier auch noch einen Spinn off.
Als vielleicht ist ja jemand Interesse !! alles andere im BTYH board.
Gruß
Bibo
Wie sich heute im nachhinein zeit. Bin ich zur richtigen Zeit ausgestiegen !
Hier nun ein Aktientipp von mir:
BAd Toys Holding A0ETLS
Vielleicht auch was für euch. Ist zwar nicht Biotech aber die Firma macht Gewinne und wird derzeit mit einem KGV von 6,25 für 2005 und 3,75 für 2006 bewertet !
Am 12.1.2006 gibt es hier auch noch einen Spinn off.
Als vielleicht ist ja jemand Interesse !! alles andere im BTYH board.
Gruß
Bibo
Hallo an alle
Wo wierd TRIB hier in Deutschland gehandelt ?
Börseplatz???? Danke in voraus !
Grüße
Wo wierd TRIB hier in Deutschland gehandelt ?
Börseplatz???? Danke in voraus !

Grüße
hallo neue ID Nr. us8964381086 .........................nach Splitt!!!!!
Grüße
Grüße

oder VKN..... TRBA 



Hallo korrektur
Die neue vkn ist us8964383986 oder TRBA , es verden in De.
gehandelt und an der Nasdq
ciao
Die neue vkn ist us8964383986 oder TRBA , es verden in De.
gehandelt und an der Nasdq
ciao
Ich habe gerade in der Gerüchteküche gelesen das trib an Inova Diagnostic interessiert ist. Weiß jemand von euch mehr??
Ich habe bisher leider nichts über Umsatz und Mitarbeiteranzahl gefunden.
Wenn jemand mehr weiß wäre ich dankbar für ne Antwort.
gruss
vaka
Ich habe bisher leider nichts über Umsatz und Mitarbeiteranzahl gefunden.
Wenn jemand mehr weiß wäre ich dankbar für ne Antwort.
gruss
vaka
DUBLIN, IRELAND--(MARKET WIRE)--Feb 27, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2005 full year results on Monday, 6 March, 2006.
The Trinity Biotech management team will discuss full year financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
The Trinity Biotech management team will discuss full year financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
Plus 6% heute, fast typisch vor den Zahlen.
http://www.rte.ie/business/2006/Morningrep/download/0227good…
http://www.rte.ie/business/2006/Morningrep/download/0227good…
Revenues Increase 23.2%
DUBLIN, IRELAND--(MARKET WIRE)--Mar 6, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the year ended December 31, 2005.
Revenues in 2005 increased 23.2% to US$98.6 million compared to US$80.0 million in 2004. Operating profit before share option expenses increased from US$6.9 million in 2004 to US$8.0 million in the current year. Quarter on quarter operating profit before share options has increased from US$2.1 million in Quarter 3 to US$2.7 million in Quarter 4.
http://biz.yahoo.com/iw/060306/0112083.html
DUBLIN, IRELAND--(MARKET WIRE)--Mar 6, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE:TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the year ended December 31, 2005.
Revenues in 2005 increased 23.2% to US$98.6 million compared to US$80.0 million in 2004. Operating profit before share option expenses increased from US$6.9 million in 2004 to US$8.0 million in the current year. Quarter on quarter operating profit before share options has increased from US$2.1 million in Quarter 3 to US$2.7 million in Quarter 4.
http://biz.yahoo.com/iw/060306/0112083.html
Gewinn pro Aktie 0,095 US-D für das vierte Quartal 05 und 0,32 US-D fürs gesamte Jahr. Im vierten Quartal ist der Gewinn im Vergleich zum Vorjahr um fast 12 % gestiegen. Das ist ja endlich mal ein kleiner Lichtblick nach der bisherigen Gewinnstagnation der letzten Quartale.
Gewinnschätzung 2006 ? Was kann man erwarten? Ich gehe mal von 0,40 US-D aus. Bei einem KGV von 25 Kursziel 10 US-D, es sei denn die Gewinne steigen etwas stärker!! Können die nicht mal die Liquidität für Aktienrückkäufe nutzen, wenn schon keine Übernahmen erfolgen!!
Gewinnschätzung 2006 ? Was kann man erwarten? Ich gehe mal von 0,40 US-D aus. Bei einem KGV von 25 Kursziel 10 US-D, es sei denn die Gewinne steigen etwas stärker!! Können die nicht mal die Liquidität für Aktienrückkäufe nutzen, wenn schon keine Übernahmen erfolgen!!
DUBLIN, IRELAND--(MARKET WIRE)--Mar 9, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has written to the United States Food and Drug Administration (FDA) requesting a meeting to discuss the conditions under which the FDA might grant marketing clearance for Over The Counter (OTC) sale of the company`s UniGold HIV test. In addition Trinity will present at a meeting of the Blood Products Advisory Committee (BPAC) of the FDA in Washington tomorrow Friday 10th March 2006 and will present its position on home testing for HIV.
ADVERTISEMENT
Trinity`s UniGold HIV test is one of only two products which are FDA approved and CLIA waived and is marketed to hospitals, reference laboratories, physicians` offices, STD clinics, public health clinics and community-based organisations. The approval to distribute OTC would allow individuals to purchase the UniGold test in a drugstore and test themselves for HIV in the privacy of their own homes. Trinity`s UniGold HIV test is very simple to use. A drop of blood from a fingerstick is applied to a membrane in the test device. The membrane contains HIV antigens and a colour indicator. Within ten minutes the result can be read. A single red line indicates a negative result and two red lines determine a positive result.
In clinical trials on 9,000 patient samples, Trinity`s UniGold device had a sensitivity (ability to detect a true positive) of 100% and a specificity (ability to detect a true negative) of 99.7%. As the UniGold test uses a patented double antigen sandwich technique it is classified as a third generation test meaning that it can detect the presence of HIV antibodies in blood samples up to eight weeks earlier than second generation tests. Trinity`s UniGold HIV test is the only third generation rapid test available on the US market. Trinity`s product has the further advantage that it can be stored at room temperature for up to 15 months.
Commenting, Trinity Biotech`s President Brendan Farrell said, "We believe the time is now right for the home use of rapid HIV tests. Approximately 1.3 million Americans are infected with HIV and it is estimated that 300,000 of these people are unaware of their status. 40,000 new infections are occurring annually in the USA and traditional test methods have not slowed the rate of new infections. Trinity`s experience in sub-saharan Africa where 7 million people were tested in 2005 using our rapid tests has shown what a powerful tool rapid HIV tests can be in the fight to combat the spread of HIV.
Current OTC diagnostic products such as home tests for Glucose and Cholesterol use fingerstick whole blood as a sample, so the efficacy of the sample type has been well proven. Furthermore, as blood contains higher concentrations of HIV antibodies than other body fluids such as urine and oral fluid, this should be the sample of choice."
ADVERTISEMENT
Trinity`s UniGold HIV test is one of only two products which are FDA approved and CLIA waived and is marketed to hospitals, reference laboratories, physicians` offices, STD clinics, public health clinics and community-based organisations. The approval to distribute OTC would allow individuals to purchase the UniGold test in a drugstore and test themselves for HIV in the privacy of their own homes. Trinity`s UniGold HIV test is very simple to use. A drop of blood from a fingerstick is applied to a membrane in the test device. The membrane contains HIV antigens and a colour indicator. Within ten minutes the result can be read. A single red line indicates a negative result and two red lines determine a positive result.
In clinical trials on 9,000 patient samples, Trinity`s UniGold device had a sensitivity (ability to detect a true positive) of 100% and a specificity (ability to detect a true negative) of 99.7%. As the UniGold test uses a patented double antigen sandwich technique it is classified as a third generation test meaning that it can detect the presence of HIV antibodies in blood samples up to eight weeks earlier than second generation tests. Trinity`s UniGold HIV test is the only third generation rapid test available on the US market. Trinity`s product has the further advantage that it can be stored at room temperature for up to 15 months.
Commenting, Trinity Biotech`s President Brendan Farrell said, "We believe the time is now right for the home use of rapid HIV tests. Approximately 1.3 million Americans are infected with HIV and it is estimated that 300,000 of these people are unaware of their status. 40,000 new infections are occurring annually in the USA and traditional test methods have not slowed the rate of new infections. Trinity`s experience in sub-saharan Africa where 7 million people were tested in 2005 using our rapid tests has shown what a powerful tool rapid HIV tests can be in the fight to combat the spread of HIV.
Current OTC diagnostic products such as home tests for Glucose and Cholesterol use fingerstick whole blood as a sample, so the efficacy of the sample type has been well proven. Furthermore, as blood contains higher concentrations of HIV antibodies than other body fluids such as urine and oral fluid, this should be the sample of choice."
wurde der Aids-Test nicht schon Ende 2004 zugelassen oder betrifft das jetzt nur den freien Verkauf über Apotheken, damit die Leute zuhause testen können. Mal sehen, was in den nächsten Tagen noch für News folgen. Könnte doch für einen Umsatzschub sorgen, wenn der Test frei verkäuflich wäre. Bei der aktuellen Vogelgrippe-Hysterie wäre für unseren Kursverlauf wohl ein Vogelgrippe-Schnelltest prima 

[posting]20.601.177 von Dangerous am 09.03.06 17:25:59[/posting]Betrifft den freien Verkauf über Drugstores.
Im Yahoo Board reden die seit Tagen darüber.
Da hat man anscheinend besere Infos
Im Yahoo Board reden die seit Tagen darüber.
Da hat man anscheinend besere Infos

Versuche mal unter VK A0ET3S oder TRBA ciao 
Die Bezeichnung ist gehändert worden , ab Split zeit.
Salute

Die Bezeichnung ist gehändert worden , ab Split zeit.
Salute
Vakataka
Natütlich haben dort bessere INFO, der Preise wird in USA gemacht und nicht hier .Hier der MM versucht noch vor Öffnung der NASDAQ den preis runter zu drücken, es vird jeden Tag versucht .
Aber di aktionäre sind nicht doof .

Salute
Natütlich haben dort bessere INFO, der Preise wird in USA gemacht und nicht hier .Hier der MM versucht noch vor Öffnung der NASDAQ den preis runter zu drücken, es vird jeden Tag versucht .
Aber di aktionäre sind nicht doof .


Salute
Today`s the day!!!
by: charlie11908 (55/M/Ireland) 03/13/06 04:01 am
Msg: 61500 of 61500
Trinity Biotech (TRIB US)
Presents to FDA on home testing for HIV
Previous close: $9.25 Target: $8.40 (29/04/05; previously $13.60, issued 18/11/04) Analyst: barry.gallagher@davy.ie
Trinity Biotech will today (March 10th) present at a meeting of the Blood Products Advisory Committee of the FDA in
Washington and put forward its view on home testing for HIV.
Trinity announced on March 9th that it had written to the FDA requesting a meeting to discuss the conditions under which
the agency might grant clearance for OTC sale of the company`s UniGold HIV test. Trinity estimates this market could be
worth over $100m per annum.
Trinity`s UniGold HIV test is one of only two rapid HIV diagnostic products which are FDA-approved and with CLIA waiver and
is marketed to hospitals, reference laboratories, physicians` offices, STD clinics, public health clinics and community-based
organisations.
salute
by: charlie11908 (55/M/Ireland) 03/13/06 04:01 am
Msg: 61500 of 61500
Trinity Biotech (TRIB US)
Presents to FDA on home testing for HIV
Previous close: $9.25 Target: $8.40 (29/04/05; previously $13.60, issued 18/11/04) Analyst: barry.gallagher@davy.ie
Trinity Biotech will today (March 10th) present at a meeting of the Blood Products Advisory Committee of the FDA in
Washington and put forward its view on home testing for HIV.
Trinity announced on March 9th that it had written to the FDA requesting a meeting to discuss the conditions under which
the agency might grant clearance for OTC sale of the company`s UniGold HIV test. Trinity estimates this market could be
worth over $100m per annum.
Trinity`s UniGold HIV test is one of only two rapid HIV diagnostic products which are FDA-approved and with CLIA waiver and
is marketed to hospitals, reference laboratories, physicians` offices, STD clinics, public health clinics and community-based
organisations.
salute

Hallo
In Irland Preis pro Aktie =1.97E , und wiso hier in Deutschland auch nicht 1,97x4,1= (8,07) E
Was ist da los?
Wer kassiert di differenz?
Salute
In Irland Preis pro Aktie =1.97E , und wiso hier in Deutschland auch nicht 1,97x4,1= (8,07) E

Was ist da los?
Wer kassiert di differenz?
Salute
[posting]20.689.134 von serafino am 15.03.06 09:50:40[/posting]@serafino
Was meinst du mit "In Irland 1,97E...x4,1 = 8,07E"???
Meixnel
Was meinst du mit "In Irland 1,97E...x4,1 = 8,07E"???
Meixnel
Maxnel
Ich meinte ,dass ein Splitt stattgegeben hat 1 aktie zum 4,1 ADR neue VKN A0ET3S , und in Irland war di Aktie mit 1,97E notiert , also 1,94x4,1 = 8,077!!!!!

capisci?
salute
Ich meinte ,dass ein Splitt stattgegeben hat 1 aktie zum 4,1 ADR neue VKN A0ET3S , und in Irland war di Aktie mit 1,97E notiert , also 1,94x4,1 = 8,077!!!!!


capisci?

salute
[posting]20.695.002 von serafino am 15.03.06 12:25:10[/posting]@serafino
Der Split 1:4 war doch schon vor 9 Monaten im Juni 2005!
capisci?
Meixnelo
Der Split 1:4 war doch schon vor 9 Monaten im Juni 2005!
capisci?
Meixnelo
maxi
Na und? es hat Heute noch bestand .


Salute
Na und? es hat Heute noch bestand .


Salute
US Professional Portfolio Invest
Innovative, sicherheitsorientierte Kapitalanlage,
prognostizierte durchschnittliche Ausschüttung: 7% p.a.
15 Minuten verzögerte Kurse
Börse Symbol Währ. Vortag akt.Kurs Diff. % Handelsvol. gehand. St. Kurszeit
Frankfurt TRBA EUR 7,30 7,86 + 0,560 + 7,67 % 20.968 2.725 15. Mär 16:18
Stuttgart TRBA EUR 7,36 7,69 + 0,330 + 4,48 % 564 75 15. Mär 15:59
Berlin TRBA EUR 7,00 7,58 + 0,580 + 8,29 % - 0 15. Mär 09:15
Düsseldorf TRBA EUR 7,33 7,66 + 0,330 + 4,50 % - 0 15. Mär 09:18
München TRBA EUR 7,43 7,64 + 0,210 + 2,83 % - 0 15. Mär 09:02
Xetra TRBA EUR 6,60 6,95 + 0,350 + 5,30 % 521 0 15. Mär 13:27

 tia
tia
salute
Innovative, sicherheitsorientierte Kapitalanlage,
prognostizierte durchschnittliche Ausschüttung: 7% p.a.
15 Minuten verzögerte Kurse
Börse Symbol Währ. Vortag akt.Kurs Diff. % Handelsvol. gehand. St. Kurszeit
Frankfurt TRBA EUR 7,30 7,86 + 0,560 + 7,67 % 20.968 2.725 15. Mär 16:18
Stuttgart TRBA EUR 7,36 7,69 + 0,330 + 4,48 % 564 75 15. Mär 15:59
Berlin TRBA EUR 7,00 7,58 + 0,580 + 8,29 % - 0 15. Mär 09:15
Düsseldorf TRBA EUR 7,33 7,66 + 0,330 + 4,50 % - 0 15. Mär 09:18
München TRBA EUR 7,43 7,64 + 0,210 + 2,83 % - 0 15. Mär 09:02
Xetra TRBA EUR 6,60 6,95 + 0,350 + 5,30 % 521 0 15. Mär 13:27

 tia
tiasalute
[posting]20.704.291 von serafino am 15.03.06 17:46:29[/posting]schlaufino
Warum fragst du dann, wenn du es eh weisst???
Warum fragst du dann, wenn du es eh weisst???

[posting]20.689.134 von serafino am 15.03.06 09:50:40[/posting]Dann Vergleich mal den Kurs von Irland / USA.
Der Aktienkurs in $ ist umgerechnet fast immer niedriger wie der Aktienkurs in Irland.
Danke für den Hinweis auf das neue Kürzel, ist mir gar nicht aufgefallen
Der Aktienkurs in $ ist umgerechnet fast immer niedriger wie der Aktienkurs in Irland.
Danke für den Hinweis auf das neue Kürzel, ist mir gar nicht aufgefallen

Welcher Hirnie hat heute über Xetra 625 Stück zu € 6,83 verkauft? Rund 1 € unter aktuellem Kurs!:O
na klar der M M spielt und bringt was er kann 
Ich sagte schon der MM versucht den Ball ganz flach zu Halten . warte mal ab bald wird AVAN explodieren
warte mal ab bald wird AVAN explodieren
Salute

Ich sagte schon der MM versucht den Ball ganz flach zu Halten .
 warte mal ab bald wird AVAN explodieren
warte mal ab bald wird AVAN explodieren Salute
Pardon natürlich meinte ich TRIB ,kann passieren 

genau um 13,30 MEZ tia der MMaus der USA arbeitet mit alle Tricksen 
capisci?
salute

capisci?
salute
[posting]8.591.290 von honorar am 12.02.03 20:26:06[/posting]@ honorar
Wie waren die Zahlen? Weiß das jemand?
Danke!
Meixnel
Wie waren die Zahlen? Weiß das jemand?
Danke!
Meixnel
siehe 567! Dort sind die Quartalszahlen nachzulesen.
10:35 AM Trinity Biotech to Sell $25 Million of American Depositary Shares - Market Wire
Trinity Biotech plc (NASDAQ: TRIB), (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, is pleased to announce that it has obtained commitments from a select group of institutional investors in the USA and Europe to purchase approximately 2.675 million American Depositary Shares at a price of $8.60 per ADS in a registered public offering. This represents a discount of 4.5% to the closing price on Nasdaq on April 5, 2006.
In addition, Ronan O' Caoimh, Trinity Biotech's CEO, has agreed to purchase approximately 223,460 American Depositary Shares from Trinity Biotech, at a price of $8.95 per ADS, the closing bid price on April 5, 2006, for total proceeds of approximately $2 million. The closing for this purchase is scheduled to occur coincident with the closing of the sale to institutional investors.
Trinity Biotech expects that the net proceeds of the offering will be approximately $24 million after deducting the placement agency fees and all estimated offering expenses that are payable by Trinity Biotech. The closing is expected to take place on Tuesday, April 11, 2006, subject to the satisfaction of customary closing conditions. Roth Capital Partners, LLC, acted as lead placement agent, with J&E Davy acting as co-placement agent for this offering.
Trinity Biotech currently intends to use these funds for general corporate purposes and business development including potential acquisitions of companies, products and technologies that complement our business.
Ronan O'Caoimh commented, "We are delighted with the level of support we have received from an internationally diverse, institutional investor group. The proceeds of this offering together with our existing cash resources, operating cashflows and banking facilities will enable us to target larger acquisition opportunities than previously and to sustain our growth ambitions."
The American Depositary Shares are being sold under a previously filed shelf registration statement, which the Securities and Exchange Commission has declared effective. This press release shall not constitute an offer to sell nor the solicitation of an offer to buy, nor shall there be any sales of these securities in any jurisdiction in which such an offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction.
Trinity Biotech plc (NASDAQ: TRIB), (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, is pleased to announce that it has obtained commitments from a select group of institutional investors in the USA and Europe to purchase approximately 2.675 million American Depositary Shares at a price of $8.60 per ADS in a registered public offering. This represents a discount of 4.5% to the closing price on Nasdaq on April 5, 2006.
In addition, Ronan O' Caoimh, Trinity Biotech's CEO, has agreed to purchase approximately 223,460 American Depositary Shares from Trinity Biotech, at a price of $8.95 per ADS, the closing bid price on April 5, 2006, for total proceeds of approximately $2 million. The closing for this purchase is scheduled to occur coincident with the closing of the sale to institutional investors.
Trinity Biotech expects that the net proceeds of the offering will be approximately $24 million after deducting the placement agency fees and all estimated offering expenses that are payable by Trinity Biotech. The closing is expected to take place on Tuesday, April 11, 2006, subject to the satisfaction of customary closing conditions. Roth Capital Partners, LLC, acted as lead placement agent, with J&E Davy acting as co-placement agent for this offering.
Trinity Biotech currently intends to use these funds for general corporate purposes and business development including potential acquisitions of companies, products and technologies that complement our business.
Ronan O'Caoimh commented, "We are delighted with the level of support we have received from an internationally diverse, institutional investor group. The proceeds of this offering together with our existing cash resources, operating cashflows and banking facilities will enable us to target larger acquisition opportunities than previously and to sustain our growth ambitions."
The American Depositary Shares are being sold under a previously filed shelf registration statement, which the Securities and Exchange Commission has declared effective. This press release shall not constitute an offer to sell nor the solicitation of an offer to buy, nor shall there be any sales of these securities in any jurisdiction in which such an offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction.
Antwort auf Beitrag Nr.: 21.109.110 von Vakataka am 06.04.06 19:20:10Dillution? Ja, aber nicht nervös werden
Bis zu 30% werden jetzt von Insidern und Institutionellen gehalten. (ohne Gewähr )
Ceo kauft auch eigene Aktien, wieder!
Die Instis haben bei den geringen Umsätzen kaum Möglichkeiten ne vernünftige Aktienanzahl zu kaufen ohne den Kurs zu treiben, von daher ist das nachvollziehbar, aber warum kaufen die Instis diese langweilige Aktie ?
Bestimmt nicht zum Vergnügen!
Egal, das kann man so oder so sehen.
Fakt ist das Trinity jetzt 34 Mio USD cash hat!
Die nächste Aquisition sollten wir bald sehen.
Die Frage ist
Wer wird gekauft?
oder
Was wird von wem gekauft?
Außerdem erhoffen wir uns noch einiges vo
Primus Diabetis
Bird Flu Test ??
Antworten erwünscht, ist ganz schön langweilig hier
Gruß
vaka

Bis zu 30% werden jetzt von Insidern und Institutionellen gehalten. (ohne Gewähr )
Ceo kauft auch eigene Aktien, wieder!
Die Instis haben bei den geringen Umsätzen kaum Möglichkeiten ne vernünftige Aktienanzahl zu kaufen ohne den Kurs zu treiben, von daher ist das nachvollziehbar, aber warum kaufen die Instis diese langweilige Aktie ?
Bestimmt nicht zum Vergnügen!
Egal, das kann man so oder so sehen.
Fakt ist das Trinity jetzt 34 Mio USD cash hat!
Die nächste Aquisition sollten wir bald sehen.
Die Frage ist
Wer wird gekauft?
oder
Was wird von wem gekauft?
Außerdem erhoffen wir uns noch einiges vo
Primus Diabetis
Bird Flu Test ??
Antworten erwünscht, ist ganz schön langweilig hier

Gruß
vaka
Antwort auf Beitrag Nr.: 21.111.326 von Vakataka am 06.04.06 21:34:05270 000 Aktien gehandelt,
plus 5,5%
werde mir doch noch ein paar ins Depot legen, nur zum traden
plus 5,5%
werde mir doch noch ein paar ins Depot legen, nur zum traden

Antwort auf Beitrag Nr.: 21.111.326 von Vakataka am 06.04.06 21:34:05Hallo Vaka,
auf den Vogelgrippe-Virentest würde ich nur dann etwas geben, wenn man die genauen Meßzeiträume kenn. Qiagen hat auch einen auf den Markt gebracht, wo der Testzeitraum nur einige Stunden beträgt. Bisherige Test brauchen für die Auswertung bis zu einer Woche. Totzdem könnte sich TRIB eine kleine Scheibe vom Kuchen abschneiden und auch etwas Fantasie in die Aktie kommen. Die IR-Arbeit sollte deutlich verbessert werden. In TRIB schlummert so viel Potential.
Zu den Major Holders 2005
HEARTLAND ADVISORS INC. 388,000 2.79 $3,166,080 31-Dec-05
PERRITT CAPITAL MANAGEMENT, INC. 359,301 2.59 $2,931,896 31-Dec-05
HIGHBRIDGE CAPITAL MANAGEMENT, LLC. 289,721 2.08 $2,364,123 31-Dec-05
FMR CORPORATION (FIDELITY MANAGEMENT & RESEARCH CORP) 227,325 1.64 $1,854,972 31-Dec-05
CAXTON ASSOCIATES, L.L.C. 46,712 .34 $381,169 31-Dec-05
Triathlon Fund Management, LLC 25,000 .18 $204,000 31-Dec-05
GARTMORE MUTUAL FUND CAPITAL TRUST 19,425 .14 $158,508 31-Dec-05
Gartmore Global Asset Management Ltd 19,425 .14 $158,508 31-Dec-05
HBK INVESTMENTS, L.P. 11,476 .08 $93,644 31-Dec-05
ALLIED IRISH BANKS, P.L.C. 5,100 .04 $41,616 31-Dec-05
Da die erfolgreichste Fondsgesellschaft der Welt mit Fidelity an Board ist, spricht das für die Qualität des Unternehmens.
Wenn endlich auch mal die Gewinne genauso wachsen wie die Umsätze, dann sehen wir bestimmt wieder Kurse im Bereich 15 Dollar.
Die mögliche Übernahme wird wohl etwas größer Ausfallen. Hoffentlich nicht zu teuer erkauft, sonst steigen die Gewinne weiterhin unterproportional!
auf den Vogelgrippe-Virentest würde ich nur dann etwas geben, wenn man die genauen Meßzeiträume kenn. Qiagen hat auch einen auf den Markt gebracht, wo der Testzeitraum nur einige Stunden beträgt. Bisherige Test brauchen für die Auswertung bis zu einer Woche. Totzdem könnte sich TRIB eine kleine Scheibe vom Kuchen abschneiden und auch etwas Fantasie in die Aktie kommen. Die IR-Arbeit sollte deutlich verbessert werden. In TRIB schlummert so viel Potential.
Zu den Major Holders 2005
HEARTLAND ADVISORS INC. 388,000 2.79 $3,166,080 31-Dec-05
PERRITT CAPITAL MANAGEMENT, INC. 359,301 2.59 $2,931,896 31-Dec-05
HIGHBRIDGE CAPITAL MANAGEMENT, LLC. 289,721 2.08 $2,364,123 31-Dec-05
FMR CORPORATION (FIDELITY MANAGEMENT & RESEARCH CORP) 227,325 1.64 $1,854,972 31-Dec-05
CAXTON ASSOCIATES, L.L.C. 46,712 .34 $381,169 31-Dec-05
Triathlon Fund Management, LLC 25,000 .18 $204,000 31-Dec-05
GARTMORE MUTUAL FUND CAPITAL TRUST 19,425 .14 $158,508 31-Dec-05
Gartmore Global Asset Management Ltd 19,425 .14 $158,508 31-Dec-05
HBK INVESTMENTS, L.P. 11,476 .08 $93,644 31-Dec-05
ALLIED IRISH BANKS, P.L.C. 5,100 .04 $41,616 31-Dec-05
Da die erfolgreichste Fondsgesellschaft der Welt mit Fidelity an Board ist, spricht das für die Qualität des Unternehmens.
Wenn endlich auch mal die Gewinne genauso wachsen wie die Umsätze, dann sehen wir bestimmt wieder Kurse im Bereich 15 Dollar.
Die mögliche Übernahme wird wohl etwas größer Ausfallen. Hoffentlich nicht zu teuer erkauft, sonst steigen die Gewinne weiterhin unterproportional!
Antwort auf Beitrag Nr.: 21.118.099 von Dangerous am 07.04.06 12:28:38Hi Dangerous,
nach viel Spekulationen hat es anscheinend der erste Ami im Yahoo Board gemerkt um welche Firma es geht.
Könnte der Hammer werden wenn die anderen wach werden. Hoffe ich zumindest.
Trinity Umsatzplus von 60%, das wär doch was.
Aber wir unterhalten uns lieber per Bm, hab keinen Bock hier Alleinunterhalter zu spielen
vaka
nach viel Spekulationen hat es anscheinend der erste Ami im Yahoo Board gemerkt um welche Firma es geht.
Könnte der Hammer werden wenn die anderen wach werden. Hoffe ich zumindest.
Trinity Umsatzplus von 60%, das wär doch was.
Aber wir unterhalten uns lieber per Bm, hab keinen Bock hier Alleinunterhalter zu spielen

vaka
Antwort auf Beitrag Nr.: 21.227.928 von Vakataka am 18.04.06 17:58:46Hi Vaka,
ab und zu BM ist okay. Aber ein wenig müssen wir auch das Forum füllen, damit die stillen Mitleser auch Gesprächsstoff haben.
60 % Umsatzwachstum das wäre ja Wahnsinn!!! Wenn die Gewinne genauso steigen, dann sitzen wir gerade auf einer neuen Zündstufe für unsere Rakete. Mal sehen, wie die Quartalszahlen ausfallen und die mögliche Übernahme.
beste Grüße
ab und zu BM ist okay. Aber ein wenig müssen wir auch das Forum füllen, damit die stillen Mitleser auch Gesprächsstoff haben.
60 % Umsatzwachstum das wäre ja Wahnsinn!!! Wenn die Gewinne genauso steigen, dann sitzen wir gerade auf einer neuen Zündstufe für unsere Rakete. Mal sehen, wie die Quartalszahlen ausfallen und die mögliche Übernahme.
beste Grüße
Antwort auf Beitrag Nr.: 21.229.961 von Dangerous am 18.04.06 19:57:13Dangerous
Rakete ist gut
Für die Q Zahlen erwarte ich eigentlich nichts überraschendes.
Interessant wird sein wieviel die Iren für den Umsatzzuwachs ausgeben.
Da die aber in den letzten Jahren günstig eingekauft haben erwarte ich hier keine Horrorsummen.
Aber um hier nicht in die reine Spekulation abzudriften sollten die jetzt langsam die PR raushauen.
Dann können wir drüber herziehen
@all
verkaufen würde ich jetzt nicht, wer sich nicht traut zu kaufen sollte die Aktie noch ein bischen halten.
Die 10$ sollten wir demnächst locker hinter uns lassen.
Hoffe ich.
Gruss
vaka
Rakete ist gut

Für die Q Zahlen erwarte ich eigentlich nichts überraschendes.
Interessant wird sein wieviel die Iren für den Umsatzzuwachs ausgeben.
Da die aber in den letzten Jahren günstig eingekauft haben erwarte ich hier keine Horrorsummen.
Aber um hier nicht in die reine Spekulation abzudriften sollten die jetzt langsam die PR raushauen.
Dann können wir drüber herziehen

@all
verkaufen würde ich jetzt nicht, wer sich nicht traut zu kaufen sollte die Aktie noch ein bischen halten.
Die 10$ sollten wir demnächst locker hinter uns lassen.
Hoffe ich.
Gruss
vaka
Antwort auf Beitrag Nr.: 21.232.210 von Vakataka am 18.04.06 22:19:05Hallo Vaka,
absolute Zustimmung. Steht schon der Termin für die Veröffentlichung der Quartalszahlen fest.
Die mögliche Übernahme wird dann die Fantasie in die Aktie bringen. We will see.
Bye Remo
absolute Zustimmung. Steht schon der Termin für die Veröffentlichung der Quartalszahlen fest.
Die mögliche Übernahme wird dann die Fantasie in die Aktie bringen. We will see.
Bye Remo
Antwort auf Beitrag Nr.: 21.235.817 von Dangerous am 19.04.06 10:27:19Q1 2006 Trinity Biotech Earnings Release
27 April 2006*
AGM 13 June 2006
Q2 2006 Trinity Biotech Earnings Release
27 July 2006*
Q3 2006 Trinity Biotech Earnings Release
26 Oct 2006*
(*Preliminary Reporting Dates)
27 April 2006*
AGM 13 June 2006
Q2 2006 Trinity Biotech Earnings Release
27 July 2006*
Q3 2006 Trinity Biotech Earnings Release
26 Oct 2006*
(*Preliminary Reporting Dates)
Antwort auf Beitrag Nr.: 21.242.100 von Vakataka am 19.04.06 17:12:28danke vielleicht kommt ja etwas Pre-Earnings-Fantasie in die Aktie. Ob dann wohl die mögliche Übernahme bekannt gegeben wird. Zum Glück brauchen wir nicht mehr lange warten.
Antwort auf Beitrag Nr.: 21.245.372 von Dangerous am 19.04.06 20:50:58Dangerous
Ich bekomme das irgendwie nicht verlinkt,
kopiers Dir mal und lies den Artikel
http://today.reuters.com/news/articlenews.aspx?type=reutersE…
168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=&cap=&sz=13" target="_blank" rel="nofollow ugc noopener">http://today.reuters.com/news/articlenews.aspx?type=reutersE…
168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=&cap=&sz=13
Ich bekomme das irgendwie nicht verlinkt,
kopiers Dir mal und lies den Artikel
http://today.reuters.com/news/articlenews.aspx?type=reutersE…
168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=&cap=&sz=13" target="_blank" rel="nofollow ugc noopener">http://today.reuters.com/news/articlenews.aspx?type=reutersE…
168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=&cap=&sz=13
Antwort auf Beitrag Nr.: 21.246.868 von Vakataka am 19.04.06 22:26:36Nochmal
http://today.reuters.com/news/articlenews.aspx?type=reutersE… 11T132831Z_01_PAC006168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=∩=&s z=13
http://today.reuters.com/news/articlenews.aspx?type=reutersE… 11T132831Z_01_PAC006168_RTRUKOC_0_US-BIOMERIEUX.xml&pageNumber=0&imageid=∩=&s z=13
Antwort auf Beitrag Nr.: 21.246.893 von Vakataka am 19.04.06 22:28:28Hallo Vaka,
leider geht der zweite Link auch nicht
We're sorry... this story is not currently available
Du hast ja schon mal über die BM dazu was geschrieben. Wenn die News in den nächsten Tagen kommen, reicht mir das aus. TRIB ist ein Long für mich.
Vielen Dank für deine Mühe. Sehr nett von dir.
beste Grüße Remo
leider geht der zweite Link auch nicht
We're sorry... this story is not currently available
Du hast ja schon mal über die BM dazu was geschrieben. Wenn die News in den nächsten Tagen kommen, reicht mir das aus. TRIB ist ein Long für mich.
Vielen Dank für deine Mühe. Sehr nett von dir.
beste Grüße Remo
Trinity Biotech Announces Quarter 1 Results; Revenues Increase 16.4% and Profits Increase 23.5%
Trinity Biotech plc (NASDAQ: TRIB) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended March 31, 2006.
Revenues in quarter 1, 2006 increased 16.4% to US$24.7 million compared to US$21.2 million in quarter 1 2005. During the same period operating profit before share option expenses increased from US$1.4 million to US$2.0 million. Profit after tax increased from US$1.0 million in quarter 1 2005 to US$1.2 million in quarter 1 2006, representing an increase of 23.5%
Revenues for the quarter by key product area were as follows:
2005 2006
US$000 US$000 % Increase
Infectious Diseases 9,422 9,992 6.0
Coagulation 7,325 6,821 -6.9
Clinical Chemistry 1,454 4,007 175.6
Point of Care 3,026 3,894 28.7
Total 21,227 24,714 16.4
Revenues for the quarter by geographical location were as follows:
2005 2006
US$000 US$000 % Increase
USA 10,812 12,266 13.4
Europe 6,017 6,966 15.8
Asia / Africa 4,398 5,482 24.6
Total 21,227 24,714 16.4
Gross profit for the quarter amounted to US$12.5 million, representing a gross margin of 51%. This compares to a gross profit of US$10.3 million and gross margin of 49% for the same period in 2005.
R&D expenses have increased from US$1.3 million in quarter 1 2005 to US$1.4 million in quarter 1 2006. The increase in selling, general and administrative expenses from US$7.6 million in 2005 to US$9.3 million in the current year is primarily attributable to the impact of the acquisitions of RDI and Primus in March 2005 and July 2005 respectively.
The net effect of the above was an operating profit before share options of US$2.0 million for the quarter compared to US$1.4 million for the quarter 1 2005 which represents a margin of 7.9% and 6.8% respectively.
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "We have experienced strong growth in both revenues and profits versus the comparable period in 2005. This growth has been achieved in each of our geographical markets. In this context we are particularly pleased with the increase in our operating profit before share option expense which has increased by 36%. We are confident of achieving our organic growth target for the year of 12%."
Ronan O'Caoimh, CEO, commented that, "During the quarter we attended a meeting of the Blood Products Advisory Committee (BPAC) and presented our position on home testing for HIV. Our UniGold HIV test is one of only two products which are FDA approved and CLIA waived and potentially suitable for this estimated US$100 million OTC market. Trinity has submitted its clinical trial plans to the FDA and will be discussing these with the FDA in May. It is our intention to commence clinical trials following this meeting once protocols have been agreed with the FDA.
"We are also very pleased with the level of support received for our recent US$25m share offering. The offering was oversubscribed and was taken up by institutions in the USA, the UK, Switzerland and Ireland. The proceeds raised, together with existing cash resources, operating cashflows and bank facilities will enable the company to target larger earnings accretive acquisitions in order to achieve its growth strategy."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Income Statements
(US$000's except share data) Three Months Three Months
Ended March Ended March
31, 2006 31, 2005
(unaudited) (unaudited)
Revenues 24,714 21,227
Cost of sales (12,200) (10,866)
Cost of sales - share based payments (26) (16)
---------- ----------
Gross profit 12,488 10,345
Other operating income 60 43
Research & development expenses (1,365) (1,320)
Selling , general and administrative expenses (9,253) (7,646)
Indirect share based payments (344) (196)
---------- ----------
Operating profit 1,586 1,226
Financial income 154 113
Financial expenses (342) (238)
---------- ----------
Net financing costs (188) (125)
---------- ----------
Profit before tax 1,398 1,101
Income tax (159) (98)
---------- ----------
Profit for the period 1,239 1,003
Earnings per ADR (US cents) 8.0 7.2
Earnings per share (US cents) 2.0 1.8
Diluted earnings per ADR (US cents) 8.0 7.2
Diluted earnings per share (US cents) 2.0 1.8
Weighted average no. of shares used in
computing earnings per share 61,461,903 56,954,314
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
Consolidated Balance Sheets
March 31, December 31,
2006 2005
US$ '000 US$ '000
(unaudited)
ASSETS
Non-current assets
Property, plant and equipment 19,003 19,202
Goodwill and intangible assets 85,914 85,197
Deferred tax assets 3,314 3,277
Other assets 61 61
---------- ----------
Total non-current assets 108,292 107,737
---------- ----------
Current assets
Inventories 38,270 36,450
Trade and other receivables 21,562 20,885
Income tax receivable 688 649
Financial assets - restricted cash 9,000 9,000
Cash and cash equivalents 757 9,881
Derivative financial instruments 33 -
---------- ----------
Total current assets 70,310 76,865
---------- ----------
TOTAL ASSETS 178,602 184,602
========== ==========
EQUITY AND LIABILITIES
Equity attributable to the equity holders
of the parent
Share capital 830 830
Share premium 124,273 124,227
Retained earnings 7,888 6,280
Translation reserve (1,497) (1,622)
Other reserves 3,972 3,903
---------- ----------
Total equity 135,466 133,618
---------- ----------
Current liabilities
Interest-bearing loans and borrowings 7,726 7,720
Convertible notes 7,257 7,203
Income tax payable 385 260
Trade and other payables 11,267 12,768
Other financial liabilities 302 3,707
Derivative financial instruments - 44
Provisions 236 199
---------- ----------
Total current liabilities 27,173 31,901
---------- ----------
Non-current liabilities
Interest-bearing loans and borrowings 9,054 10,369
Convertible notes - 1,836
Other income tax payable 49 48
Other payables 102 102
Deferred tax liabilities 6,758 6,728
---------- ----------
Total non-current liabilities 15,963 19,083
---------- ----------
TOTAL LIABILITIES 43,136 50,984
---------- ----------
TOTAL EQUITY AND LIABILITIES 178,602 184,602
========== ==========
Prepared in accordance with International Financial Reporting Standards.
Source: Market Wire (April 27, 2006 - 8:16 AM EDT)
News by QuoteMedia
www.quotemedia.com
Trinity Biotech plc (NASDAQ: TRIB) (ISE: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended March 31, 2006.
Revenues in quarter 1, 2006 increased 16.4% to US$24.7 million compared to US$21.2 million in quarter 1 2005. During the same period operating profit before share option expenses increased from US$1.4 million to US$2.0 million. Profit after tax increased from US$1.0 million in quarter 1 2005 to US$1.2 million in quarter 1 2006, representing an increase of 23.5%
Revenues for the quarter by key product area were as follows:
2005 2006
US$000 US$000 % Increase
Infectious Diseases 9,422 9,992 6.0
Coagulation 7,325 6,821 -6.9
Clinical Chemistry 1,454 4,007 175.6
Point of Care 3,026 3,894 28.7
Total 21,227 24,714 16.4
Revenues for the quarter by geographical location were as follows:
2005 2006
US$000 US$000 % Increase
USA 10,812 12,266 13.4
Europe 6,017 6,966 15.8
Asia / Africa 4,398 5,482 24.6
Total 21,227 24,714 16.4
Gross profit for the quarter amounted to US$12.5 million, representing a gross margin of 51%. This compares to a gross profit of US$10.3 million and gross margin of 49% for the same period in 2005.
R&D expenses have increased from US$1.3 million in quarter 1 2005 to US$1.4 million in quarter 1 2006. The increase in selling, general and administrative expenses from US$7.6 million in 2005 to US$9.3 million in the current year is primarily attributable to the impact of the acquisitions of RDI and Primus in March 2005 and July 2005 respectively.
The net effect of the above was an operating profit before share options of US$2.0 million for the quarter compared to US$1.4 million for the quarter 1 2005 which represents a margin of 7.9% and 6.8% respectively.
Commenting on the results, Rory Nealon, Chief Financial Officer, said, "We have experienced strong growth in both revenues and profits versus the comparable period in 2005. This growth has been achieved in each of our geographical markets. In this context we are particularly pleased with the increase in our operating profit before share option expense which has increased by 36%. We are confident of achieving our organic growth target for the year of 12%."
Ronan O'Caoimh, CEO, commented that, "During the quarter we attended a meeting of the Blood Products Advisory Committee (BPAC) and presented our position on home testing for HIV. Our UniGold HIV test is one of only two products which are FDA approved and CLIA waived and potentially suitable for this estimated US$100 million OTC market. Trinity has submitted its clinical trial plans to the FDA and will be discussing these with the FDA in May. It is our intention to commence clinical trials following this meeting once protocols have been agreed with the FDA.
"We are also very pleased with the level of support received for our recent US$25m share offering. The offering was oversubscribed and was taken up by institutions in the USA, the UK, Switzerland and Ireland. The proceeds raised, together with existing cash resources, operating cashflows and bank facilities will enable the company to target larger earnings accretive acquisitions in order to achieve its growth strategy."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own salesforce and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Income Statements
(US$000's except share data) Three Months Three Months
Ended March Ended March
31, 2006 31, 2005
(unaudited) (unaudited)
Revenues 24,714 21,227
Cost of sales (12,200) (10,866)
Cost of sales - share based payments (26) (16)
---------- ----------
Gross profit 12,488 10,345
Other operating income 60 43
Research & development expenses (1,365) (1,320)
Selling , general and administrative expenses (9,253) (7,646)
Indirect share based payments (344) (196)
---------- ----------
Operating profit 1,586 1,226
Financial income 154 113
Financial expenses (342) (238)
---------- ----------
Net financing costs (188) (125)
---------- ----------
Profit before tax 1,398 1,101
Income tax (159) (98)
---------- ----------
Profit for the period 1,239 1,003
Earnings per ADR (US cents) 8.0 7.2
Earnings per share (US cents) 2.0 1.8
Diluted earnings per ADR (US cents) 8.0 7.2
Diluted earnings per share (US cents) 2.0 1.8
Weighted average no. of shares used in
computing earnings per share 61,461,903 56,954,314
Prepared in accordance with International Financial Reporting Standards.
Trinity Biotech plc
Consolidated Balance Sheets
March 31, December 31,
2006 2005
US$ '000 US$ '000
(unaudited)
ASSETS
Non-current assets
Property, plant and equipment 19,003 19,202
Goodwill and intangible assets 85,914 85,197
Deferred tax assets 3,314 3,277
Other assets 61 61
---------- ----------
Total non-current assets 108,292 107,737
---------- ----------
Current assets
Inventories 38,270 36,450
Trade and other receivables 21,562 20,885
Income tax receivable 688 649
Financial assets - restricted cash 9,000 9,000
Cash and cash equivalents 757 9,881
Derivative financial instruments 33 -
---------- ----------
Total current assets 70,310 76,865
---------- ----------
TOTAL ASSETS 178,602 184,602
========== ==========
EQUITY AND LIABILITIES
Equity attributable to the equity holders
of the parent
Share capital 830 830
Share premium 124,273 124,227
Retained earnings 7,888 6,280
Translation reserve (1,497) (1,622)
Other reserves 3,972 3,903
---------- ----------
Total equity 135,466 133,618
---------- ----------
Current liabilities
Interest-bearing loans and borrowings 7,726 7,720
Convertible notes 7,257 7,203
Income tax payable 385 260
Trade and other payables 11,267 12,768
Other financial liabilities 302 3,707
Derivative financial instruments - 44
Provisions 236 199
---------- ----------
Total current liabilities 27,173 31,901
---------- ----------
Non-current liabilities
Interest-bearing loans and borrowings 9,054 10,369
Convertible notes - 1,836
Other income tax payable 49 48
Other payables 102 102
Deferred tax liabilities 6,758 6,728
---------- ----------
Total non-current liabilities 15,963 19,083
---------- ----------
TOTAL LIABILITIES 43,136 50,984
---------- ----------
TOTAL EQUITY AND LIABILITIES 178,602 184,602
========== ==========
Prepared in accordance with International Financial Reporting Standards.
Source: Market Wire (April 27, 2006 - 8:16 AM EDT)
News by QuoteMedia
www.quotemedia.com
Antwort auf Beitrag Nr.: 21.348.354 von Vakataka am 27.04.06 16:30:23Ich habe auch auf 10cent/eps gehofft, aber das die Amis jetzt so abladen
Minus 5% in USA. Da kommen die Aktien nur so geflogen. 177 000 bis jetzt.
Bei 8,5 sind dann wieder größere Käufe. Mal schaun wo wir heute schließen.
Echt grausam.
In ein paar Tagen/ Wochen sieht das ganz anders aus.

Minus 5% in USA. Da kommen die Aktien nur so geflogen. 177 000 bis jetzt.
Bei 8,5 sind dann wieder größere Käufe. Mal schaun wo wir heute schließen.
Echt grausam.
In ein paar Tagen/ Wochen sieht das ganz anders aus.
Hier noch was lesenswertes von jemanden der den CC mitgehört hat.
by: tallyrand2003
Long-Term Sentiment: Buy 04/27/06 12:38 pm
Msg: 62362 of 62362
My take on the CC and TRIB's future is:
1. Gel approved in Q2 - profits projected in 2007 and 2008
2. Dilution in Q2 going forward until acquistion, organic growth (at 12%) and increased margins (from 8% to 12%)offset the dilution. The dilution was about 17%. I estimate this year's earnings to approximate last years (not great news hence the drop in price). They said the organic growth wouild translate into $5M to $6M bottom line.
3. Over the counter HIV, diabetes, 2 additional FDA products in the pipeline, sales force eficiency will over the next 12 to 24 months produce signficiant bottom line growth. Western Blot to be approved Q1 of 2007.
4. Wampole litigation going well. Depositions and discovery support breach of contract by Wampole. Only question will be measuring damages (IMO) which may be nominal or some millions of dollars.
5. Lots of US analysts/managers on the call. Great news.
6. This is a great stock to buy now and hold for 2 years. For those of us who have owned it for 8 years we are disappointed that top line growth is done at the expense of bottom line profits. I am more than impatient at the timeline and of course future uncertainties.
7. There will be a pop when diabetes test is approved. It is a $100M/year US market with only one other player. Sell then and buy back when TRIB drops back.
All the above my thoughts and info gleaned from the CC. I share the optimism of those who are long and share the skepticism of others. If TRIB closes 2006 at $10 it is a substantial increase over current price. The question is will it (or even better going into 2007) or is money spent better elsewhere?
Only hindsight can ever answer that.
Good luck to us all.
http://finance.messages.yahoo.com/bbs?.mm=FN&action=m&board=…
by: tallyrand2003
Long-Term Sentiment: Buy 04/27/06 12:38 pm
Msg: 62362 of 62362
My take on the CC and TRIB's future is:
1. Gel approved in Q2 - profits projected in 2007 and 2008
2. Dilution in Q2 going forward until acquistion, organic growth (at 12%) and increased margins (from 8% to 12%)offset the dilution. The dilution was about 17%. I estimate this year's earnings to approximate last years (not great news hence the drop in price). They said the organic growth wouild translate into $5M to $6M bottom line.
3. Over the counter HIV, diabetes, 2 additional FDA products in the pipeline, sales force eficiency will over the next 12 to 24 months produce signficiant bottom line growth. Western Blot to be approved Q1 of 2007.
4. Wampole litigation going well. Depositions and discovery support breach of contract by Wampole. Only question will be measuring damages (IMO) which may be nominal or some millions of dollars.
5. Lots of US analysts/managers on the call. Great news.
6. This is a great stock to buy now and hold for 2 years. For those of us who have owned it for 8 years we are disappointed that top line growth is done at the expense of bottom line profits. I am more than impatient at the timeline and of course future uncertainties.
7. There will be a pop when diabetes test is approved. It is a $100M/year US market with only one other player. Sell then and buy back when TRIB drops back.
All the above my thoughts and info gleaned from the CC. I share the optimism of those who are long and share the skepticism of others. If TRIB closes 2006 at $10 it is a substantial increase over current price. The question is will it (or even better going into 2007) or is money spent better elsewhere?
Only hindsight can ever answer that.
Good luck to us all.
http://finance.messages.yahoo.com/bbs?.mm=FN&action=m&board=…
Antwort auf Beitrag Nr.: 21.351.437 von Vakataka am 27.04.06 18:54:54350 000 , entspricht 1,4 Mio ADR.
Wahnsinn.
Und immer noch wird bei 8,5 mit großen Blöcken aufgefangen.
Sieht so aus ich bin der einzige der hier mitleidet.
Na vaka, denn mal Prost
Wahnsinn.
Und immer noch wird bei 8,5 mit großen Blöcken aufgefangen.
Sieht so aus ich bin der einzige der hier mitleidet.
Na vaka, denn mal Prost

Antwort auf Beitrag Nr.: 21.352.898 von Vakataka am 27.04.06 20:09:45Hallo Vaka,
das habe ich geahnt, dass unter 10 Cent geschmissen wird. Die Firma hat so gute Voraussetzungen und Zukunftsaussichten, aber so langsam verlässt mich die Zuversicht bei permanent schwachen Quartalszahlen. Ich verstehe einfach nicht, warum die Umsätze ständig steigen und der Gewinn stagniert. Ich bin wirklich enttäuscht vom Ergebnis. Wie lange sollen die Aktionäre denn noch warten, bis mal was positives rüber kommt. Die ewiger Warterei auf bessere Zeiten nervt ganz schön. Dann wird es bald mal Zeit für eine Ad-Hoc zur Übernahme, damit die Aktie nicht unter den flachen Aufwärtstrend fällt. Eigentlich war TRIB ein Long für mich, aber wenn nicht die nächsten Quartale die Gewinne steigen, schmeisse ich auch. Man kann leider bei anderen Werten besseres Geld verdienen. Bisher sind es ja nur Verluste. Naja morgen sieht die Welt schon wieder anders aus. Meine Firma hat heute enttäuscht. Bin aber nicht investiert, bekomme aber demnächst ein paar Optionen und kann mich dann mit dem Kursverlauf tief bei 9,80 € in 2003 und jetzt 60 € etwas trösten. Da soll TRIB auch mal hin Wäre schön, ist aber derzeit unrealistisch. We will see! Vielen Dank für deinen Fleiß hier im Forum. Bye
Wäre schön, ist aber derzeit unrealistisch. We will see! Vielen Dank für deinen Fleiß hier im Forum. Bye
das habe ich geahnt, dass unter 10 Cent geschmissen wird. Die Firma hat so gute Voraussetzungen und Zukunftsaussichten, aber so langsam verlässt mich die Zuversicht bei permanent schwachen Quartalszahlen. Ich verstehe einfach nicht, warum die Umsätze ständig steigen und der Gewinn stagniert. Ich bin wirklich enttäuscht vom Ergebnis. Wie lange sollen die Aktionäre denn noch warten, bis mal was positives rüber kommt. Die ewiger Warterei auf bessere Zeiten nervt ganz schön. Dann wird es bald mal Zeit für eine Ad-Hoc zur Übernahme, damit die Aktie nicht unter den flachen Aufwärtstrend fällt. Eigentlich war TRIB ein Long für mich, aber wenn nicht die nächsten Quartale die Gewinne steigen, schmeisse ich auch. Man kann leider bei anderen Werten besseres Geld verdienen. Bisher sind es ja nur Verluste. Naja morgen sieht die Welt schon wieder anders aus. Meine Firma hat heute enttäuscht. Bin aber nicht investiert, bekomme aber demnächst ein paar Optionen und kann mich dann mit dem Kursverlauf tief bei 9,80 € in 2003 und jetzt 60 € etwas trösten. Da soll TRIB auch mal hin
 Wäre schön, ist aber derzeit unrealistisch. We will see! Vielen Dank für deinen Fleiß hier im Forum. Bye
Wäre schön, ist aber derzeit unrealistisch. We will see! Vielen Dank für deinen Fleiß hier im Forum. Bye
Antwort auf Beitrag Nr.: 21.353.616 von Dangerous am 27.04.06 20:58:50Da muß ich Dir voll zustimmen.
Habe auch langsam die Schnauze voll.
Aber wenn ich morgen verkaufe kommt in den nächsten Tagen/Wochen die Aquisition.
Ein bischen Fantasie habe ich noch, OTC HIV , Primus Diabetic, Western Blot.
Die Coagulation die uns jetzt das Quartal verhagelt hat wird auch wieder anziehen. Vor allen nach der Aquisition. Da sind noch Synergiepotentiale vorhanden.
Aber alles in allen warten aufs nächste Quartal, wie immer.
Gerade minus 7% in USA.
Wie immer zu früh nachgekauft.
Macht keine Freude.
Ich denke ich klinke mich jetzt aus für die nächsten Tage und melde mich in 4 Wochen wieder.
Wenn die PR rauskommt können wir vielleicht mal ein bischen feiern.
Good trades
vaka
Habe auch langsam die Schnauze voll.
Aber wenn ich morgen verkaufe kommt in den nächsten Tagen/Wochen die Aquisition.
Ein bischen Fantasie habe ich noch, OTC HIV , Primus Diabetic, Western Blot.
Die Coagulation die uns jetzt das Quartal verhagelt hat wird auch wieder anziehen. Vor allen nach der Aquisition. Da sind noch Synergiepotentiale vorhanden.
Aber alles in allen warten aufs nächste Quartal, wie immer.
Gerade minus 7% in USA.
Wie immer zu früh nachgekauft.
Macht keine Freude.
Ich denke ich klinke mich jetzt aus für die nächsten Tage und melde mich in 4 Wochen wieder.
Wenn die PR rauskommt können wir vielleicht mal ein bischen feiern.
Good trades
vaka
Antwort auf Beitrag Nr.: 21.354.032 von Vakataka am 27.04.06 21:23:48Den Schlusskurs will ich noch nachreichen
370 000 Aktien
Minus 6,5%
8,42 $
Bis in ein paar Wochen
Waren heute ne Menge Klicks hier, können andere mal das Maul aufreissen
370 000 Aktien
Minus 6,5%
8,42 $
Bis in ein paar Wochen
Waren heute ne Menge Klicks hier, können andere mal das Maul aufreissen

Antwort auf Beitrag Nr.: 21.355.288 von Vakataka am 27.04.06 22:37:42@ Vakataka
Leider, leider, leider...
Was tun, sprach Zeus!
Meixnel
Leider, leider, leider...
Was tun, sprach Zeus!
Meixnel
Antwort auf Beitrag Nr.: 21.429.632 von Meixnel am 04.05.06 14:29:45Meixnel
Das muss jeder selbst entscheiden.
Verluste begrenzen ist manchmal auch nicht das schlechteste.
Buchwert laut Yahoo Finance 8,79$.
Lies nochmal die letzten 2 Seiten hier.
Alles was ich geschrieben habe glaube oder weiß ich.
Ich habe vor 2 Wochen bei 7,4€ nachgekauft.
Ich werde den Juni noch abwarten.
vaka
Das muss jeder selbst entscheiden.
Verluste begrenzen ist manchmal auch nicht das schlechteste.
Buchwert laut Yahoo Finance 8,79$.
Lies nochmal die letzten 2 Seiten hier.
Alles was ich geschrieben habe glaube oder weiß ich.
Ich habe vor 2 Wochen bei 7,4€ nachgekauft.
Ich werde den Juni noch abwarten.
vaka
Trinity Biotech (Add, Closing Price $8.53)
Q1'06 results disappoint but model is still holding.
Analyst: Ian Hunter
We have revised our Trinity Biotech model to incorporate Q1'06 numbers and management guidance, reducing forecast fully diluted earnings per share for 2006 by 3.2% to 41.3c from 42.7c. This comes from a 3.4% clip in forecast revenue to $110.4m from $114.3m. This pull back runs through FY07 and FY08 numbers with forecast earnings down 1.8% and 3.7% to 50.1c and 61.6c respectively. Running these changes through our DCF for Trinity Biotech (but including the $24m raised in the recent placement), values the company in the $8.82 to $12.23 per share range (up from our previous $8.10 to $12.11 range established on 23 March). On traditional multiples, the company is trading at 20.6x FY06 earnings, a 38.2% discount to peers. Given, (i) management's guidance on recovery in the second quarter; (ii) the potential for upside surprise on acquisitive activity (the recent placement should see the company with net cash of $20.4m at the end of 2006); and (iii) the unduly large discount at which the stock is trading relative to its peers, we believe the risk is weighted to the upside. We have, therefore, retained our Add recommendation but, reflecting the pull back in earnings forecasts, have reduced our price target to $9.75 from $10.10, which would have the shares trading at an relatively undemanding 23.6x FY06 earnings.
Q1'06 results disappoint but model is still holding.
Analyst: Ian Hunter
We have revised our Trinity Biotech model to incorporate Q1'06 numbers and management guidance, reducing forecast fully diluted earnings per share for 2006 by 3.2% to 41.3c from 42.7c. This comes from a 3.4% clip in forecast revenue to $110.4m from $114.3m. This pull back runs through FY07 and FY08 numbers with forecast earnings down 1.8% and 3.7% to 50.1c and 61.6c respectively. Running these changes through our DCF for Trinity Biotech (but including the $24m raised in the recent placement), values the company in the $8.82 to $12.23 per share range (up from our previous $8.10 to $12.11 range established on 23 March). On traditional multiples, the company is trading at 20.6x FY06 earnings, a 38.2% discount to peers. Given, (i) management's guidance on recovery in the second quarter; (ii) the potential for upside surprise on acquisitive activity (the recent placement should see the company with net cash of $20.4m at the end of 2006); and (iii) the unduly large discount at which the stock is trading relative to its peers, we believe the risk is weighted to the upside. We have, therefore, retained our Add recommendation but, reflecting the pull back in earnings forecasts, have reduced our price target to $9.75 from $10.10, which would have the shares trading at an relatively undemanding 23.6x FY06 earnings.
Buy confirmed.
Heute letzte Chance für 6,5€ ??
http://www.americanbulls.com/StockPage.asp?CompanyTicker=TRI…
Heute letzte Chance für 6,5€ ??
http://www.americanbulls.com/StockPage.asp?CompanyTicker=TRI…
Antwort auf Beitrag Nr.: 21.666.987 von Vakataka am 19.05.06 13:32:09May 22
Trinity Biotech signs agreement with 340B Prime
(English : DOC 36.50KB)
Trinity Biotech plc (Nasdaq NM: TRIB-News) (ISE: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, announced today that the 340B Prime Vendor Program, managed by HealthCare Purchasing Partners International™ (HPPI), has signed an agreement with Trinity Biotech to provide the Uni-Gold™ Recombigen® Rapid HIV antibody test to 340B Prime Vendor participants. There are currently in excess of 12,000 public hospitals, community health centers, clinics, state-operated AIDS programs and other safety-net healthcare providers serving low income and uninsured patients that are eligible for 340B. Previously, rapid HIV testing was only available to these grantees through a special bulk purchase program administered by the Centers for Disease Control (CDC).
http://www.trinitybiotech.com/
Trinity Biotech signs agreement with 340B Prime
(English : DOC 36.50KB)
Trinity Biotech plc (Nasdaq NM: TRIB-News) (ISE: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, announced today that the 340B Prime Vendor Program, managed by HealthCare Purchasing Partners International™ (HPPI), has signed an agreement with Trinity Biotech to provide the Uni-Gold™ Recombigen® Rapid HIV antibody test to 340B Prime Vendor participants. There are currently in excess of 12,000 public hospitals, community health centers, clinics, state-operated AIDS programs and other safety-net healthcare providers serving low income and uninsured patients that are eligible for 340B. Previously, rapid HIV testing was only available to these grantees through a special bulk purchase program administered by the Centers for Disease Control (CDC).
http://www.trinitybiotech.com/
Na, alle am Bier trinken?
May 25, 2006 - 1:00 AM EDT
Trinity Biotech Acquires Coagulation Product Line of bioMérieux
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has signed a binding agreement to acquire the Coagulation product line of bioMérieux for a total consideration of up to $51.9m. The bioMérieux Coagulation product line comprises a comprehensive portfolio of diagnostic tests manufactured primarily in Durham, North Carolina and a complete range of automated instruments primarily manufactured in St Louis.
Trinity will pay $40m on closing together with a maximum of $6.4m after 12 months and a maximum of a further $5.5m after 24 months. Of these combined deferred payments, an amount of $5.5m is contingent on the achievement of certain milestones for the product line during the remainder of 2006. The transaction will be funded from a combination of cash and bank debt and is expected to close in late June.
The worldwide Coagulation testing market is valued at $600m and is growing at 5% per annum. Every hospital needs to undertake Coagulation testing resulting in more than 25,000 clinical laboratories performing Coagulation testing globally. Following the merger of Trinity's existing Biopool and Amax Coagulation product lines with the bioMérieux product line it is estimated that Trinity's share of the worldwide Coagulation market will be 13%.
The bioMérieux product portfolio comprises a range of automated instruments including the MDA, MTX and Thrombolyzer which are comparable with various instruments within the Trinity Destiny instrument range. bioMérieux has a strong global position in the Coagulation market. In particular bioMérieux is strong in the U.S., U.K. and Germany where Trinity sells directly and this will result in considerable synergies in that Trinity will incur modest incremental sales and marketing costs.
As part of the transition, Trinity will transfer production of the diagnostic tests and instruments from bioMérieux's facilities in North Carolina to Dublin and from St Louis to Jamestown, New York respectively. During this transition process Trinity will combine the best of the bioMérieux, Biopool and Amax instrument and reagent product ranges with a view to creating a best-in-class instrument and reagent platform for the combined customer base. In this context, Trinity will be discontinuing various existing Coagulation products and this will result in a once-off write-off of inventory in the amount of $5.8 million.
Ronan O'Caoimh, CEO said: "This acquisition is our largest to date and will, we believe, be transformational for Trinity. We currently have a 5% Coagulation market share. This acquisition will significantly increase our installed base of instruments to 3,000 putting us in 4th place worldwide with a market share of 13%. The combination of the existing and the acquired business gives us a full range of state-of-the-art instruments for every size of hospital, with an excellent range of both routine and specialty diagnostic tests to accompany them. Given this combination and the scale achieved through the increased customer base, we are confident of increasing our market share aggressively in the future."
Ronan O'Caoimh further stated: "The transfer of reagent manufacturing from bioMérieux's plant in the U.S. to our existing reagent plant in Dublin will give rise to significant manufacturing efficiencies. Similarly, and more significantly, as the bioMérieux Coagulation revenues are concentrated in the U.S., Germany and the U.K. we will benefit from considerable and immediate synergies in these markets, given that we have existing salesforces in place. Despite the fact that there will be substantial costs incurred during the next 18 months on the technology transfer from the U.S. to Ireland, we expect the acquisition to be immediately earnings accretive and in 2007 it should add approximately $40m to our revenue and increase our operating profit by between $5m and $6m."
Trinity will provide additional information on an investor conference call on Tuesday 30th May at 11am EDT (4pm GMT). Details of the conference call will be available on www.trinitybiotech.com
About Trinity Biotech
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Forward-Looking Statements
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Source: Market Wire (May 25, 2006 - 1:00 AM EDT)
News by QuoteMedia
www.quotemedia.com
May 25, 2006 - 1:00 AM EDT
Trinity Biotech Acquires Coagulation Product Line of bioMérieux
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has signed a binding agreement to acquire the Coagulation product line of bioMérieux for a total consideration of up to $51.9m. The bioMérieux Coagulation product line comprises a comprehensive portfolio of diagnostic tests manufactured primarily in Durham, North Carolina and a complete range of automated instruments primarily manufactured in St Louis.
Trinity will pay $40m on closing together with a maximum of $6.4m after 12 months and a maximum of a further $5.5m after 24 months. Of these combined deferred payments, an amount of $5.5m is contingent on the achievement of certain milestones for the product line during the remainder of 2006. The transaction will be funded from a combination of cash and bank debt and is expected to close in late June.
The worldwide Coagulation testing market is valued at $600m and is growing at 5% per annum. Every hospital needs to undertake Coagulation testing resulting in more than 25,000 clinical laboratories performing Coagulation testing globally. Following the merger of Trinity's existing Biopool and Amax Coagulation product lines with the bioMérieux product line it is estimated that Trinity's share of the worldwide Coagulation market will be 13%.
The bioMérieux product portfolio comprises a range of automated instruments including the MDA, MTX and Thrombolyzer which are comparable with various instruments within the Trinity Destiny instrument range. bioMérieux has a strong global position in the Coagulation market. In particular bioMérieux is strong in the U.S., U.K. and Germany where Trinity sells directly and this will result in considerable synergies in that Trinity will incur modest incremental sales and marketing costs.
As part of the transition, Trinity will transfer production of the diagnostic tests and instruments from bioMérieux's facilities in North Carolina to Dublin and from St Louis to Jamestown, New York respectively. During this transition process Trinity will combine the best of the bioMérieux, Biopool and Amax instrument and reagent product ranges with a view to creating a best-in-class instrument and reagent platform for the combined customer base. In this context, Trinity will be discontinuing various existing Coagulation products and this will result in a once-off write-off of inventory in the amount of $5.8 million.
Ronan O'Caoimh, CEO said: "This acquisition is our largest to date and will, we believe, be transformational for Trinity. We currently have a 5% Coagulation market share. This acquisition will significantly increase our installed base of instruments to 3,000 putting us in 4th place worldwide with a market share of 13%. The combination of the existing and the acquired business gives us a full range of state-of-the-art instruments for every size of hospital, with an excellent range of both routine and specialty diagnostic tests to accompany them. Given this combination and the scale achieved through the increased customer base, we are confident of increasing our market share aggressively in the future."
Ronan O'Caoimh further stated: "The transfer of reagent manufacturing from bioMérieux's plant in the U.S. to our existing reagent plant in Dublin will give rise to significant manufacturing efficiencies. Similarly, and more significantly, as the bioMérieux Coagulation revenues are concentrated in the U.S., Germany and the U.K. we will benefit from considerable and immediate synergies in these markets, given that we have existing salesforces in place. Despite the fact that there will be substantial costs incurred during the next 18 months on the technology transfer from the U.S. to Ireland, we expect the acquisition to be immediately earnings accretive and in 2007 it should add approximately $40m to our revenue and increase our operating profit by between $5m and $6m."
Trinity will provide additional information on an investor conference call on Tuesday 30th May at 11am EDT (4pm GMT). Details of the conference call will be available on www.trinitybiotech.com
About Trinity Biotech
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits are used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Forward-Looking Statements
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Source: Market Wire (May 25, 2006 - 1:00 AM EDT)
News by QuoteMedia
www.quotemedia.com
Price Size Exch Time
7.32 712 NSD 13:11:27
7.32 625 NSD 13:11:11
7.25 215712 NSD 13:07:21
7.37 100 NSD 12:49:25
7.34 236 PAC 12:40:24
7.33 100 PAC 12:39:37
Da hatte wohl jemand die Schnauze voll!
Ich war es nicht
Aber irgendjemand hat den Block auch genommen, war
wohl abgesprochen.
7.32 712 NSD 13:11:27
7.32 625 NSD 13:11:11
7.25 215712 NSD 13:07:21
7.37 100 NSD 12:49:25
7.34 236 PAC 12:40:24
7.33 100 PAC 12:39:37
Da hatte wohl jemand die Schnauze voll!
Ich war es nicht

Aber irgendjemand hat den Block auch genommen, war
wohl abgesprochen.
July 19, 2006 - 12:01 PM EDT
Dutton Associates Announces Investment Opinion: Trinity Biotech Strong Buy Rating Maintained By Dutton Associates
Dutton Associates updates its coverage of Trinity Biotech plc (Nasdaq:TRIB) maintaining a Strong Speculative Buy rating and a 12 month $12 price target. The 8-page report by Dutton senior analyst Steve Handley is available at www.jmdutton.com as well as from First Call, Bloomberg, Zacks, Reuters, Knobias, and other leading financial portals.
Recently, on June 27, the Company announced that it had concluded its largest acquisition to date, which, in management's words, is expected to be "transformational" for Trinity. The acquired diagnostic product line is expected to immediately be accretive to earnings, and in 2007 is projected to add $40 million to revenues and $5-$6 million to operating profits. Specifically, this relates to the acquisition of bioMerieux's coagulation business, which has been acquired for a total cash consideration of up to $51.9 million. Its coagulation instrument and reagent product lines have immediately increased Trinity's installed base of automated coagulation instruments from 1,000 to 3,000, and thereby moved the Company into fourth place in this market; specifically, this will increase its share of the worldwide coagulation market from 5% to about 13%. Management's stated objective is to compound revenues over the next few years at an average annual rate of 20%, through a combination of organic growth and acquisitions, and to increase operating margins from 7.9% in the recent quarter (before share-based payments) to over 12.0%. This targeted operating margin is expected to be achieved by early 2007.
About Dutton Associates
Dutton Associates is one of the largest independent investment research firms in the U.S. Its 31 senior analysts are primarily CFAs, and have expertise in many industries. Dutton Associates provides continuing analyst coverage of over 140 enrolled companies, and its research, estimates, and ratings are carried in all the major databases serving institutions and online investors.
The cost of enrollment in our one-year continuing research program is US $35,000 prepaid for 4 Research Reports, typically published quarterly, and requisite Research Notes. Dutton Associates received $71,000 from the Company for 12 Research Reports with coverage commencing on 1/27/2003. We do not accept payment of our fees in company stock. Our principals and analysts are prohibited from owning or trading in securities of covered companies. The views expressed in this research report accurately reflect the analyst's personal views about the subject securities or issuer. Neither the analyst's compensation nor the compensation received by us is in any way related to the specific ratings or views contained in this research report or note. Please read full disclosures and analyst background at www.jmdutton.com before investing.
Dutton Associates
John M. Dutton, 916-941-8119
Source: Business Wire (July 19, 2006 - 12:01 PM EDT)
News by QuoteMedia
www.quotemedia.com
Dutton Associates Announces Investment Opinion: Trinity Biotech Strong Buy Rating Maintained By Dutton Associates
Dutton Associates updates its coverage of Trinity Biotech plc (Nasdaq:TRIB) maintaining a Strong Speculative Buy rating and a 12 month $12 price target. The 8-page report by Dutton senior analyst Steve Handley is available at www.jmdutton.com as well as from First Call, Bloomberg, Zacks, Reuters, Knobias, and other leading financial portals.
Recently, on June 27, the Company announced that it had concluded its largest acquisition to date, which, in management's words, is expected to be "transformational" for Trinity. The acquired diagnostic product line is expected to immediately be accretive to earnings, and in 2007 is projected to add $40 million to revenues and $5-$6 million to operating profits. Specifically, this relates to the acquisition of bioMerieux's coagulation business, which has been acquired for a total cash consideration of up to $51.9 million. Its coagulation instrument and reagent product lines have immediately increased Trinity's installed base of automated coagulation instruments from 1,000 to 3,000, and thereby moved the Company into fourth place in this market; specifically, this will increase its share of the worldwide coagulation market from 5% to about 13%. Management's stated objective is to compound revenues over the next few years at an average annual rate of 20%, through a combination of organic growth and acquisitions, and to increase operating margins from 7.9% in the recent quarter (before share-based payments) to over 12.0%. This targeted operating margin is expected to be achieved by early 2007.
About Dutton Associates
Dutton Associates is one of the largest independent investment research firms in the U.S. Its 31 senior analysts are primarily CFAs, and have expertise in many industries. Dutton Associates provides continuing analyst coverage of over 140 enrolled companies, and its research, estimates, and ratings are carried in all the major databases serving institutions and online investors.
The cost of enrollment in our one-year continuing research program is US $35,000 prepaid for 4 Research Reports, typically published quarterly, and requisite Research Notes. Dutton Associates received $71,000 from the Company for 12 Research Reports with coverage commencing on 1/27/2003. We do not accept payment of our fees in company stock. Our principals and analysts are prohibited from owning or trading in securities of covered companies. The views expressed in this research report accurately reflect the analyst's personal views about the subject securities or issuer. Neither the analyst's compensation nor the compensation received by us is in any way related to the specific ratings or views contained in this research report or note. Please read full disclosures and analyst background at www.jmdutton.com before investing.
Dutton Associates
John M. Dutton, 916-941-8119
Source: Business Wire (July 19, 2006 - 12:01 PM EDT)
News by QuoteMedia
www.quotemedia.com
10 August 2006
Q2 2006 Trinity Biotech Earnings Release
26 Oct 2006*
Q3 2006 Trinity Biotech Earnings Release
http://www.trinitybiotech.com/
Q2 2006 Trinity Biotech Earnings Release
26 Oct 2006*
Q3 2006 Trinity Biotech Earnings Release
http://www.trinitybiotech.com/
Trinity Biotech and Inverness Settle Dispute and Enter Manufacturing and Licence Agreements
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, is pleased to announce that it has reached an amicable settlement in its dispute with Inverness Medical Innovations, Inc.
Under the terms of the settlement Inverness has granted Trinity a royalty bearing licence to its lateral flow patents for all diagnostic uses with the exception of women's health and cardiology, including an Over the Counter ("OTC") license for Trinity's Unigold HIV products. In addition, Inverness has agreed to manufacture Trinity's Unigold HIV tests primarily for the African market in its new facility in Hangzhou, China, and will reimburse Trinity for its costs of litigation.
Ronan O'Caoimh commented, "We are pleased to have settled this litigation and to have obtained a licence which covers the potentially lucrative HIV OTC market in the USA. We believe that the combination of our platform Unigold technology, as proven with our HIV products, and the broad licence from Inverness will enable us to develop a substantial and vibrant point-of-care business."
"We are pleased to have reached a settlement in this dispute, and are excited about the prospect of making Trinity's Unigold HIV product in our new China plant," said Ron Zwanziger, CEO of Inverness.
Further details will be provided on Trinity Biotech's 2nd quarter conference call on Wednesday 9 August at 4.00pm BST (11.00am ET). The call is being webcast and can be accessed at www.vcall.com
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB.I), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, is pleased to announce that it has reached an amicable settlement in its dispute with Inverness Medical Innovations, Inc.
Under the terms of the settlement Inverness has granted Trinity a royalty bearing licence to its lateral flow patents for all diagnostic uses with the exception of women's health and cardiology, including an Over the Counter ("OTC") license for Trinity's Unigold HIV products. In addition, Inverness has agreed to manufacture Trinity's Unigold HIV tests primarily for the African market in its new facility in Hangzhou, China, and will reimburse Trinity for its costs of litigation.
Ronan O'Caoimh commented, "We are pleased to have settled this litigation and to have obtained a licence which covers the potentially lucrative HIV OTC market in the USA. We believe that the combination of our platform Unigold technology, as proven with our HIV products, and the broad licence from Inverness will enable us to develop a substantial and vibrant point-of-care business."
"We are pleased to have reached a settlement in this dispute, and are excited about the prospect of making Trinity's Unigold HIV product in our new China plant," said Ron Zwanziger, CEO of Inverness.
Further details will be provided on Trinity Biotech's 2nd quarter conference call on Wednesday 9 August at 4.00pm BST (11.00am ET). The call is being webcast and can be accessed at www.vcall.com
Trinity Biotech Announces Quarter 2 Results Revenues Increase 20.3% and Profits Increase 95%
DUBLIN, IRELAND -- (MARKET WIRE) -- August 09, 2006 -- Trinity Biotech plc (NASDAQ: TRIB) (ISE: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2006. Revenues in quarter 2, 2006 increased 20.3% to US$27.3 million compared to US$22.7 million in quarter 2 2005. During the same period operating profit before share option expenses increased from US$1.69 million to US$2.34 million before the impact of the once off inventory write off of US$5.8 million. Profit for the period increased from US$1.08 million in quarter 2 2005 to US$2.11 million before the inventory write off in quarter 2 2006, representing an increase of 95%.
Revenues for the six months by key product area were as follows :
H1 2005 H1 2006
------------ ------------ -----------
US$000 US$000 % Increase
------------ ------------ -----------
Clinical Chemistry* 3,043 7,502 146.6%
------------ ------------ -----------
Haemostasis 14,399 14,782 2.7%
------------ ------------ -----------
Infectious Diseases 20,179 21,027 4.2%
------------ ------------ -----------
Point of Care 6,147 8,716 41.8%
------------ ------------ -----------
Total 43,768 52,027 18.8%
------------ ------------ -----------
* Primus acquired in July 2005
http://www.marketwire.com/mw/release_html_b1?release_id=1522…
DUBLIN, IRELAND -- (MARKET WIRE) -- August 09, 2006 -- Trinity Biotech plc (NASDAQ: TRIB) (ISE: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2006. Revenues in quarter 2, 2006 increased 20.3% to US$27.3 million compared to US$22.7 million in quarter 2 2005. During the same period operating profit before share option expenses increased from US$1.69 million to US$2.34 million before the impact of the once off inventory write off of US$5.8 million. Profit for the period increased from US$1.08 million in quarter 2 2005 to US$2.11 million before the inventory write off in quarter 2 2006, representing an increase of 95%.
Revenues for the six months by key product area were as follows :
H1 2005 H1 2006
------------ ------------ -----------
US$000 US$000 % Increase
------------ ------------ -----------
Clinical Chemistry* 3,043 7,502 146.6%
------------ ------------ -----------
Haemostasis 14,399 14,782 2.7%
------------ ------------ -----------
Infectious Diseases 20,179 21,027 4.2%
------------ ------------ -----------
Point of Care 6,147 8,716 41.8%
------------ ------------ -----------
Total 43,768 52,027 18.8%
------------ ------------ -----------
* Primus acquired in July 2005
http://www.marketwire.com/mw/release_html_b1?release_id=1522…
Antwort auf Beitrag Nr.: 23.372.291 von Vakataka am 09.08.06 18:02:55so schlecht klingt das ja eigentlich nicht oder, wo lagen die Erwartungen für den Berichtszeitraum?
Antwort auf Beitrag Nr.: 23.385.541 von girokonto am 10.08.06 14:53:58Nun, ich bin erstmal beruhigt, die scheinen jetzt endlich auf den richtigen Weg zu sein.
Wenn trib in Q3 13-14c und Q4 15-16c erreicht bin ich sogar zufrieden.
Auf dem Link unten siehst Du die Schätzung und andere Zahlen, einfach mal durchklicken.
http://www.nasdaq.com/earnings/earnings_forecast.asp?symbol=…
Wenn trib in Q3 13-14c und Q4 15-16c erreicht bin ich sogar zufrieden.
Auf dem Link unten siehst Du die Schätzung und andere Zahlen, einfach mal durchklicken.
http://www.nasdaq.com/earnings/earnings_forecast.asp?symbol=…
Antwort auf Beitrag Nr.: 23.390.051 von Vakataka am 10.08.06 19:41:53danke,
ehrlich gesagt schlummern die trinity's bei mir nur so neben bei und ich habe ihnen nicht sehr viel Aufmerksamkeit in der letzten Zeit geschenkt.
Zwischenzeitlich hatte ich mal gut verdient, aber nachdem ich wieder eingestiegen bin geht es bisschen seitwärts, eben weil die Ergebnisse noch nicht überzeugen konnten.
Wenn jetzt eine solide Gewinnentwicklung für die nächsten Quartale kommt wird das der Kurs sicher auch bald würdigen.
ehrlich gesagt schlummern die trinity's bei mir nur so neben bei und ich habe ihnen nicht sehr viel Aufmerksamkeit in der letzten Zeit geschenkt.
Zwischenzeitlich hatte ich mal gut verdient, aber nachdem ich wieder eingestiegen bin geht es bisschen seitwärts, eben weil die Ergebnisse noch nicht überzeugen konnten.
Wenn jetzt eine solide Gewinnentwicklung für die nächsten Quartale kommt wird das der Kurs sicher auch bald würdigen.
langsam entwickelt sich der Kurs ja wieder, aber in Deutschland kann man die Umsätze echt vergessen, der Kurs hängt voll an Amerika.
Langsam geht es hoch.
Es würde mich nicht wundern wenn wir in Q3 oder Q4 etwas von Primus hören.
Früher hätte ich gesagt wir sehen einen richtigen Hype.
Heute bin ich etwas schlauer, wahrscheinlich passiert gar nichts.
Falls hier einer bei WO auf die Umsätze in den USA achtet.
Die Umsätze die bei WO angezeigt werden stimmen nicht.
Ein Beispiel heute Umsatz ca 28000 Aktien
Orderbuch kann man einsehen bei otcbb.com
oder onvista.de
Time & Sales
Price Size Exch Time
8.30 100 NGS 15:58:58
8.30 100 NGS 15:58:55
8.25 100 NGS 15:58:50
8.30 300 NGS 15:52:01
8.30 225 NDD 15:52:01
8.30 200 NGS 15:41:09
8.30 173 NGS 15:41:09
8.30 427 NGS 15:41:09
8.30 100 PAC 15:41:09
8.30 100 PAC 15:41:09
8.32 100 PAC 15:36:34
8.25 400 NDD 15:31:17
8.27 100 NGS 15:31:09
8.25 10000 NDD 14:54:44
8.25 100 NGS 14:53:27
8.31 100 PAC 14:43:31
8.21 250 PAC 14:11:05
8.20 485 NDD 14:10:11
8.20 485 NGS 14:10:11
8.20 100 NDD 14:10:11
8.20 100 NGS 14:10:11
8.18 250 NGS 13:43:23
8.18 250 NGS 13:43:23
8.17 100 NGS 13:43:21
8.15 100 NGS 13:43:20
8.13 600 PAC 13:43:05
8.13 100 NGS 13:38:59
8.15 100 PAC 13:32:05
8.15 100 PAC 13:09:06
8.11 100 PAC 13:09:06
WO zeigt nur einen Umsatz von ca 8700 Aktien
Times & Sales (25.08.2006 von 15:30:06 bis 22:01:33) Weiter Zeit Umsatz Volumen Geld (Bid) Geldvolumen Brief (Ask) Briefvolumen
22:01:33 8,30 0 - - - -
21:58:58 8,30 100 - - - -
21:58:55 8,30 100 - - - -
21:58:50 8,25 100 - - - -
21:52:01 8,30 300 - - - -
21:41:09 8,30 200 - - - -
21:41:09 8,30 173 - - - -
21:41:09 8,30 427 - - - -
21:31:09 8,27 100 - - - -
20:53:27 8,25 100 - - - -
20:10:11 8,20 485 - - - -
20:10:11 8,20 100 - - - -
19:43:23 8,18 250 - - - -
19:43:23 8,18 250 - - - -
19:43:21 8,17 100 - - - -
19:43:20 8,15 100 - - - -
19:38:58 8,13 100 - - - -
19:08:30 8,11 100 - - - -
18:49:39 8,12 100 - - - -
17:50:27 8,20 100 - - - -
17:00:32 8,10 1.300 - - - -
17:00:32 8,10 1.100 - - - -
17:00:32 8,10 100 - - - -
16:54:18 8,10 161 - - - -
15:51:18 8,10 839 - - - -
15:51:06 8,10 100 - - - -
15:51:06 8,10 400 - - - -
15:51:06 8,10 500 - - - -
15:51:06 8,10 100 - - - -
15:30:07 8,10 787 - - - -
15:30:06 8,10 787
Es würde mich nicht wundern wenn wir in Q3 oder Q4 etwas von Primus hören.
Früher hätte ich gesagt wir sehen einen richtigen Hype.
Heute bin ich etwas schlauer, wahrscheinlich passiert gar nichts.
Falls hier einer bei WO auf die Umsätze in den USA achtet.
Die Umsätze die bei WO angezeigt werden stimmen nicht.
Ein Beispiel heute Umsatz ca 28000 Aktien
Orderbuch kann man einsehen bei otcbb.com
oder onvista.de
Time & Sales
Price Size Exch Time
8.30 100 NGS 15:58:58
8.30 100 NGS 15:58:55
8.25 100 NGS 15:58:50
8.30 300 NGS 15:52:01
8.30 225 NDD 15:52:01
8.30 200 NGS 15:41:09
8.30 173 NGS 15:41:09
8.30 427 NGS 15:41:09
8.30 100 PAC 15:41:09
8.30 100 PAC 15:41:09
8.32 100 PAC 15:36:34
8.25 400 NDD 15:31:17
8.27 100 NGS 15:31:09
8.25 10000 NDD 14:54:44
8.25 100 NGS 14:53:27
8.31 100 PAC 14:43:31
8.21 250 PAC 14:11:05
8.20 485 NDD 14:10:11
8.20 485 NGS 14:10:11
8.20 100 NDD 14:10:11
8.20 100 NGS 14:10:11
8.18 250 NGS 13:43:23
8.18 250 NGS 13:43:23
8.17 100 NGS 13:43:21
8.15 100 NGS 13:43:20
8.13 600 PAC 13:43:05
8.13 100 NGS 13:38:59
8.15 100 PAC 13:32:05
8.15 100 PAC 13:09:06
8.11 100 PAC 13:09:06
WO zeigt nur einen Umsatz von ca 8700 Aktien
Times & Sales (25.08.2006 von 15:30:06 bis 22:01:33) Weiter Zeit Umsatz Volumen Geld (Bid) Geldvolumen Brief (Ask) Briefvolumen
22:01:33 8,30 0 - - - -
21:58:58 8,30 100 - - - -
21:58:55 8,30 100 - - - -
21:58:50 8,25 100 - - - -
21:52:01 8,30 300 - - - -
21:41:09 8,30 200 - - - -
21:41:09 8,30 173 - - - -
21:41:09 8,30 427 - - - -
21:31:09 8,27 100 - - - -
20:53:27 8,25 100 - - - -
20:10:11 8,20 485 - - - -
20:10:11 8,20 100 - - - -
19:43:23 8,18 250 - - - -
19:43:23 8,18 250 - - - -
19:43:21 8,17 100 - - - -
19:43:20 8,15 100 - - - -
19:38:58 8,13 100 - - - -
19:08:30 8,11 100 - - - -
18:49:39 8,12 100 - - - -
17:50:27 8,20 100 - - - -
17:00:32 8,10 1.300 - - - -
17:00:32 8,10 1.100 - - - -
17:00:32 8,10 100 - - - -
16:54:18 8,10 161 - - - -
15:51:18 8,10 839 - - - -
15:51:06 8,10 100 - - - -
15:51:06 8,10 400 - - - -
15:51:06 8,10 500 - - - -
15:51:06 8,10 100 - - - -
15:30:07 8,10 787 - - - -
15:30:06 8,10 787
Antwort auf Beitrag Nr.: 23.618.052 von Vakataka am 25.08.06 23:08:22Damit Ihr nicht glaubt das ich hier Mist erzähle nochmal Times und Sale von onvista.
Wer von Euch hat um 20.54 gekauft
Onvista Stand: 25. 08. 2006
TRINITY BIOTECH PLC REG.SHS A (SP.ADRS NEW) 4/O.N.
WKN: A0ET3S ISIN: US8964383066 Branche: Biotechnologie Land: Irland
Times & Sales
Uhrzeit Kurs letztes Volumen kumuliert
21:58:59 8,30 100 28.851
21:58:56 8,30 100 28.751
21:58:51 8,25 100 28.651
21:52:02 8,30 300 28.551
21:52:02 8,2975 225 28.251
21:41:10 8,30 200 28.026
21:41:10 8,30 173 27.826
21:41:09 8,30 427 27.653
21:41:09 8,30 100 27.226
21:41:09 8,30 100 27.126
21:36:34 8,32 100 27.026
21:31:17 8,25 400 26.926
21:31:09 8,27 100 26.526
20:54:44 8,25 10.000 26.426
20:53:28 8,25 100 16.426
20:43:31 8,31 100 16.326
20:11:05 8,21 250 16.226
20:10:11 8,20 485 15.976
20:10:11 8,20 485 15.491
20:10:11 8,20 100 15.006
20:10:11 8,20 100 14.906
19:43:24 8,18 250 14.806
19:43:24 8,18 250 14.556
19:43:21 8,17 100 14.306
19:43:20 8,15 100 14.206
19:43:05 8,13 600 14.106
19:38:59 8,13 100 13.506
19:32:05 8,15 100 13.406
19:09:06 8,15 100 13.306
19:09:06 8,11 100 13.206
19:08:30 8,11 100 13.106
18:56:58 8,13 600 13.006
18:49:40 8,12 100 12.406
18:17:58 8,1272 100 12.306
18:17:58 8,12 150 12.206
18:11:50 8,126 1.000 12.056
18:11:11 8,1224 500 11.056
17:50:27 8,20 100 10.556
17:19:33 8,10 500 10.456
17:00:32 8,10 1.100 9.956
17:00:32 8,10 1.300 8.856
17:00:32 8,10 1.300 7.556
17:00:32 8,10 1.100 6.256
17:00:32 8,10 100 5.156
16:56:38 8,122 190 5.056
16:54:19 8,10 161 4.866
16:53:07 8,095 300 4.705
16:10:02 8,07 679 4.405
15:51:18 8,10 839 3.726
15:51:07 8,10 100 2.887
15:51:07 8,10 400 2.787
15:51:07 8,10 500 2.387
15:51:07 8,10 100 1.887
15:51:01 8,10 1.000 1.787
15:30:07 8,10 787 787
Wer von Euch hat um 20.54 gekauft

Onvista Stand: 25. 08. 2006
TRINITY BIOTECH PLC REG.SHS A (SP.ADRS NEW) 4/O.N.
WKN: A0ET3S ISIN: US8964383066 Branche: Biotechnologie Land: Irland
Times & Sales
Uhrzeit Kurs letztes Volumen kumuliert
21:58:59 8,30 100 28.851
21:58:56 8,30 100 28.751
21:58:51 8,25 100 28.651
21:52:02 8,30 300 28.551
21:52:02 8,2975 225 28.251
21:41:10 8,30 200 28.026
21:41:10 8,30 173 27.826
21:41:09 8,30 427 27.653
21:41:09 8,30 100 27.226
21:41:09 8,30 100 27.126
21:36:34 8,32 100 27.026
21:31:17 8,25 400 26.926
21:31:09 8,27 100 26.526
20:54:44 8,25 10.000 26.426
20:53:28 8,25 100 16.426
20:43:31 8,31 100 16.326
20:11:05 8,21 250 16.226
20:10:11 8,20 485 15.976
20:10:11 8,20 485 15.491
20:10:11 8,20 100 15.006
20:10:11 8,20 100 14.906
19:43:24 8,18 250 14.806
19:43:24 8,18 250 14.556
19:43:21 8,17 100 14.306
19:43:20 8,15 100 14.206
19:43:05 8,13 600 14.106
19:38:59 8,13 100 13.506
19:32:05 8,15 100 13.406
19:09:06 8,15 100 13.306
19:09:06 8,11 100 13.206
19:08:30 8,11 100 13.106
18:56:58 8,13 600 13.006
18:49:40 8,12 100 12.406
18:17:58 8,1272 100 12.306
18:17:58 8,12 150 12.206
18:11:50 8,126 1.000 12.056
18:11:11 8,1224 500 11.056
17:50:27 8,20 100 10.556
17:19:33 8,10 500 10.456
17:00:32 8,10 1.100 9.956
17:00:32 8,10 1.300 8.856
17:00:32 8,10 1.300 7.556
17:00:32 8,10 1.100 6.256
17:00:32 8,10 100 5.156
16:56:38 8,122 190 5.056
16:54:19 8,10 161 4.866
16:53:07 8,095 300 4.705
16:10:02 8,07 679 4.405
15:51:18 8,10 839 3.726
15:51:07 8,10 100 2.887
15:51:07 8,10 400 2.787
15:51:07 8,10 500 2.387
15:51:07 8,10 100 1.887
15:51:01 8,10 1.000 1.787
15:30:07 8,10 787 787
Auch wenn es keinen interessiert
Geiler Tag!
512k ( die WO Zahlen passen wieder nicht )
plus 13.4% ( in der Spitze bis 20% plus )
Geiler Tag!
512k ( die WO Zahlen passen wieder nicht )
plus 13.4% ( in der Spitze bis 20% plus )
Antwort auf Beitrag Nr.: 24.093.017 von Vakataka am 21.09.06 22:29:04mich interessiert es freilich was war der grund hatte noch keine Zeit nachzusehn, bin grad erst heim gekomm!
was war der grund hatte noch keine Zeit nachzusehn, bin grad erst heim gekomm!
 was war der grund hatte noch keine Zeit nachzusehn, bin grad erst heim gekomm!
was war der grund hatte noch keine Zeit nachzusehn, bin grad erst heim gekomm!
Antwort auf Beitrag Nr.: 24.093.337 von girokonto am 21.09.06 22:57:16Ich habe auch stundenlang geraten, dann habe ich im Yahoo Board was gefunden.
TRIB on CNBC again (1 Rating) 21-Sep-06 04:56 pm
CNBC just mentioned TRIB again in its story regarding the new HIV guidelines. OSUR was the only other mentioned and the story said more guidelines were coming. Let's hope the WS Journal picks up on this tomorrow and mentions TRIB. The CNBC story should run again tonight.
TRIB on CNBC again (1 Rating) 21-Sep-06 04:56 pm
CNBC just mentioned TRIB again in its story regarding the new HIV guidelines. OSUR was the only other mentioned and the story said more guidelines were coming. Let's hope the WS Journal picks up on this tomorrow and mentions TRIB. The CNBC story should run again tonight.
250 Million in Revenue? (Not rated) 23-Sep-06 07:57 pm Given the CDC's recommendation to routinely test ALL American's between the ages of 13 & 64, I'd say it's a safe bet that TRIB will hit $250 Million in annual revenue without another acqusition! 200+ million people, 1 test each per year? There's going to be an HIV test sales boom for everybody! If TRIB gets 10% additional market share (20 million tests), $5 gross per test adds $100 million to revenues. I'm willing to bet gross is closer to $20 per test. That equates to $400 MILLION in ADDITIONAL Revenue! Somebody posted earlier that margins on Uni-Gold HIV tests are in the 80% range vs 50% for other products. Get ready to rock! We're all about to make some money!!!!



tia und kommt noch besser!!!!!!!!!!!



tia und kommt noch besser!!!!!!!!!!!

Antwort auf Beitrag Nr.: 24.152.225 von serafino am 24.09.06 09:08:54Sorry, der Typ hat keine Ahnung.
Die Zahlen sind nicht nur übertrieben, das sind Mondzahlen
Die Zahlen sind nicht nur übertrieben, das sind Mondzahlen

Antwort auf Beitrag Nr.: 24.180.653 von Vakataka am 25.09.06 09:34:08Hallo Vaka,
was ist denn deine guidiance für das aktuelle Quartal. Jetzt sollte doch endlich der Zukauf sich voll auf das Q-Ergebnis auswirken. Ich hoffe, dass wir in den nächsten Tagen den Widerstand von $ 9,50 brechen und der Weg bis 12 $ frei ist. Mit endlich wieder steigenden Gewinnen wäre die Aktie unterbewertet. Ich hoffe, dass wir nicht wieder durch stagnierende Gewinne bei steigenden Umsätze enttäuscht werden. Aber die Kursentwicklung ist sicher ein Vorbote auf sehr gute Zahlen?
Ciao Danger
was ist denn deine guidiance für das aktuelle Quartal. Jetzt sollte doch endlich der Zukauf sich voll auf das Q-Ergebnis auswirken. Ich hoffe, dass wir in den nächsten Tagen den Widerstand von $ 9,50 brechen und der Weg bis 12 $ frei ist. Mit endlich wieder steigenden Gewinnen wäre die Aktie unterbewertet. Ich hoffe, dass wir nicht wieder durch stagnierende Gewinne bei steigenden Umsätze enttäuscht werden. Aber die Kursentwicklung ist sicher ein Vorbote auf sehr gute Zahlen?
Ciao Danger
Antwort auf Beitrag Nr.: 24.417.671 von Dangerous am 05.10.06 12:41:16Hallo Dangerous,
ich teile Deine Hoffnungen.
Ich denke die Kursentwicklung und das anziehende Volumen hängt mit der Sendung auf CNBC zusammen. Das scheint jetzt aber abzuflauen. Bisher war es so das der Kurs vor den Q-Zahlen anzog, da sollte bis zum 26. noch etwas drin sein.
Zu den Zahlen mag ich nicht spekulieren. Aber alles andere als ein Zuwachs ist enttäuschend.
Wenn der BMX Zukauf voll durchschlägt sollte der Umsatz um bis zu 10 Mio zulegen. Bei dem Gewinn bin ich mir nicht sicher. Wenn ich das richtig gelesen habe wird jetzt in Irland eine neue Produktionshalle gebaut. Wenn dort produziert wird sollte der Gewinn nochmal anziehen.
Lassen wir uns mal überraschen.
Gruss vaka
PS: Schau Dir mal die Bewertung von orasure an, die werden oft mit trinity verglichen
ich teile Deine Hoffnungen.
Ich denke die Kursentwicklung und das anziehende Volumen hängt mit der Sendung auf CNBC zusammen. Das scheint jetzt aber abzuflauen. Bisher war es so das der Kurs vor den Q-Zahlen anzog, da sollte bis zum 26. noch etwas drin sein.
Zu den Zahlen mag ich nicht spekulieren. Aber alles andere als ein Zuwachs ist enttäuschend.
Wenn der BMX Zukauf voll durchschlägt sollte der Umsatz um bis zu 10 Mio zulegen. Bei dem Gewinn bin ich mir nicht sicher. Wenn ich das richtig gelesen habe wird jetzt in Irland eine neue Produktionshalle gebaut. Wenn dort produziert wird sollte der Gewinn nochmal anziehen.
Lassen wir uns mal überraschen.
Gruss vaka
PS: Schau Dir mal die Bewertung von orasure an, die werden oft mit trinity verglichen
Schaffen wir den Sprung nach oben?
cherrs

cherrs
Antwort auf Beitrag Nr.: 24.626.710 von serafino am 14.10.06 22:15:23fallende Kurse vor den Q-Zahlen lassen nichts Gutes erahnen. Ich hoffe, der Markt liegt diesmal falsch und es wird endlich mal eine positive Gewinnüberraschung geben.
We will see. Morgen sind wir schlauer
We will see. Morgen sind wir schlauer
Antwort auf Beitrag Nr.: 24.877.811 von serafino am 26.10.06 22:06:56Hätte ja mal jemand den link reinstellen können
http://www.nasdaq.com/aspxcontent/NewsStory.aspx?cpath=20061…
http://www.nasdaq.com/aspxcontent/NewsStory.aspx?cpath=20061…
Antwort auf Beitrag Nr.: 24.878.510 von Vakataka am 26.10.06 22:34:26das schaut doch garnicht so schlecht aus, also nach den jüngsten Aquisitionen sind sie gut aufgestellt und wir können ein kontinuierliches Wachstum erwarten, das sollte dem Kurs auch langfristig gut aussehen lassen.
Mal schaun
Mal schaun

Antwort auf Beitrag Nr.: 24.859.424 von Dangerous am 26.10.06 09:09:41Hallo Dangerous
Nach trägen Handel sind die Amis wohl doch zufrieden mit dem Ergebnis.
Plus 11% und 148k sind nicht schlecht, hoffen wir mal das es nachhaltig ist.
Ich finde den Umsatz sehr schwach, da habe ich mehr erwartet. Ich kann mir das nicht erklären. Plus 6 Mio? von BMX , wenn das nächstes Quartal so bleibt war das ein glatter Fehlkauf. Naja, abwarten.
EPS ist im Rahmen meiner Erwartung ( siehe @ 621 ).
Völlig daneben ist die Meldung über Nephrotek und David Oxley, da hätte man in den nächsten Tagen 2 Pressemitteilungen rausgeben können.
Von PR haben die noch nie was verstanden
Was meinst Du?
Gruss vaka
Nach trägen Handel sind die Amis wohl doch zufrieden mit dem Ergebnis.
Plus 11% und 148k sind nicht schlecht, hoffen wir mal das es nachhaltig ist.
Ich finde den Umsatz sehr schwach, da habe ich mehr erwartet. Ich kann mir das nicht erklären. Plus 6 Mio? von BMX , wenn das nächstes Quartal so bleibt war das ein glatter Fehlkauf. Naja, abwarten.
EPS ist im Rahmen meiner Erwartung ( siehe @ 621 ).
Völlig daneben ist die Meldung über Nephrotek und David Oxley, da hätte man in den nächsten Tagen 2 Pressemitteilungen rausgeben können.
Von PR haben die noch nie was verstanden

Was meinst Du?
Gruss vaka
Antwort auf Beitrag Nr.: 24.878.510 von Vakataka am 26.10.06 22:34:26Hi Vaka! Na endlich. Darauf haben wir so lange gewartet. Die Analystenerwartungen von 12,5 Cent wurden übertroffen. Prima finde ich, dass mal die Kosten halbwegs konstant geblieben sind und damit die Umsatzsteigerung fast vollständig auf das Ergebnis durchgeschlagen hat. Ich unterstelle mal einen zukünftigen Gewinn pro Quartal von 15 Cent ab 2007. Dann ergäbe sich ein Jahresergebnis von 60 Cent. Bei einem guten KGV von 30 könnten das Potential durchaus bis in den Bereich von 18 Dollar reichen. Allerdings wird das sicher nicht vor Mitte nächsten Jahres sein.
Antwort auf Beitrag Nr.: 24.878.875 von Vakataka am 26.10.06 22:55:51Hallo Vaka, da ich nicht wusste wieviel Umsatz und Gewinn von Biomerix dazugekauft wurde, kann ich nichts zu meinen Erwartungen sagen. Ich habe nochmal nachgelesen. Die Frage ist, ab wann wurden die Ergebnisse von Biomerix mit einbezogen. Da die Vereinbarung Ende Juni geschlossen wurde, könnte man annehmen, dass bereits das volle Quartal eingerechnet wurde. Dafür ist die Umsatzsteigerung wirklich enttäuschend. Man müsste jetzt wissen, wie hoch der Umsatzanteil von Biomerix war, um das ausreichend beurteilen zu können. Aber ich gebe dir Recht, wenn ich folgende Meldung noch einmal lese, dann muss man über die Umsatzsteigerung enttäuscht sein.
DUBLIN, IRELAND--(MARKET WIRE)--Jun 27, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has completed the acquisition of the Coagulation product line of bioMérieux for a total consideration of up to $51.9m. The bioMérieux Coagulation product line comprises a comprehensive portfolio of diagnostic tests manufactured primarily in Durham, North Carolina, and a complete range of automated instruments primarily manufactured in St Louis.
ADVERTISEMENT
Trinity has paid $40m on closing and will pay a maximum of $6.4m after 12 months and a maximum of a further $5.5m after 24 months. Of these combined deferred payments, an amount of $5.5m is contingent on the achievement of certain milestones for the product line during the remainder of 2006. The transaction is being funded from a combination of cash and bank debt.
DUBLIN, IRELAND--(MARKET WIRE)--Jun 27, 2006 -- Trinity Biotech plc (NasdaqNM:TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has completed the acquisition of the Coagulation product line of bioMérieux for a total consideration of up to $51.9m. The bioMérieux Coagulation product line comprises a comprehensive portfolio of diagnostic tests manufactured primarily in Durham, North Carolina, and a complete range of automated instruments primarily manufactured in St Louis.
ADVERTISEMENT
Trinity has paid $40m on closing and will pay a maximum of $6.4m after 12 months and a maximum of a further $5.5m after 24 months. Of these combined deferred payments, an amount of $5.5m is contingent on the achievement of certain milestones for the product line during the remainder of 2006. The transaction is being funded from a combination of cash and bank debt.
Antwort auf Beitrag Nr.: 24.878.875 von Vakataka am 26.10.06 22:55:51wenn die IR-Arbeit besser wäre, dann würde der Kurs nicht unter 10 Dollar stehen und es wären mehr Fonds investiert. Die Meldung über den Zukauf (Nephrothek) und die Abwerbung des Managers beim Mitkonkurrenten Orasure hätte sicher für weitere steigende Kurse gesorgt.
Leider erfahren wir nicht, wieviel Umsatz über Nephrotek dazu gekauft wurde. Allerdings bei so einem geringen Kaufpreis war wohl nur der Marktzugang in Frankreich wichtig.
Leider erfahren wir nicht, wieviel Umsatz über Nephrotek dazu gekauft wurde. Allerdings bei so einem geringen Kaufpreis war wohl nur der Marktzugang in Frankreich wichtig.
Antwort auf Beitrag Nr.: 24.882.611 von Dangerous am 27.10.06 09:42:34Hallo Dangerous
Folgendes posting ist aus dem yahoo board, der hat den CC mitgehört. Wenn ich das richtig verstehe ist der Biomerieux Zukauf
umatzmäßig nicht voll in das Quartal eingegangen.
Bin beruhigt und sehe jetzt gelassen in die Zukunft.
und sehe jetzt gelassen in die Zukunft.
Man beachte den letzten Absatz unten!
earnings (2 Ratings) 26-Oct-06 11:11 pm
Best CC in a long time. Numbers were not only good but detailed. Projections were detailed. And, by the way, it is $250M in 2009, not 2008. Note that TRIB has never made more than $.04 per share on a sustained basis before diluting. They are now again over $.03 (before reverse split) with only a small part of the Biomiriex (sp?) acquisition income coming in. The bottom line is finally benefitting. As I noted previously the major "if" is further dilution. However, since they have over three years to make the $250M, organic growth and new products should do it. $170M next year with 12% growth from this year and the $40M from the acquistion (Rory Nealon, CFO, said he feels even stronger now about he $40M number) is expected. At 12% organic growth per year 2008 would be $190M and 2009 $213M -- close enough for my comfort level. As the gross goes up so does the margin and the net. Today we saw the beginning of a solid move forward. There were more American analysts/investors on the CC. Shares were 153,000 traded -- on a CC!! Price was up $0.91 on earnings!! Yes, they can pull the old Sally/Charlie Brown yank the football trick again but they really don't need to. They beat Wampole in court, have a China manufacturing facility, are making hundreds of millions. They have the respect they wanted now they can share with the shareholders. Again, yes, "if" no dilution and "if" they absord the acquistion without screwing up. I'm willing to stick around another 12-18 months (as I said before)to see how it all comes out -- after years of being on a slow moving train. Time will tell and we will all revisit our predictions at the next CC. Hope the bottom line keeps going up.
And yes, the CLIA waiver for Tri-stat(Diabetes test) is expected by year end. The European launch will be 11/15 in Dusseldorf. Western Blot Q2 of next year. More in the pipeline.
Great luck to us all!
Folgendes posting ist aus dem yahoo board, der hat den CC mitgehört. Wenn ich das richtig verstehe ist der Biomerieux Zukauf
umatzmäßig nicht voll in das Quartal eingegangen.
Bin beruhigt
 und sehe jetzt gelassen in die Zukunft.
und sehe jetzt gelassen in die Zukunft.Man beachte den letzten Absatz unten!
earnings (2 Ratings) 26-Oct-06 11:11 pm
Best CC in a long time. Numbers were not only good but detailed. Projections were detailed. And, by the way, it is $250M in 2009, not 2008. Note that TRIB has never made more than $.04 per share on a sustained basis before diluting. They are now again over $.03 (before reverse split) with only a small part of the Biomiriex (sp?) acquisition income coming in. The bottom line is finally benefitting. As I noted previously the major "if" is further dilution. However, since they have over three years to make the $250M, organic growth and new products should do it. $170M next year with 12% growth from this year and the $40M from the acquistion (Rory Nealon, CFO, said he feels even stronger now about he $40M number) is expected. At 12% organic growth per year 2008 would be $190M and 2009 $213M -- close enough for my comfort level. As the gross goes up so does the margin and the net. Today we saw the beginning of a solid move forward. There were more American analysts/investors on the CC. Shares were 153,000 traded -- on a CC!! Price was up $0.91 on earnings!! Yes, they can pull the old Sally/Charlie Brown yank the football trick again but they really don't need to. They beat Wampole in court, have a China manufacturing facility, are making hundreds of millions. They have the respect they wanted now they can share with the shareholders. Again, yes, "if" no dilution and "if" they absord the acquistion without screwing up. I'm willing to stick around another 12-18 months (as I said before)to see how it all comes out -- after years of being on a slow moving train. Time will tell and we will all revisit our predictions at the next CC. Hope the bottom line keeps going up.
And yes, the CLIA waiver for Tri-stat(Diabetes test) is expected by year end. The European launch will be 11/15 in Dusseldorf. Western Blot Q2 of next year. More in the pipeline.
Great luck to us all!
Antwort auf Beitrag Nr.: 24.888.973 von Vakataka am 27.10.06 14:37:42Ich stelle hier nochmal eine Aussage von tallyrand rein.
Die Zahlen die er nennt sind denen ähnlich die wir vor ein oder zwei Jahren hier genannt haben. Kurzfristig kann man mit anderen Aktien sicher mehr verdienen, langfristig bin ich weiter positiv.
Re: earnings (Not rated) 20 minutes ago
I am using a 20-30 p/e multiple. I think in 2007 they will come close to $.20/share (pre-split). This means $4-$6 ($16-$24)per share. However, we have seen that excitement over a product that can give an artifical pop. I would hope that OTC for the HIV Unigold comes by the end of next year. That could move the stock -- at least temporarily -- towards the 30 p/e range. Simply, there are exit points -- "if" they don't play games.
In 2009 $220M will generate about $22M in net profits after taxes, etc. Net, net. With 75M (18.6M pre-split)shares that translates into $1.30/share. Share price $26-$39 depending upon organic growth potential (will the history support 12% continued growth organically) are there products in the FDA pipeline with good income potential, is their chemical platform still competitive (are their products still cutting edge)does a bigger competitor go after them with a price war or more, etc.
I'd like to sell next year or beginning of 2008. A short enough tmeline where too much change is not expected (and no more dilution). I'd hope to exit at $20-$25 and it may be on an FDA approval "pop."
A lot of variables. The knowns are the company always makes money, grows year after year, has numerous products, its own mature sales force around the world, licenses from Walmpole, manufacturing economies from its China plant, new products emerging as I write this, and a vision to be in the top four diagnostic companies globally (and of course dilution). I'd love to see a 2-1 split at $16 next year. Each person must balance his/her own goals, his/her own timeline for holding, and all the variables. Giving weight to some over others becomes much more of an art than a science.
Best of luck to us all!
Die Zahlen die er nennt sind denen ähnlich die wir vor ein oder zwei Jahren hier genannt haben. Kurzfristig kann man mit anderen Aktien sicher mehr verdienen, langfristig bin ich weiter positiv.
Re: earnings (Not rated) 20 minutes ago
I am using a 20-30 p/e multiple. I think in 2007 they will come close to $.20/share (pre-split). This means $4-$6 ($16-$24)per share. However, we have seen that excitement over a product that can give an artifical pop. I would hope that OTC for the HIV Unigold comes by the end of next year. That could move the stock -- at least temporarily -- towards the 30 p/e range. Simply, there are exit points -- "if" they don't play games.
In 2009 $220M will generate about $22M in net profits after taxes, etc. Net, net. With 75M (18.6M pre-split)shares that translates into $1.30/share. Share price $26-$39 depending upon organic growth potential (will the history support 12% continued growth organically) are there products in the FDA pipeline with good income potential, is their chemical platform still competitive (are their products still cutting edge)does a bigger competitor go after them with a price war or more, etc.
I'd like to sell next year or beginning of 2008. A short enough tmeline where too much change is not expected (and no more dilution). I'd hope to exit at $20-$25 and it may be on an FDA approval "pop."
A lot of variables. The knowns are the company always makes money, grows year after year, has numerous products, its own mature sales force around the world, licenses from Walmpole, manufacturing economies from its China plant, new products emerging as I write this, and a vision to be in the top four diagnostic companies globally (and of course dilution). I'd love to see a 2-1 split at $16 next year. Each person must balance his/her own goals, his/her own timeline for holding, and all the variables. Giving weight to some over others becomes much more of an art than a science.
Best of luck to us all!
hallo? noch jemand investiert?  gibt leider nicht viel neues oder?
gibt leider nicht viel neues oder?
 gibt leider nicht viel neues oder?
gibt leider nicht viel neues oder?
Antwort auf Beitrag Nr.: 25.979.767 von girokonto am 06.12.06 19:58:25Hey
Werden wir die 25D bald sehen?
cheers

Werden wir die 25D bald sehen?
cheers
Antwort auf Beitrag Nr.: 26.825.519 von serafino am 11.01.07 09:32:37Wie kommst du auf 25USD? zwar erholt sich der Kurs die letzten Tage wieder, aber ich hab noch nichts neues gehört. Der Kurs dümpelt mehr oder weniger seitwärts, da müssen wohl mal sehr gute Zahlen her das er sich mehr nach Norden bewegt. Ich hatte nochmal paar Stücke nachgelegt mal sehn was die nächsten Wochen passiert.
Kommen bald NEWS raus?
Antwort auf Beitrag Nr.: 27.107.102 von serafino am 22.01.07 11:47:51Ich denke im Februar /März kommen Zahlen fürs 4.Quartal zumindest lt. Homepage, warten wir mal ab, wüßte nicht was jetzt demnächst kommen könnte.
Hoffe der Kurs kann die 7EUR bzw. 9USD mal dauerhaft überspringen.
Hoffe der Kurs kann die 7EUR bzw. 9USD mal dauerhaft überspringen.
Warum steigt nicht auch bei uns?  in den USA aber sehr wohl . Spielt der MM alles runter?
in den USA aber sehr wohl . Spielt der MM alles runter?
cheers
 in den USA aber sehr wohl . Spielt der MM alles runter?
in den USA aber sehr wohl . Spielt der MM alles runter?cheers
Antwort auf Beitrag Nr.: 27.143.405 von serafino am 24.01.07 09:44:14Naja ich finde die Entwicklung gar nicht so schlecht. Leider ziehen wir immer nur nach. D.h in orientiert der Kurs immer nur am Vortagesschluß von USA.
Der liegt oft in der mitte von BID/ASK und in Frankfurt hält sich der Kurs immer am unteren Ende. Wir legen leider nie etwas vor.
Dennoch bin ich nach den letzten Tagen wieder optimistisch, hatte ja letztes Jahr nochmal nachgelegt.
Der liegt oft in der mitte von BID/ASK und in Frankfurt hält sich der Kurs immer am unteren Ende. Wir legen leider nie etwas vor.
Dennoch bin ich nach den letzten Tagen wieder optimistisch, hatte ja letztes Jahr nochmal nachgelegt.
NEWS? An der NASDAQ + über 3%





das Mehjahreshoch bei 9,50 US-D wurde gebrochen. Wenn wir dann noch die 10 Dollar überwinden, kann es Richtung 12-15 US-D laufen. Hoffen wir auf baldige gute Quartalszahlen.
An der NASDAQ heute schon über 10$?
trong buy
trong buy
S...
Antwort auf Beitrag Nr.: 27.743.694 von Dangerous am 16.02.07 11:58:21alle 4 Jahre geht es Richtung 25$
http://finance.yahoo.com/q/bc?s=TRIB&t=my&l=on&z=m&q=l&c=
http://finance.yahoo.com/q/bc?s=TRIB&t=my&l=on&z=m&q=l&c=
Antwort auf Beitrag Nr.: 27.761.166 von Vakataka am 16.02.07 21:11:39Da hast du schon recht. Aber meist gab es auch einen Grund dafür.
Vor 3 Jahren die Zulassung des HIV Tests durch die FDA, vor 7 Jahren der allgemeine Run auf Biotech-Aktien.
Und diesmal???
Vor 3 Jahren die Zulassung des HIV Tests durch die FDA, vor 7 Jahren der allgemeine Run auf Biotech-Aktien.
Und diesmal???
Antwort auf Beitrag Nr.: 27.783.079 von Silenttrader am 17.02.07 19:48:49Hey man kann immer noch einsteigen !
Bis 25$ ist noch viel Luft ,noch über 100% steigerung
Ende des Monat glaube ich schon über 12$=über 9E
An der NASDAQ ist was los !
cheers

Bis 25$ ist noch viel Luft ,noch über 100% steigerung

Ende des Monat glaube ich schon über 12$=über 9E

An der NASDAQ ist was los !
cheers
Na ja vielleicht sehen wir doch die 26$ wieder 
cheers

cheers
Das war heute gar nicht übel
10.45 Change: +0.57(+5.77%) Volume: 189.55 k
10.45 Change: +0.57(+5.77%) Volume: 189.55 k
Antwort auf Beitrag Nr.: 27.863.473 von Vakataka am 20.02.07 22:33:26Yesss der Truck ist im Bewegung geraten gen NORD
Bald 25$?
cheers

Bald 25$?
cheers
Antwort auf Beitrag Nr.: 27.864.235 von serafino am 20.02.07 22:54:41Und jetzt ist er gerade beim Boxenstopp! 
Die neuen Zahlen müssten eigentlich bald kommen, die geben hoffentlich weiter Auftrieb.
Full speed ahead!!!

Die neuen Zahlen müssten eigentlich bald kommen, die geben hoffentlich weiter Auftrieb.
Full speed ahead!!!

March 5, 2007 - 10:19 AM EST
Trinity Biotech plc: 2006 Full Year Results
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2006 full year results on Tuesday, 6 March, 2007.
The Trinity Biotech management team will discuss full year financial performance with investors and analysts on a conference call scheduled for 3.00pm BST (10.00am ET) on that date.
Dial-in details as follows:
USA 877 407 9210
International +1 201 689 8049
Playback Dial-In as follows:
USA 877 660 6853
International +1 201 612 7415
Playback Account # 286
Playback Conference ID # 233853
Playback is available until March 7.
Webcast:
The call is being webcast and can be accessed at www.vcall.com
Source: Market Wire (March 5, 2007 - 10:19 AM EST)
Trinity Biotech plc: 2006 Full Year Results
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2006 full year results on Tuesday, 6 March, 2007.
The Trinity Biotech management team will discuss full year financial performance with investors and analysts on a conference call scheduled for 3.00pm BST (10.00am ET) on that date.
Dial-in details as follows:
USA 877 407 9210
International +1 201 689 8049
Playback Dial-In as follows:
USA 877 660 6853
International +1 201 612 7415
Playback Account # 286
Playback Conference ID # 233853
Playback is available until March 7.
Webcast:
The call is being webcast and can be accessed at www.vcall.com
Source: Market Wire (March 5, 2007 - 10:19 AM EST)
Antwort auf Beitrag Nr.: 28.132.659 von Vakataka am 05.03.07 19:56:42Auf www.nasdaq.com sind sie raus. Unter company news "trib" eingeben.
Antwort auf Beitrag Nr.: 28.147.993 von Ren am 06.03.07 15:59:36Na, das Ergebnis wird den Kurs wieder schön zurückbringen.
Umsatz rauf, Profit runter, wie jedes Mal.
Umsatz rauf, Profit runter, wie jedes Mal.

Antwort auf Beitrag Nr.: 28.148.287 von Lubus104 am 06.03.07 16:09:29Ich hab die Zahlen noch nicht gesehen, aber wenn ich nach USA schaue und den Kurs sehe, dann ist wirklich wieder alles klar, kann die Truppe nicht auch mal ne Überraschung raushauen. 

Antwort auf Beitrag Nr.: 28.148.861 von girokonto am 06.03.07 16:31:03Und ich hab` mal gedacht ich hätte da eine Perle erwischt. Aber irgendwie kriegen die das nicht auf die Reihe den Gewinn stärker als den Umsatz steigen zu lassen.
Na ja, so schlecht sind die Zahlen auf den zweiten Blick gar nicht. Keine Ahnung, was jetzt da erwartet wurde.
Die Abschreibung war klar, gut die Kosten in "selling, general and administrative expenses" sind etwas hoch, aber das kommt nur wegen der Übernahme vom Juni und den Aufbau des Vertriebs in Frankreich. Und da müssen wir jetzt durch, um neue Höhen zu erreichen.
Ich denke, dass wird sich auch heute im Tageskursverlauf zeigen, erst der Schreck und dann das "AHA".
@Lubus104
Hast schon eine Perle erwischt! Der Schlamm muss nur noch abgewaschen werden!
Die Abschreibung war klar, gut die Kosten in "selling, general and administrative expenses" sind etwas hoch, aber das kommt nur wegen der Übernahme vom Juni und den Aufbau des Vertriebs in Frankreich. Und da müssen wir jetzt durch, um neue Höhen zu erreichen.
Ich denke, dass wird sich auch heute im Tageskursverlauf zeigen, erst der Schreck und dann das "AHA".
@Lubus104
Hast schon eine Perle erwischt! Der Schlamm muss nur noch abgewaschen werden!
@ Silenttrader
Im groben sehe ich das ähnlich.
Die negativen Dinge möchte ich nicht hervorheben, die positiven werden hoffentlich überwiegen.
Am 7. April kommen die Q1 Zahlen
Im groben sehe ich das ähnlich.
Die negativen Dinge möchte ich nicht hervorheben, die positiven werden hoffentlich überwiegen.
Am 7. April kommen die Q1 Zahlen
Antwort auf Beitrag Nr.: 28.780.314 von serafino am 12.04.07 17:40:47nö, noch dabei,aber es gibt meines Wissens nichts Neues, das sich lohnt zu posten und die geringen Kursaufschläge der letzten Tage hatten wir immer wieder, um dann wieder zurück zu gehen. Also warten wir auf bessere Tage!
Bis dann,
Lubus
Bis dann,
Lubus
Antwort auf Beitrag Nr.: 28.347.780 von Vakataka am 17.03.07 20:53:40wurden die Quartalszahlen verschoben?
beste Grüße Danger
beste Grüße Danger
Antwort auf Beitrag Nr.: 28.788.487 von Dangerous am 13.04.07 08:48:21Ich habe mich wohl irgendwo verlesen.
2006 war es der 27. April.
Gruß vaka
2006 war es der 27. April.
Gruß vaka
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will release 2007 first quarter results on Thursday, 26 April, 2007.
The Trinity Biotech management team will discuss first quarter financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
Dial-in details as follows:
USA 877 407 8031
International +1 201 689 8031
Playback Dial-In as follows:
USA 877 660 6853
International +1 201 612 7415
Playback Account # 286
Playback Conference ID # 239805
Playback is available until 27 April.
Webcast:
The call is being webcast and can be accessed at www.vcall.com
Further details are available from
K Capital Source
+353 1 631 5500
Email Contact
Source: Market Wire (April 24, 2007 - 8:35 AM EDT)
The Trinity Biotech management team will discuss first quarter financial performance with investors and analysts on a conference call scheduled for 4.00pm BST (11.00am ET) on that date.
Dial-in details as follows:
USA 877 407 8031
International +1 201 689 8031
Playback Dial-In as follows:
USA 877 660 6853
International +1 201 612 7415
Playback Account # 286
Playback Conference ID # 239805
Playback is available until 27 April.
Webcast:
The call is being webcast and can be accessed at www.vcall.com
Further details are available from
K Capital Source
+353 1 631 5500
Email Contact
Source: Market Wire (April 24, 2007 - 8:35 AM EDT)




Was geht denn jetzt los, hab ich etwas verpasst
Geld 7,97 14:30:28 1.500 Stk.
Brief 8,60 14:30:28 1.500 Stk.
Ist ja geil aktuell schon 9% plus



Geld 8,37 15:10:12 1.500 Stk.
Brief 9,00 15:10:12 1.500 Stk.
Also wenn das so bleibt und weitergeht, dann bekomm ich Angst


Brief 9,00 15:10:12 1.500 Stk.
Also wenn das so bleibt und weitergeht, dann bekomm ich Angst



Antwort auf Beitrag Nr.: 29.014.943 von girokonto am 26.04.07 15:21:29Sind die Zahlen schon raus??
Antwort auf Beitrag Nr.: 29.015.614 von Silenttrader am 26.04.07 15:50:26hab sie noch nicht gefunden aber heute war ja der conference call, ich nehme an die Zahlen sind gut

Antwort auf Beitrag Nr.: 29.015.614 von Silenttrader am 26.04.07 15:50:26Ach du meine Fresse! Upps, 'tschuldigung. Na, endlich! Die Zahlen klingen gut.
http://biz.yahoo.com/iw/070426/0244434.html
http://biz.yahoo.com/iw/070426/0244434.html

Antwort auf Beitrag Nr.: 29.015.705 von Silenttrader am 26.04.07 15:54:36endlich mal sehr gute Zahlen auf die ich schon seit 3 Jahren warte. Die Analystenerwartung lag bei 0,10 US-$ Gewinn je Aktie. Das die Erwartung deutlich übertroffen wurde, zeigt sich im heutigen Kurssprung. Gut wäre, wenn es jetzt ein Update zum Forecast für 2007 geben könnte. Man könnte von einem Jahresgewinn von 0,60-0,70 US-$ träumen. Bei einem KGV von ca. 25 wäre ein Kurs so um die 15 US-$ angemessen. Vielleicht gibt es dann wieder den im Chart zu erkennenden vierjährlichen Ausbruch auf 25 US-$. Dann hätte sich endlich das lange Warten gelohnt 

Die Zahlen sind raus und der Kurs geht nicht nach unten!
Das ich das noch erleben darf
Sieht auf den ersten Blick gar nicht so schlecht aus.
Hoffentlich nehmen die Trader nicht alle ihre Gewinne
mit.
Das ich das noch erleben darf

Sieht auf den ersten Blick gar nicht so schlecht aus.
Hoffentlich nehmen die Trader nicht alle ihre Gewinne
mit.
Hat hier jemand den CC mitgehört?
Hier noch eine Zusammenfassung, Meinung, aus dem Yahoo Board:
The CC made clear that only $500,000 of the deferred came in Q1, not $1.7M. They grossed $36.7 million! Only $500,000 was a carryover. This compared to a gross of $24.7 same quarter last year.
2007 will be a good year but will suffer some from the prior acquisition. The costs of transferring products, training personnel,etc. will peak in 2007 and in 2008 (8 mos. away) will decline. Inventories taken from Biomerieux that had been stockpiled in advance of acquistion should be utilized by end of June, the invenstories will decline.
HIV market in US continues to grow. CDC was giving away OSUR product initially. TRIB sales force has gotten market share by educating those that were unaware or Unigold (and had been lured to Oraquick) that TRIB UniGold is 1) more accurate, 2) quicker and 3) has a longer shelf life.
Diabetes test to be apprvoed with CLIA waiver Q3. Delay was due to change in instrumentation to allow for 3 simoulteneous tests and easier use. It will be best on the market. FDA required resubmission and trial tests. This is all done and Q3 launch is expected. This is a $100 million market.
Western Blot expected to be approved Q1 of 2008. Submitted and should be fast tracked since FDA wants a second product on the market. There is currently only one. Last time a number was attached (years ago) this was a $40Million market.
African Unigold tests being moved to China for production. This will be finished Q3 and will lower production costs/increase profit margins.
A large version of one of their instrumentation products will be demonstrated in San Diego later this year. They expect this product to generate additional revenue.
Organic growth still factored at over 10% per annum.
Over the counter approval of HIV unigold is optimistically expected next year.
More detail on specifics was gicen but the above is a flavor for the future.
As always, if they do not dillute or otherwise stumble and can keep up the good work for three more quarters the year should end at $12-$18 depending on p/e ($.60 or more for the year). 2008 would then be dramrically good. They just have to grow what they have and stop buying.
Good luck to us all!
Hier noch eine Zusammenfassung, Meinung, aus dem Yahoo Board:
The CC made clear that only $500,000 of the deferred came in Q1, not $1.7M. They grossed $36.7 million! Only $500,000 was a carryover. This compared to a gross of $24.7 same quarter last year.
2007 will be a good year but will suffer some from the prior acquisition. The costs of transferring products, training personnel,etc. will peak in 2007 and in 2008 (8 mos. away) will decline. Inventories taken from Biomerieux that had been stockpiled in advance of acquistion should be utilized by end of June, the invenstories will decline.
HIV market in US continues to grow. CDC was giving away OSUR product initially. TRIB sales force has gotten market share by educating those that were unaware or Unigold (and had been lured to Oraquick) that TRIB UniGold is 1) more accurate, 2) quicker and 3) has a longer shelf life.
Diabetes test to be apprvoed with CLIA waiver Q3. Delay was due to change in instrumentation to allow for 3 simoulteneous tests and easier use. It will be best on the market. FDA required resubmission and trial tests. This is all done and Q3 launch is expected. This is a $100 million market.
Western Blot expected to be approved Q1 of 2008. Submitted and should be fast tracked since FDA wants a second product on the market. There is currently only one. Last time a number was attached (years ago) this was a $40Million market.
African Unigold tests being moved to China for production. This will be finished Q3 and will lower production costs/increase profit margins.
A large version of one of their instrumentation products will be demonstrated in San Diego later this year. They expect this product to generate additional revenue.
Organic growth still factored at over 10% per annum.
Over the counter approval of HIV unigold is optimistically expected next year.
More detail on specifics was gicen but the above is a flavor for the future.
As always, if they do not dillute or otherwise stumble and can keep up the good work for three more quarters the year should end at $12-$18 depending on p/e ($.60 or more for the year). 2008 would then be dramrically good. They just have to grow what they have and stop buying.
Good luck to us all!
Hey  werden wir schon im dieses Jahr die 16E..1/4,1 Splitting
werden wir schon im dieses Jahr die 16E..1/4,1 Splitting
sehen?
cheers
 werden wir schon im dieses Jahr die 16E..1/4,1 Splitting
werden wir schon im dieses Jahr die 16E..1/4,1 Splitting sehen?

cheers
Antwort auf Beitrag Nr.: 29.035.829 von serafino am 27.04.07 20:33:44Hey ! Endlich haben wir di Kurve geschaft 
Eigentlich sollte die AZH wieder erreichbar sein
cheers

Eigentlich sollte die AZH wieder erreichbar sein

cheers
Antwort auf Beitrag Nr.: 29.129.150 von serafino am 04.05.07 11:31:18Hallo,
kannste mal genauer ausführen, welche Kurve wir geschafft haben und wie du darauf kommst, dass das AZH in nächster Zeit erreichbar ist? Scheint mir doch ein etwas überzogener Optimismus auf diese kleine Kursbewegung zu sein.
kannste mal genauer ausführen, welche Kurve wir geschafft haben und wie du darauf kommst, dass das AZH in nächster Zeit erreichbar ist? Scheint mir doch ein etwas überzogener Optimismus auf diese kleine Kursbewegung zu sein.
Antwort auf Beitrag Nr.: 29.130.181 von Lubus104 am 04.05.07 12:36:36Naja das Gequatsche vom ATH finde ich etwas übertrieben, aber zumindest haben die Zahlen diesmal nicht wieder enttäuscht und so kann man zumindest auf eine positive Entwicklung setzen.
Was allerdings passiert wenn der Markt dreht bleibt abzuwarten ich werde meine Gewinne mit SL absichern, später wieder rein geht immer. Hab ich schon oft bei TB gemacht und der Chart gibt es auch her.
Was allerdings passiert wenn der Markt dreht bleibt abzuwarten ich werde meine Gewinne mit SL absichern, später wieder rein geht immer. Hab ich schon oft bei TB gemacht und der Chart gibt es auch her.
Antwort auf Beitrag Nr.: 29.132.780 von girokonto am 04.05.07 15:14:45Gequatsche?
Bist Du ausgestiegen? na ja kannst Du wieder einsteigen
cheers

Bist Du ausgestiegen? na ja kannst Du wieder einsteigen

cheers
Antwort auf Beitrag Nr.: 29.132.780 von girokonto am 04.05.07 15:14:45Danke für deine Meinung. Sag` mal, ist das über SL nicht ziemlich gefährlich bei dem geringen Volumen in Old Germany, oder handelst du direkt an der Nasdaq ?
Antwort auf Beitrag Nr.: 29.146.380 von Lubus104 am 05.05.07 07:01:20Ich bin mittlerweile wieder ordentlich im Plus und von daher sichere ich mich lieber ab als wieder 1 Jahr zu warten. Ich sehe den Gesamtmarkt vor einer Korrektur wann auch immer die kommt und ich möchte vermeiden, danach wieder ewig warten zu müssen bis TB wieder anspringt.
Bin schon einige Jahre in TB und bisher hatte ich Glück mit der Strategie. So konnte ich nahe am ATH verkaufen und bin danach wieder in mehreren Schritten rein. Natürlich hoffe ich der Chart läuft jetzt weiter.
Bin noch drin, handel aber nur in Deutschland, denn so groß ist meine Position auch nicht.
Bin schon einige Jahre in TB und bisher hatte ich Glück mit der Strategie. So konnte ich nahe am ATH verkaufen und bin danach wieder in mehreren Schritten rein. Natürlich hoffe ich der Chart läuft jetzt weiter.
Bin noch drin, handel aber nur in Deutschland, denn so groß ist meine Position auch nicht.
 gen Nord
gen Nord  AZH bald
AZH bald
cheers
Antwort auf Beitrag Nr.: 29.192.152 von serafino am 07.05.07 17:52:08
AZH lag irgend was bei 30 $ !!
erst informieren und nicht von dem kurzen Chart verwirren lassen....

AZH lag irgend was bei 30 $ !!
erst informieren und nicht von dem kurzen Chart verwirren lassen....
Antwort auf Beitrag Nr.: 29.193.588 von LastDrow am 07.05.07 18:59:16sag ich ja kaum steigen wir ein bisschen wird ATH geschrien....
Bin gut dabei, von daher ist jeder weitere Anstieg prima

Bin gut dabei, von daher ist jeder weitere Anstieg prima
May 23, 2007 - 10:00 AM EDT
TRIB 10.98 0.09
Today 5d 1m 3m 1y 5y 10y
Trinity Biotech Announces Agreement With the Clinton Foundation HIV/AIDS Initiative (CHAI)
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced an agreement with the Clinton Foundation HIV/AIDS Initiative (CHAI) in connection with its work on behalf of the International Drug Purchase Facility (UNITAID) established by the governments of Brazil, France, Chile, Norway and the United Kingdom. UNITAID is a newly established funding mechanism to accelerate access to high-quality drugs and diagnostics for HIV/AIDS, malaria and tuberculosis in countries with a high burden of disease.
Under the agreement, Trinity Biotech will immediately begin offering its Uni-Gold and Capillus rapid HIV test kits at a discounted price to UNITAID-eligible countries, together with members of CHAI's Procurement Consortium, which is a coalition of more than 60 developing countries around the world.
Ira Magaziner, the Chairman of CHAI, commented on the agreement saying that: "We face a global HIV/AIDS emergency. More than 90% of the 40 million people living with HIV in the world, including 2-3 million children under the age of 15, do not know that they are HIV-infected. And with 5 million new infections each year (one person every 6 seconds), increasing use of rapid HIV tests is and must be a vital component of our global HIV/AIDS prevention strategy. In order to reach treatment targets, developing countries need to run at least 200 million HIV tests in the next four years. That is why our ability to scale up the use of highly accurate and efficient rapid HIV testing is a first line defense. As such, we are pleased to work with Trinity and commend them for helping to make Uni-Gold and Capillus available to members of our Procurement Consortium and UNITAID countries."
Commenting on the agreement, Brendan Farrell, President of Trinity Biotech, said: "We are delighted to work with the Clinton Foundation in conjunction with its work for UNITAID. We believe there are synergies between our demonstrated ability to supply large volume, highly accurate, rapid HIV test kits and the goals of both President Clinton's Foundation and UNITAID. We look forward to assisting both organizations as they support countries across much of the developing world in their scale-up of HIV rapid testing initiatives."
Since 2002, the Clinton Foundation HIV/AIDS Initiative has been assisting countries in implementing large-scale, integrated care and treatment programs. It partners with 25 countries in Africa, the Caribbean and Asia. Individual governments take the lead and the Foundation provides technical assistance, mobilizes human and financial resources, and facilitates the sharing of best practices across projects. The Foundation also provides access to reduced prices for HIV/AIDS drugs and diagnostics to more than 60 countries.
UNITAID was officially launched on 19 September 2006 in New York on the occasion of the Opening Session of the United Nations General Assembly by France's President Jacques Chirac, Brazil's President Luiz Inacio Lula da Silva, Norway's Prime Minister Jens Stoltenberg, Chile's Foreign Minister Alejandro Foxley Rioseco, Great Britain's Parliamentary Under-Secretary of International Development Gareth Thomas, United Nations Secretary-General Kofi Annan, the African Union's President Denis Sassou-Nguesso and the former U.S. President, Bill Clinton. The mission of UNITAID is to scale up access to treatment for HIV/AIDS, tuberculosis and malaria for the poor in developing countries by lowering the price of drugs and diagnostics and accelerating the pace at which they are made available.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits is used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Contact:
Trinity Biotech plc
Brendan Farrell
+ 353 1 276 9800
E-mail: Email Contact
Clinton Foundation HIV/AIDS Initiative (CHAI)
New York Office
+1 212 348 0360
Source: Market Wire (May 23, 2007 - 10:00 AM EDT)
News by QuoteMedia
www.quotemedia.com
TRIB 10.98 0.09
Today 5d 1m 3m 1y 5y 10y
Trinity Biotech Announces Agreement With the Clinton Foundation HIV/AIDS Initiative (CHAI)
Trinity Biotech plc (NASDAQ: TRIB) (DUBLIN: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced an agreement with the Clinton Foundation HIV/AIDS Initiative (CHAI) in connection with its work on behalf of the International Drug Purchase Facility (UNITAID) established by the governments of Brazil, France, Chile, Norway and the United Kingdom. UNITAID is a newly established funding mechanism to accelerate access to high-quality drugs and diagnostics for HIV/AIDS, malaria and tuberculosis in countries with a high burden of disease.
Under the agreement, Trinity Biotech will immediately begin offering its Uni-Gold and Capillus rapid HIV test kits at a discounted price to UNITAID-eligible countries, together with members of CHAI's Procurement Consortium, which is a coalition of more than 60 developing countries around the world.
Ira Magaziner, the Chairman of CHAI, commented on the agreement saying that: "We face a global HIV/AIDS emergency. More than 90% of the 40 million people living with HIV in the world, including 2-3 million children under the age of 15, do not know that they are HIV-infected. And with 5 million new infections each year (one person every 6 seconds), increasing use of rapid HIV tests is and must be a vital component of our global HIV/AIDS prevention strategy. In order to reach treatment targets, developing countries need to run at least 200 million HIV tests in the next four years. That is why our ability to scale up the use of highly accurate and efficient rapid HIV testing is a first line defense. As such, we are pleased to work with Trinity and commend them for helping to make Uni-Gold and Capillus available to members of our Procurement Consortium and UNITAID countries."
Commenting on the agreement, Brendan Farrell, President of Trinity Biotech, said: "We are delighted to work with the Clinton Foundation in conjunction with its work for UNITAID. We believe there are synergies between our demonstrated ability to supply large volume, highly accurate, rapid HIV test kits and the goals of both President Clinton's Foundation and UNITAID. We look forward to assisting both organizations as they support countries across much of the developing world in their scale-up of HIV rapid testing initiatives."
Since 2002, the Clinton Foundation HIV/AIDS Initiative has been assisting countries in implementing large-scale, integrated care and treatment programs. It partners with 25 countries in Africa, the Caribbean and Asia. Individual governments take the lead and the Foundation provides technical assistance, mobilizes human and financial resources, and facilitates the sharing of best practices across projects. The Foundation also provides access to reduced prices for HIV/AIDS drugs and diagnostics to more than 60 countries.
UNITAID was officially launched on 19 September 2006 in New York on the occasion of the Opening Session of the United Nations General Assembly by France's President Jacques Chirac, Brazil's President Luiz Inacio Lula da Silva, Norway's Prime Minister Jens Stoltenberg, Chile's Foreign Minister Alejandro Foxley Rioseco, Great Britain's Parliamentary Under-Secretary of International Development Gareth Thomas, United Nations Secretary-General Kofi Annan, the African Union's President Denis Sassou-Nguesso and the former U.S. President, Bill Clinton. The mission of UNITAID is to scale up access to treatment for HIV/AIDS, tuberculosis and malaria for the poor in developing countries by lowering the price of drugs and diagnostics and accelerating the pace at which they are made available.
Trinity Biotech develops, acquires, manufactures and markets over 500 diagnostic products for the point-of-care and clinical laboratory segments of the diagnostic market. The broad line of test kits is used to detect infectious diseases, sexually transmitted diseases, blood coagulation disorders, and autoimmune diseases. Trinity Biotech sells worldwide in over 80 countries through its own sales force and a network of international distributors and strategic partners. For further information please see the Company's website: www.trinitybiotech.com.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Contact:
Trinity Biotech plc
Brendan Farrell
+ 353 1 276 9800
E-mail: Email Contact
Clinton Foundation HIV/AIDS Initiative (CHAI)
New York Office
+1 212 348 0360
Source: Market Wire (May 23, 2007 - 10:00 AM EDT)
News by QuoteMedia
www.quotemedia.com
Hey 

Bin ganz allein noch investiert?
oder seit alle short ? Na ja wenn ja habt Euch vertan ahhhhhhhh
cheers


Bin ganz allein noch investiert?
oder seit alle short ? Na ja wenn ja habt Euch vertan ahhhhhhhh

cheers
Antwort auf Beitrag Nr.: 29.483.720 von serafino am 27.05.07 11:06:27keine Sorge, bist nicht allein 
Bin immernoch long und mittlerweile wieder gut dabei!

Bin immernoch long und mittlerweile wieder gut dabei!
Antwort auf Beitrag Nr.: 29.484.319 von girokonto am 27.05.07 13:08:59
25$ kommen bald strong buy
strong buy
cheers

25$ kommen bald
 strong buy
strong buycheers
Antwort auf Beitrag Nr.: 29.581.968 von serafino am 01.06.07 21:46:43Hallo serafinoxx
vorher müssen wir die 12$ knacken
vorher müssen wir die 12$ knacken

Hey die 12$ sind so gut wie schon gefallen ,nur der MM spielt ein wenig 
Ich denke wir werden AZH bald sehen ,die Weiche sind sehr gut angebracht und sehr Erfolgreicht gerichtet

cheers

Ich denke wir werden AZH bald sehen ,die Weiche sind sehr gut angebracht und sehr Erfolgreicht gerichtet


cheers
wie sich wohl die Übernahme von Digene von Qiagen auf TRIB auswirkt? Hoffentlich überspringen wir bald den Widerstand bei 10 US-D.
Antwort auf Beitrag Nr.: 29.886.150 von Dangerous am 14.06.07 10:08:42wir stehen bei USD 11,45
wie meinen?
wie meinen?

sorry 12 US-D. Das ist mein damaliger Kaufkurs von vor 3 Jahren. Leider ist seitdem der Euro so stark gewurden, so dass mein Break-even erst bei 15 US-D liegt 

Antwort auf Beitrag Nr.: 29.936.832 von Dangerous am 15.06.07 11:43:39hm also ich steh dick im plus bin bei 8 eur nochmal rein nach dem ich vorher schon seit 2000 dabei und nahe dem allzeithoch verkauft hatte, war echt mal richtig glück.
Mit denen die ich wieder im Depot habe bin ich jetzt bei knapp 20% plus, aber bin recht positiv auch wenn man den Chart betrachtet
Mit denen die ich wieder im Depot habe bin ich jetzt bei knapp 20% plus, aber bin recht positiv auch wenn man den Chart betrachtet
Aktie vor dem Ausbruch nach oben! Wann folgt der Vierjahresrhythmus Richtung 25 US-D?
Antwort auf Beitrag Nr.: 30.586.708 von Dangerous am 09.07.07 09:24:22noch kein Ausbruch  Wann kommen die Q-Zahlen?
Wann kommen die Q-Zahlen?
 Wann kommen die Q-Zahlen?
Wann kommen die Q-Zahlen?
Antwort auf Beitrag Nr.: 30.647.930 von Dangerous am 12.07.07 08:21:05Google + yahoo finance sagen am 8. August.
Antwort auf Beitrag Nr.: 30.659.910 von Silenttrader am 12.07.07 20:14:03So, Q2-Zahlen gibt's am Do., 9.August 07.
Antwort auf Beitrag Nr.: 31.009.965 von Silenttrader am 03.08.07 19:16:01So hier ein kleiner Auszug aus den Q2-Zahlen:
Aug 9 (Reuters) - TRINITY BIOTECH PLC (TRIB.O: Quote, Profile, Research)
(In $) ............... ............ Latest .... Yr ago qtr
Earnings/ADR (diluted) ..... 0.1 ........ -0.12
Net (thousands) ............... 2.6 ........ -2.1
Revenues (million) .......... 37.4 ........ 27.3
Reuters Estimates: EPS $0.14 (one analyst), revenue $38.4
million (one analyst)
Die kompletten Zahlen könnt ihr hier nachlesen:
http://www.trinitybiotech.com/data/press/PR%20Q2%20FY07%209%…
Aug 9 (Reuters) - TRINITY BIOTECH PLC (TRIB.O: Quote, Profile, Research)
(In $) ............... ............ Latest .... Yr ago qtr
Earnings/ADR (diluted) ..... 0.1 ........ -0.12
Net (thousands) ............... 2.6 ........ -2.1
Revenues (million) .......... 37.4 ........ 27.3
Reuters Estimates: EPS $0.14 (one analyst), revenue $38.4
million (one analyst)
Die kompletten Zahlen könnt ihr hier nachlesen:
http://www.trinitybiotech.com/data/press/PR%20Q2%20FY07%209%…
Hey TRIB ist kurz davor, bald wird exsplodieren 
strong buy
cheers

strong buy
cheers
Hey!
Wann ist A.T.H. fällig?
Wann kommen NEWS?
cheers
Wann ist A.T.H. fällig?
Wann kommen NEWS?
cheers
Was war denn da gestern plötzlich los? Riesenumsätze, die ich bei Trib noch nie gesehen habe.
Ab 5. November verlässt Trib die Heimatbörse in Irland. Das sollte aber keine Auswirkungen auf die Nasdaq haben, denn dort ist die Hauptbörse für Trib. Übrigens das jetzt seit genau 15 Jahren.
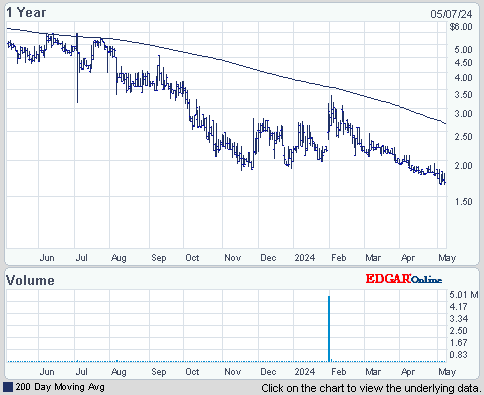
Ab 5. November verlässt Trib die Heimatbörse in Irland. Das sollte aber keine Auswirkungen auf die Nasdaq haben, denn dort ist die Hauptbörse für Trib. Übrigens das jetzt seit genau 15 Jahren.
Antwort auf Beitrag Nr.: 31.937.142 von honorar am 11.10.07 12:05:30Hier noch der Intraday-Chart von gestern, wo die Pakete ersichtlich sind:
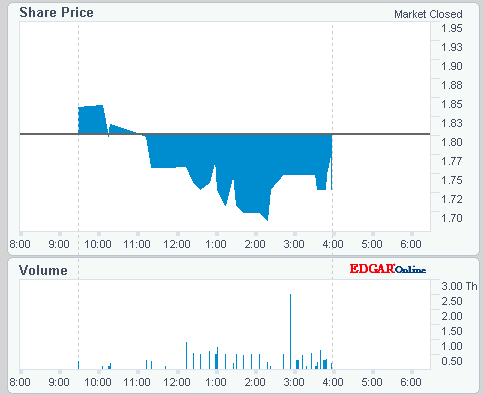
alle raus aus Trib? Die Quartalszahlen waren ja mal wieder ein Witz. Umsatz hui Gewinn pfui. Vielleicht sollten sie mal ein Aktienrückkauf starten und die Kosten reduzieren. Cash müsste ja genug vorhanden sein. Wahrscheinlich werden die Übernahmen zu teuer bezahlt und die Kostensynagieeffekte nicht genutzt.
Hey!
NEWS!!!!
Trinity Biotech names new CEO, CFO
Nov 8 (Reuters) - Trinity Biotech plc (TRIB.O: Quote, Profile, Research) said Chief Executive Officer Ronan O'Caoimh would be replaced by President Brendan Farrell.
O'Caoimh will be executive chairman, the company said in a statement.
Trinity Biotech said it named Chief Accounting Officer Kevin Tansley as chief financial officer. (Reporting by Sharangdhar Limaye in Bangalore
cheers
NEWS!!!!
Trinity Biotech names new CEO, CFO
Nov 8 (Reuters) - Trinity Biotech plc (TRIB.O: Quote, Profile, Research) said Chief Executive Officer Ronan O'Caoimh would be replaced by President Brendan Farrell.
O'Caoimh will be executive chairman, the company said in a statement.
Trinity Biotech said it named Chief Accounting Officer Kevin Tansley as chief financial officer. (Reporting by Sharangdhar Limaye in Bangalore
cheers

November 29, 2007 - 8:03 AM EST
Trinity Biotech Announces FDA Clearance of TRIstat(TM) Point-of-Care HbA1c Test
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that the US Food and Drug Administration has given clearance to market the rapid, point-of-care TRIstat(TM) HbA1c system.
The TRIstat(TM) point-of-care platform is designed to measure HbA1c, also known as glycated hemoglobin, which is a measure of a patient's blood sugar control over the previous two to three months. The availability of the TRIstat(TM) system will improve overall efficiency of diabetes care by delivering HbA1c values during patient's visits to their physicians and outpatient clinics. Utilizing a patented Affinity Two-Phase Optical system, together with a simple, fully automated, or plug-and-play design, TRIstat(TM) offers highly accurate results within minutes while eliminating complex, hands-on operator requirements found with other point-of-care technologies.
Diabetes is the fourth leading cause of death by disease in the world. According to the International Diabetes Federation, the number of people diagnosed with Type 2 diabetes worldwide has increased from 30 million to more than 246 million over the last two decades. This figure is projected to increase further to 380 million by 2025. In the USA, 20.8 million people (7 percent of the population) have the disease with a further 54 million considered to be pre-diabetic, according to the Centers for Disease Control and Prevention (CDC) and the American Diabetes Association (ADA).
The ADA strongly recommends that patients be tested for HbA1c at least 4 times a year. Testing should also be performed during treatment changes or after periods of elevated blood glucose levels. The ADA added a recommendation for point-of-care HbA1c monitoring to their 2006 professional practice guidelines emphasizing the importance of routine real-time HbA1c monitoring of persons with diabetes.
Commenting on the news, Brendan Farrell, CEO, said, "FDA Clearance of the TRIstat(TM) product is an extremely exciting development for Trinity Biotech and enables us to compete in the high growth point-of-care segment of the HbA1c market. As TRIstat(TM) provides test results within 10 minutes, patients will know their HbA1c status during their visit to their physician or diabetic clinic. This will assist the physician and patient to make immediate diabetes management decisions and ultimately improve outcomes."
"Our Ultra2(TM)and PDQ(TM) systems already address the $700m clinical lab market for HbA1c and the launch of TRIstat(TM) means that Trinity will now have a presence in the point-of-care segment which is estimated at $300m world wide. Discussions with distribution partners in the USA and Europe are well advanced and will be concluded shortly."
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Contact :
Trinity Biotech plc
Brendan Farrell
CEO
(353)-1-2769800
E-mail: Email Contact
Source: Marketwire (November 29, 2007 - 8:03 AM EST)
News by QuoteMedia
www.quotemedia.com
Trinity Biotech Announces FDA Clearance of TRIstat(TM) Point-of-Care HbA1c Test
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that the US Food and Drug Administration has given clearance to market the rapid, point-of-care TRIstat(TM) HbA1c system.
The TRIstat(TM) point-of-care platform is designed to measure HbA1c, also known as glycated hemoglobin, which is a measure of a patient's blood sugar control over the previous two to three months. The availability of the TRIstat(TM) system will improve overall efficiency of diabetes care by delivering HbA1c values during patient's visits to their physicians and outpatient clinics. Utilizing a patented Affinity Two-Phase Optical system, together with a simple, fully automated, or plug-and-play design, TRIstat(TM) offers highly accurate results within minutes while eliminating complex, hands-on operator requirements found with other point-of-care technologies.
Diabetes is the fourth leading cause of death by disease in the world. According to the International Diabetes Federation, the number of people diagnosed with Type 2 diabetes worldwide has increased from 30 million to more than 246 million over the last two decades. This figure is projected to increase further to 380 million by 2025. In the USA, 20.8 million people (7 percent of the population) have the disease with a further 54 million considered to be pre-diabetic, according to the Centers for Disease Control and Prevention (CDC) and the American Diabetes Association (ADA).
The ADA strongly recommends that patients be tested for HbA1c at least 4 times a year. Testing should also be performed during treatment changes or after periods of elevated blood glucose levels. The ADA added a recommendation for point-of-care HbA1c monitoring to their 2006 professional practice guidelines emphasizing the importance of routine real-time HbA1c monitoring of persons with diabetes.
Commenting on the news, Brendan Farrell, CEO, said, "FDA Clearance of the TRIstat(TM) product is an extremely exciting development for Trinity Biotech and enables us to compete in the high growth point-of-care segment of the HbA1c market. As TRIstat(TM) provides test results within 10 minutes, patients will know their HbA1c status during their visit to their physician or diabetic clinic. This will assist the physician and patient to make immediate diabetes management decisions and ultimately improve outcomes."
"Our Ultra2(TM)and PDQ(TM) systems already address the $700m clinical lab market for HbA1c and the launch of TRIstat(TM) means that Trinity will now have a presence in the point-of-care segment which is estimated at $300m world wide. Discussions with distribution partners in the USA and Europe are well advanced and will be concluded shortly."
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Contact :
Trinity Biotech plc
Brendan Farrell
CEO
(353)-1-2769800
E-mail: Email Contact
Source: Marketwire (November 29, 2007 - 8:03 AM EST)
News by QuoteMedia
www.quotemedia.com
noch jemand investiert?, ich bin bei 8,40 raus und hab keine News mehr gefunden warum der Kurs jetzt so einknickt.
Ich überlege ob das Kaufkurse sind oder ob es noch weiter geht.
Ich überlege ob das Kaufkurse sind oder ob es noch weiter geht.
Antwort auf Beitrag Nr.: 32.735.400 von girokonto am 12.12.07 09:42:28
Hey ist aber schon 5 Tage alt.
Trinity Biotech Cuts Jobs, Products
Friday December 7, 12:52 pm ET
Trinity Biotech to Cut Work Force, Close Manufacturing Plant in Major Restructuring
NEW YORK (AP) -- Trinity Biotech PLC, a developer and manufacturer of diagnostic products, said Friday it is reducing its work force to save $5 million annually, shuttering its Swedish manufacturing plant and discontinuing products as part of a major restructuring plan.
ADVERTISEMENT
The Irish company said the restructuring plan will reduce complexities in the business and allow it to target research and development.
As a result of the restructuring, the company will incur a one-time write-off of about $39.5 million before taxes in the fourth quarter. The company said except for about $2 million for redundancy costs and the closure of the Swedish plant, the write-off has no cash impact.
Trinity decided to discontinue 106 hemostasis and 69 infectious-disease products. The company offers hemostasis test kits for the detection of blood disorders. The company said the halting of the hemostasis products will be negligible on revenue. Eliminating the infectious-disease products will have less than a 1 percent impact on the company's revenue, Trinity said.
In addition, Trinity is suspending development of a number of ongoing projects, the two most significant being an HIV over-the-counter product and the development of an HIV confirmatory test. The project suspensions will allow the company to focus on a small number of key projects in each division, Trinity said.
Meanwhile, the closing of the plant in Umea, Sweden, will result in an overall net savings of about $500,000 a year, Trinity said. The company will benefit from some of these savings in 2008, with the full impact in 2009.
The company is also recognizing an impairment of about $20.2 million against its intangible assets, primarily goodwill.
Trinity did not disclose in its press release the number of positions it is cutting and could not be reached by the Associated Press.
Shares of Trinity fell 30 cents, or 3.6 percent, to $8.15 in afternoon trading. The stock has ranged between $7.93 and $11.85 over the past year.
Bemerkung von mir ... die Restrekturierung ist ok ,was ich nicht verstehen ist die Reaktion an der NASDAQ.
Der PS sollte zumindestens halten und nicht fallen.
Hummmm vielleicht Leerverkäufer?
cheers
Hey ist aber schon 5 Tage alt.
Trinity Biotech Cuts Jobs, Products
Friday December 7, 12:52 pm ET
Trinity Biotech to Cut Work Force, Close Manufacturing Plant in Major Restructuring
NEW YORK (AP) -- Trinity Biotech PLC, a developer and manufacturer of diagnostic products, said Friday it is reducing its work force to save $5 million annually, shuttering its Swedish manufacturing plant and discontinuing products as part of a major restructuring plan.
ADVERTISEMENT
The Irish company said the restructuring plan will reduce complexities in the business and allow it to target research and development.
As a result of the restructuring, the company will incur a one-time write-off of about $39.5 million before taxes in the fourth quarter. The company said except for about $2 million for redundancy costs and the closure of the Swedish plant, the write-off has no cash impact.
Trinity decided to discontinue 106 hemostasis and 69 infectious-disease products. The company offers hemostasis test kits for the detection of blood disorders. The company said the halting of the hemostasis products will be negligible on revenue. Eliminating the infectious-disease products will have less than a 1 percent impact on the company's revenue, Trinity said.
In addition, Trinity is suspending development of a number of ongoing projects, the two most significant being an HIV over-the-counter product and the development of an HIV confirmatory test. The project suspensions will allow the company to focus on a small number of key projects in each division, Trinity said.
Meanwhile, the closing of the plant in Umea, Sweden, will result in an overall net savings of about $500,000 a year, Trinity said. The company will benefit from some of these savings in 2008, with the full impact in 2009.
The company is also recognizing an impairment of about $20.2 million against its intangible assets, primarily goodwill.
Trinity did not disclose in its press release the number of positions it is cutting and could not be reached by the Associated Press.
Shares of Trinity fell 30 cents, or 3.6 percent, to $8.15 in afternoon trading. The stock has ranged between $7.93 and $11.85 over the past year.
Bemerkung von mir ... die Restrekturierung ist ok ,was ich nicht verstehen ist die Reaktion an der NASDAQ.
Der PS sollte zumindestens halten und nicht fallen.
Hummmm vielleicht Leerverkäufer?

cheers
Hey!
Der tief Fall war gekonnt.
Ich glaube war ein Institutioneller, dass günstig kaufen wollte und dürft auch ganz normaler verfahren , sehr SMART.
ganz normaler verfahren , sehr SMART.
Nach ein Restrukturierungsmassnahme ein Produkt oder Firma geht nicht nach unten, sondern nach oben, man ist bestrebt effizient zu werden, also im Sinne der Aktionären.
cheers
Der tief Fall war gekonnt.
Ich glaube war ein Institutioneller, dass günstig kaufen wollte und dürft auch
 ganz normaler verfahren , sehr SMART.
ganz normaler verfahren , sehr SMART.Nach ein Restrukturierungsmassnahme ein Produkt oder Firma geht nicht nach unten, sondern nach oben, man ist bestrebt effizient zu werden, also im Sinne der Aktionären.
cheers
Hallo.
Anscheinend sind alle mit dem tiefen Fall aus dieser Aktie ausgestiegen.
Falls doch noch jemand da ist, gibt es eine Erklärung für die Kurshalbierung in den letzten 3 Monaten?
Grüße
Lubus104
Anscheinend sind alle mit dem tiefen Fall aus dieser Aktie ausgestiegen.
Falls doch noch jemand da ist, gibt es eine Erklärung für die Kurshalbierung in den letzten 3 Monaten?
Grüße
Lubus104
Antwort auf Beitrag Nr.: 32.826.421 von Lubus104 am 20.12.07 14:40:57Nein, nicht alle.
Keep cool, von diesem Kurs aus gesehen wird 2008 ein gutes Jahr
Keep cool, von diesem Kurs aus gesehen wird 2008 ein gutes Jahr

January 24, 2008 - 8:00 AM EST
Trinity Biotech Announces the Launch of New Lyme Products for the European Market
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced the launch of its new Lyme Enzyme Immunoassay (EIA) products specifically designed for the European market.
The new products, the Trinity Biotech EU Lyme IgG + VlsE EIA and the EU Lyme IgM EIA are designed for use as screening assays in clinical laboratories. They are qualitative serological tests for the detection of IgG and IgM antibodies to the most prevalent and clinically important Borrelia species in Europe, i.e. Borrerlia afzelii, Borrelia garinii, and Borrelia burgdorferi. The addition of VlsE to the product significantly boosts sensitivity of the assay. These new products complete Trinity Biotech's product offering for Lyme Disease in Europe, complementing the existing EU Lyme Western Blot confirmatory assays.
Borrelia species cause Lyme Disease in humans following transmission via a tick bite. Lyme Disease is the most common tick-borne disease in the EU. The most frequent symptoms include skin rash, diffuse flu-like reaction and joint pains. The characteristic rash is not always present making diagnosis more difficult. If untreated or inadequately treated, neurological, cardiac, or serious joint abnormalities may follow.
Eurosurveillance (www.eurosurveillance.org) reports that the incidence of Lyme Disease is on the increase across Europe with particularly high increases seen in Northern and Eastern Europe. The continued expansion of this market segment is driven by factors that increase exposure to ticks. Greater participation in outdoor activities such as hiking, hunting and fishing and recent milder winters have contributed to the proliferation of tick numbers with a consequent increased incidence of Lyme Disease.
"Due to the diverse nature of clinical symptoms, diagnosis of Lyme Disease is difficult, thus making confirmation with a diagnostic assay always advisable. The excellent performance of Trinity's new EIAs in combination with the existing EU Lyme Western Blot products will provide invaluable information to clinicians to aid diagnosis," commented Brendan K. Farrell CEO of Trinity Biotech. "Trinity Biotech is the global leader in Lyme Disease diagnostics. The launch of our new EU Lyme EIA assays extends our product offering to European customers. Trinity now offers both screening and confirmatory assays to all markets, thus reinforcing its number one position for Lyme detection in the world."
Contact :
Trinity Biotech plc
Brendan Farrell
CEO
(353)-1-2769800
E-mail: Email Contact
Trinity Biotech Announces the Launch of New Lyme Products for the European Market
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced the launch of its new Lyme Enzyme Immunoassay (EIA) products specifically designed for the European market.
The new products, the Trinity Biotech EU Lyme IgG + VlsE EIA and the EU Lyme IgM EIA are designed for use as screening assays in clinical laboratories. They are qualitative serological tests for the detection of IgG and IgM antibodies to the most prevalent and clinically important Borrelia species in Europe, i.e. Borrerlia afzelii, Borrelia garinii, and Borrelia burgdorferi. The addition of VlsE to the product significantly boosts sensitivity of the assay. These new products complete Trinity Biotech's product offering for Lyme Disease in Europe, complementing the existing EU Lyme Western Blot confirmatory assays.
Borrelia species cause Lyme Disease in humans following transmission via a tick bite. Lyme Disease is the most common tick-borne disease in the EU. The most frequent symptoms include skin rash, diffuse flu-like reaction and joint pains. The characteristic rash is not always present making diagnosis more difficult. If untreated or inadequately treated, neurological, cardiac, or serious joint abnormalities may follow.
Eurosurveillance (www.eurosurveillance.org) reports that the incidence of Lyme Disease is on the increase across Europe with particularly high increases seen in Northern and Eastern Europe. The continued expansion of this market segment is driven by factors that increase exposure to ticks. Greater participation in outdoor activities such as hiking, hunting and fishing and recent milder winters have contributed to the proliferation of tick numbers with a consequent increased incidence of Lyme Disease.
"Due to the diverse nature of clinical symptoms, diagnosis of Lyme Disease is difficult, thus making confirmation with a diagnostic assay always advisable. The excellent performance of Trinity's new EIAs in combination with the existing EU Lyme Western Blot products will provide invaluable information to clinicians to aid diagnosis," commented Brendan K. Farrell CEO of Trinity Biotech. "Trinity Biotech is the global leader in Lyme Disease diagnostics. The launch of our new EU Lyme EIA assays extends our product offering to European customers. Trinity now offers both screening and confirmatory assays to all markets, thus reinforcing its number one position for Lyme detection in the world."
Contact :
Trinity Biotech plc
Brendan Farrell
CEO
(353)-1-2769800
E-mail: Email Contact
Hey !
Woher kommt so ein bewegung am heutigen Tag?

NEWS? Positives NEWS?
cheers
Woher kommt so ein bewegung am heutigen Tag?


NEWS? Positives NEWS?
cheers
Antwort auf Beitrag Nr.: 33.399.388 von serafino am 18.02.08 11:47:47wo bitte siehst Du eine Bewegung?
Antwort auf Beitrag Nr.: 33.399.482 von Lubus104 am 18.02.08 11:56:52hat jemand Informationen oder eine Erklärung für den rasanten Kursverfall von Trinity?
Antwort auf Beitrag Nr.: 33.513.780 von Lubus104 am 29.02.08 11:58:38ich find kein richtigen Grund, ich wollte evtl. wieder einsteigen, aber im Moment ist es ja ein Fass ohne Boden. Bin heil froh bei 8,40 dochmal Gewinn realisiert zu haben
Antwort auf Beitrag Nr.: 33.528.169 von girokonto am 02.03.08 15:57:21geht mir zum Glück ähnlich, wobei ich eine kleine Posi behalten habe. Vielleicht gab es mal wieder eine Kapitalerhöhung von der die breite Öffentlichkeit nichts weiss.
March 14, 2008 - 11:00 AM Eastern
Fourth Quarter 2007 Financial Results Call
Trinity Biotech Announces Quarter 4 Results
Operating Profits Increase 77%
Mar 14, 2008 -- Trinity Biotech plc (NasdaqGS: TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended December 31, 2007.
Revenues for 2007 increased 21% to US$143.6m compared to US$118.7m in 2006, in part reflecting the acquisition of the haemostasis product line from bioMerieux in June 2006. Operating profit (before the once-off charges associated with the restructuring, inventory write off and impairment) for the same period increased to US$10.6m from US$7.7m, representing an increase of 37%. EBITDA and share option expense for the year increased from US$15.0m in 2006 to US$19.8m in 2007, before the impact of once-off charges.
Revenues for the year by key product area were as follows:
2006 2007
US$000 US$000 % Increase
Clinical Laboratory 103,395 119,113 15.2%
Point of Care 15,279 24,504 60.4%
Total 118,674 143,617 21.0%
Revenues for the year by geographic location were as follows:
2006 2007
US$000 US$000 % Increase
Americas 60,748 68,481 12.7%
Europe 34,452 43,630 26.6%
Asia / Africa 23,474 31,506 34.2%
Total 118,674 143,617 21.0%
Revenues for the quarter were US$35.7m compared to US$33.7m in quarter 3, 2007, an increase of 6% quarter on quarter. Excluding the impact of once-off charges, operating profit for the quarter was US$2.3m which represents an increase of 77% over the previous quarter's operating profit of US$1.3m. On a similar basis, profit before tax more than doubled to US$1.5m when compared to the quarter 3, 2007 profit before tax of US$0.6m.
Gross profit before once-off charges for the quarter amounted to US$17.3m representing a gross margin of 48.3%. This compares favourably to the gross margin of 46.8% for the same period in 2006 and 45.3% for quarter 3, 2007.
Research and development expenditure remains at approximately 5% of revenues whilst the increase in selling, general and administrative expenses from US$12.5m in quarter 4, 2006 to US$13.3m in the current quarter is primarily attributable to the impact of foreign exchange movements and higher professional fees associated with the first year of compliance with the full provisions of the Sarbanes Oxley Act.
Group restructuring and impairment -- quarter 4, 2007 once-off charges
As announced on December 7, 2007 a charge arising from the restructuring of the Group's activities and the impairment of goodwill, amounting to US$39.9m, was recognized in the quarter. This consists of the following items:
US$ million
Inventory write-off - instrumentation 7.5
Inventory write-off - non-instrument related 4.2
Intangible assets write-off 6.7
Closure costs - Sweden manufacturing facility 0.7
Redundancy costs 1.7
Impairment of goodwill 19.1
Total 39.9*
* before the impact of taxation
The cash elements of the above charge amount to US$2.2m and consist of the redundancy costs and certain costs associated with the closure of the group's manufacturing facility in Sweden.
The impairment of goodwill was triggered largely as a consequence of the market capitalisation of the Company being below the carrying amount of its net assets.
In addition to the above, the Company recognized a tax credit of US$1.6m directly associated with the restructuring. This was offset by a once-off charge of US$3.8m due to the derecognition of deferred tax assets previously recognized, which primarily arose on tax losses carried forward in the Group's US operations. The derecognition of these deferred tax assets was considered appropriate in the light of the increased tax losses caused by the restructuring and uncertainty over the timing of the utilisation of the tax losses. Excluding the impact of the restructuring and the derecognition of the deferred tax assets, the tax charge would have been US$1.1m for the year and US$0.3m for the quarter giving an effective tax rate of 14% and 20% respectively.
Earnings per ADR
The loss per ADR was US$1.86 for the year with a loss per ADR of US$2.15 for the quarter. Excluding the impact of the once-off charges associated with restructuring, impairment and the once-off tax amounts mentioned above the earnings per ADR would be 35.6 US cents for the year and 6.4 US cents for quarter 4. On a similar basis the diluted earnings per ADR would be 34.8 US cents for the year and 6.2 US cents for quarter 4.
Commenting on the results, Kevin Tansley, Chief Financial Officer, said "Quarter 4 represented a strong quarter for the Company. Revenues increased by 6% over the previous quarter and more importantly we achieved a 77% growth in operating profits. In addition, EBITDA and share option expense for the quarter increased from US$3.7m to US$4.5m in the period. The growth in profit is particularly satisfying given the adverse currency movements during the period and higher professional fees associated with the implementation of Sarbanes Oxley."
Brendan Farrell, CEO, commented, "We are pleased with our continued growth in revenues year on year. In particular our point of care revenues have significantly outperformed industry growth rates. We are also pleased with the progress made implementing our group restructuring plan announced in late 2007. To date all elements of this plan are on schedule and expected to be completed within the timeframe envisaged.
We believe that Trinity Biotech is very well positioned to achieve strong growth in the coming quarters during which time we will see the impact of the launch of our Tri-stat point of care HbA1c product, our HIV Incidence Assay, our new product for the detection of neo-natal haemoglobin variant analysis and our Destiny Max haemostasis analyzer."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Income Statements
(US$000's except
share data) Three Months Three Months Year Year
Ended Ended Ended Ended
December 31, December 31, December 31, December 31,
2007 2006 2007 2006
(unaudited) (unaudited) (unaudited) (audited)
Revenues 35,725 33,328 143,617 118,674
Cost of sales (18,423) (17,703) (75,572) (62,001)
Cost of sales -
restructuring expenses (12,725) - (12,725) -
Cost of sales -
inventory write off - - - (5,800)
Cost of sales - share
based payments (18) (17) (71) (89)
----------- ----------- ----------- -----------
Gross profit 4,559 15,608 55,249 50,784
Gross profit before
restructuring expenses
& inventory write off 17,284 15,608 67,974 56,584
Other operating income 157 115 413 275
Research & development
expenses (1,667) (1,964) (6,761) (6,660)
Selling, general and
administrative
expenses (13,272) (12,503) (49,719) (41,406)
Restructuring expenses
& goodwill impairment (27,222) - (27,222) -
Indirect share based
payments (226) (136) (1,332) (1,052)
----------- ----------- ----------- -----------
Operating (loss)/
profit (37,671) 1,120 (29,372) 1,941
Operating profit before
restructuring
expenses, goodwill
impairment &
inventory write off 2,276 1,120 10,575 7,741
Financial income 57 368 457 1,164
Financial expenses (821) (929) (3,148) (2,653)
----------- ----------- ----------- -----------
Net financing costs (764) (561) (2,691) (1,489)
----------- ----------- ----------- -----------
(Loss)/ profit before
tax (38,435) 559 (32,063) 452
(Loss)/ profit before
tax before
restructuring
expenses, goodwill
impairment &
inventory write off 1,512 559 7,884 6,252
Income tax (expense) /
credit (2,494) 1,038 (3,309) 2,824
----------- ----------- ----------- -----------
(Loss)/ profit for the
period (40,929) 1,597 (35,372) 3,276
=========== =========== =========== ===========
Earnings per ADR (US
cents) (215.0) 8.5 (186.1) 18.5
Diluted earnings per
ADR (US cents) (215.0) 8.5 (186.1) 18.5
Weighted average no.
of ADR shares used in
computing earnings per
share 19,037,989 18,736,751 19,009,145 17,673,438
The above financial information has been prepared in accordance with
International Financial Reporting Standards and the Company’s accounting
policies but does not constitute full statutory financial statements of the
Company.
Trinity Biotech plc
Consolidated Balance Sheets
December December
31, 2007 31, 2006
US$ '000 US$ '000
(unaudited) (audited)
ASSETS
Non-current assets
Property, plant and equipment 26,409 22,255
Goodwill and intangible assets 104,928 121,768
Deferred tax assets 3,937 7,656
Other assets 896 515
----------- -----------
Total non-current assets 136,170 152,194
----------- -----------
Current assets
Inventories 44,420 45,572
Trade and other receivables 25,683 32,676
Income tax receivable 782 368
Derivative financial instruments 224 -
Financial assets - restricted cash - 15,500
Cash and cash equivalents 8,700 2,821
----------- -----------
Total current assets 79,809 96,937
----------- -----------
----------- -----------
TOTAL ASSETS 215,979 249,131
=========== ===========
EQUITY AND LIABILITIES
Equity attributable to the equity holders of the
parent
Share capital 991 978
Share premium 153,961 151,774
Retained earnings (22,908) 10,818
Translation reserve 797 (275)
Other reserves 4,004 3,967
----------- -----------
Total equity 136,845 167,262
----------- -----------
Current liabilities
Interest-bearing loans and borrowings 15,821 10,382
Convertible notes - interest bearing - 1,836
Income tax payable 86 44
Trade and other payables 24,779 20,459
Other financial liabilities 2,725 3,120
Provisions 100 100
----------- -----------
Total current liabilities 43,511 35,941
----------- -----------
Non-current liabilities
Interest-bearing loans and borrowings 26,312 33,076
Other financial liabilities - 2,568
Other payables 74 838
Deferred tax liabilities 9,237 9,446
----------- -----------
Total non-current liabilities 35,623 45,928
----------- -----------
----------- -----------
TOTAL LIABILITIES 79,134 81,869
----------- -----------
----------- -----------
TOTAL EQUITY AND LIABILITIES 215,979 249,131
=========== ===========
The above financial information has been prepared in accordance with
International Financial Reporting Standards and the Company’s accounting
policies but does not constitute full statutory financial statements of the
Company.
Fourth Quarter 2007 Financial Results Call
Trinity Biotech Announces Quarter 4 Results
Operating Profits Increase 77%
Mar 14, 2008 -- Trinity Biotech plc (NasdaqGS: TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended December 31, 2007.
Revenues for 2007 increased 21% to US$143.6m compared to US$118.7m in 2006, in part reflecting the acquisition of the haemostasis product line from bioMerieux in June 2006. Operating profit (before the once-off charges associated with the restructuring, inventory write off and impairment) for the same period increased to US$10.6m from US$7.7m, representing an increase of 37%. EBITDA and share option expense for the year increased from US$15.0m in 2006 to US$19.8m in 2007, before the impact of once-off charges.
Revenues for the year by key product area were as follows:
2006 2007
US$000 US$000 % Increase
Clinical Laboratory 103,395 119,113 15.2%
Point of Care 15,279 24,504 60.4%
Total 118,674 143,617 21.0%
Revenues for the year by geographic location were as follows:
2006 2007
US$000 US$000 % Increase
Americas 60,748 68,481 12.7%
Europe 34,452 43,630 26.6%
Asia / Africa 23,474 31,506 34.2%
Total 118,674 143,617 21.0%
Revenues for the quarter were US$35.7m compared to US$33.7m in quarter 3, 2007, an increase of 6% quarter on quarter. Excluding the impact of once-off charges, operating profit for the quarter was US$2.3m which represents an increase of 77% over the previous quarter's operating profit of US$1.3m. On a similar basis, profit before tax more than doubled to US$1.5m when compared to the quarter 3, 2007 profit before tax of US$0.6m.
Gross profit before once-off charges for the quarter amounted to US$17.3m representing a gross margin of 48.3%. This compares favourably to the gross margin of 46.8% for the same period in 2006 and 45.3% for quarter 3, 2007.
Research and development expenditure remains at approximately 5% of revenues whilst the increase in selling, general and administrative expenses from US$12.5m in quarter 4, 2006 to US$13.3m in the current quarter is primarily attributable to the impact of foreign exchange movements and higher professional fees associated with the first year of compliance with the full provisions of the Sarbanes Oxley Act.
Group restructuring and impairment -- quarter 4, 2007 once-off charges
As announced on December 7, 2007 a charge arising from the restructuring of the Group's activities and the impairment of goodwill, amounting to US$39.9m, was recognized in the quarter. This consists of the following items:
US$ million
Inventory write-off - instrumentation 7.5
Inventory write-off - non-instrument related 4.2
Intangible assets write-off 6.7
Closure costs - Sweden manufacturing facility 0.7
Redundancy costs 1.7
Impairment of goodwill 19.1
Total 39.9*
* before the impact of taxation
The cash elements of the above charge amount to US$2.2m and consist of the redundancy costs and certain costs associated with the closure of the group's manufacturing facility in Sweden.
The impairment of goodwill was triggered largely as a consequence of the market capitalisation of the Company being below the carrying amount of its net assets.
In addition to the above, the Company recognized a tax credit of US$1.6m directly associated with the restructuring. This was offset by a once-off charge of US$3.8m due to the derecognition of deferred tax assets previously recognized, which primarily arose on tax losses carried forward in the Group's US operations. The derecognition of these deferred tax assets was considered appropriate in the light of the increased tax losses caused by the restructuring and uncertainty over the timing of the utilisation of the tax losses. Excluding the impact of the restructuring and the derecognition of the deferred tax assets, the tax charge would have been US$1.1m for the year and US$0.3m for the quarter giving an effective tax rate of 14% and 20% respectively.
Earnings per ADR
The loss per ADR was US$1.86 for the year with a loss per ADR of US$2.15 for the quarter. Excluding the impact of the once-off charges associated with restructuring, impairment and the once-off tax amounts mentioned above the earnings per ADR would be 35.6 US cents for the year and 6.4 US cents for quarter 4. On a similar basis the diluted earnings per ADR would be 34.8 US cents for the year and 6.2 US cents for quarter 4.
Commenting on the results, Kevin Tansley, Chief Financial Officer, said "Quarter 4 represented a strong quarter for the Company. Revenues increased by 6% over the previous quarter and more importantly we achieved a 77% growth in operating profits. In addition, EBITDA and share option expense for the quarter increased from US$3.7m to US$4.5m in the period. The growth in profit is particularly satisfying given the adverse currency movements during the period and higher professional fees associated with the implementation of Sarbanes Oxley."
Brendan Farrell, CEO, commented, "We are pleased with our continued growth in revenues year on year. In particular our point of care revenues have significantly outperformed industry growth rates. We are also pleased with the progress made implementing our group restructuring plan announced in late 2007. To date all elements of this plan are on schedule and expected to be completed within the timeframe envisaged.
We believe that Trinity Biotech is very well positioned to achieve strong growth in the coming quarters during which time we will see the impact of the launch of our Tri-stat point of care HbA1c product, our HIV Incidence Assay, our new product for the detection of neo-natal haemoglobin variant analysis and our Destiny Max haemostasis analyzer."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Trinity Biotech plc
Consolidated Income Statements
(US$000's except
share data) Three Months Three Months Year Year
Ended Ended Ended Ended
December 31, December 31, December 31, December 31,
2007 2006 2007 2006
(unaudited) (unaudited) (unaudited) (audited)
Revenues 35,725 33,328 143,617 118,674
Cost of sales (18,423) (17,703) (75,572) (62,001)
Cost of sales -
restructuring expenses (12,725) - (12,725) -
Cost of sales -
inventory write off - - - (5,800)
Cost of sales - share
based payments (18) (17) (71) (89)
----------- ----------- ----------- -----------
Gross profit 4,559 15,608 55,249 50,784
Gross profit before
restructuring expenses
& inventory write off 17,284 15,608 67,974 56,584
Other operating income 157 115 413 275
Research & development
expenses (1,667) (1,964) (6,761) (6,660)
Selling, general and
administrative
expenses (13,272) (12,503) (49,719) (41,406)
Restructuring expenses
& goodwill impairment (27,222) - (27,222) -
Indirect share based
payments (226) (136) (1,332) (1,052)
----------- ----------- ----------- -----------
Operating (loss)/
profit (37,671) 1,120 (29,372) 1,941
Operating profit before
restructuring
expenses, goodwill
impairment &
inventory write off 2,276 1,120 10,575 7,741
Financial income 57 368 457 1,164
Financial expenses (821) (929) (3,148) (2,653)
----------- ----------- ----------- -----------
Net financing costs (764) (561) (2,691) (1,489)
----------- ----------- ----------- -----------
(Loss)/ profit before
tax (38,435) 559 (32,063) 452
(Loss)/ profit before
tax before
restructuring
expenses, goodwill
impairment &
inventory write off 1,512 559 7,884 6,252
Income tax (expense) /
credit (2,494) 1,038 (3,309) 2,824
----------- ----------- ----------- -----------
(Loss)/ profit for the
period (40,929) 1,597 (35,372) 3,276
=========== =========== =========== ===========
Earnings per ADR (US
cents) (215.0) 8.5 (186.1) 18.5
Diluted earnings per
ADR (US cents) (215.0) 8.5 (186.1) 18.5
Weighted average no.
of ADR shares used in
computing earnings per
share 19,037,989 18,736,751 19,009,145 17,673,438
The above financial information has been prepared in accordance with
International Financial Reporting Standards and the Company’s accounting
policies but does not constitute full statutory financial statements of the
Company.
Trinity Biotech plc
Consolidated Balance Sheets
December December
31, 2007 31, 2006
US$ '000 US$ '000
(unaudited) (audited)
ASSETS
Non-current assets
Property, plant and equipment 26,409 22,255
Goodwill and intangible assets 104,928 121,768
Deferred tax assets 3,937 7,656
Other assets 896 515
----------- -----------
Total non-current assets 136,170 152,194
----------- -----------
Current assets
Inventories 44,420 45,572
Trade and other receivables 25,683 32,676
Income tax receivable 782 368
Derivative financial instruments 224 -
Financial assets - restricted cash - 15,500
Cash and cash equivalents 8,700 2,821
----------- -----------
Total current assets 79,809 96,937
----------- -----------
----------- -----------
TOTAL ASSETS 215,979 249,131
=========== ===========
EQUITY AND LIABILITIES
Equity attributable to the equity holders of the
parent
Share capital 991 978
Share premium 153,961 151,774
Retained earnings (22,908) 10,818
Translation reserve 797 (275)
Other reserves 4,004 3,967
----------- -----------
Total equity 136,845 167,262
----------- -----------
Current liabilities
Interest-bearing loans and borrowings 15,821 10,382
Convertible notes - interest bearing - 1,836
Income tax payable 86 44
Trade and other payables 24,779 20,459
Other financial liabilities 2,725 3,120
Provisions 100 100
----------- -----------
Total current liabilities 43,511 35,941
----------- -----------
Non-current liabilities
Interest-bearing loans and borrowings 26,312 33,076
Other financial liabilities - 2,568
Other payables 74 838
Deferred tax liabilities 9,237 9,446
----------- -----------
Total non-current liabilities 35,623 45,928
----------- -----------
----------- -----------
TOTAL LIABILITIES 79,134 81,869
----------- -----------
----------- -----------
TOTAL EQUITY AND LIABILITIES 215,979 249,131
=========== ===========
The above financial information has been prepared in accordance with
International Financial Reporting Standards and the Company’s accounting
policies but does not constitute full statutory financial statements of the
Company.
Trinity Executive Chairman Purchases Shares in the Company
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that Executive Chairman, Ronan O'Caoimh, purchased 100,000 ADRs in the Company in the open market.
Ronan O'Caoimh, Executive Chairman of Trinity Biotech, commented, "This purchase reflects the confidence that I have in the future growth prospects and strategic direction of the Company. Furthermore, I will be making further share purchases in the immediate future."
Source: Marketwire (March 20, 2008 - 10:42 AM EDT)
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that Executive Chairman, Ronan O'Caoimh, purchased 100,000 ADRs in the Company in the open market.
Ronan O'Caoimh, Executive Chairman of Trinity Biotech, commented, "This purchase reflects the confidence that I have in the future growth prospects and strategic direction of the Company. Furthermore, I will be making further share purchases in the immediate future."
Source: Marketwire (March 20, 2008 - 10:42 AM EDT)
Trinity Biotech Announces the Launch of New GeneSys(TM) Neonatal Haemoglobin Variant Screen
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets and the market leader in adult haemoglobinopathy detection, is pleased to announce the introduction of its GeneSys(TM) platform for infant haemoglobinopathies. The GeneSys(TM) system will be the first on the market to offer a fully automated approach for newborn screening while providing positive identification for all major infant haemoglobin variants. The system also offers the unique time saving capability of allowing laboratories to run a confirmatory test on the same instrument for all positive results.
In the U.S. under Federal Law, it is mandatory to screen all newborns for haemoglobinopathies. As a result, over 4 million newborns in the U.S. each year are tested for genetic blood disorders, some of which have significant long term clinical implications. Demand is currently served by a small number of market participants. Trinity will market the GeneSys(TM) system to the 38 U.S. State laboratories that participate in the national neonatal screening program immediately upon its release.
"The Trinity GeneSys(TM) platform provides a number of competitive advantages to the alternative products currently used by U.S. State laboratories. It is the only platform to provide truly automated sample handling. It provides screening results in two minutes and it enables confirmatory tests to be run on the same instrument," commented Brendan K. Farrell CEO of Trinity Biotech. "Trinity Biotech is particularly pleased to bring our market leading technology from the adult haemoglobinopathy market to the neonatal market. We believe that the product will prove an immediate success and this week are shipping our first two instruments to a State laboratory."
Source: Marketwire (April 2, 2008 - 12:00 PM EDT)
Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets and the market leader in adult haemoglobinopathy detection, is pleased to announce the introduction of its GeneSys(TM) platform for infant haemoglobinopathies. The GeneSys(TM) system will be the first on the market to offer a fully automated approach for newborn screening while providing positive identification for all major infant haemoglobin variants. The system also offers the unique time saving capability of allowing laboratories to run a confirmatory test on the same instrument for all positive results.
In the U.S. under Federal Law, it is mandatory to screen all newborns for haemoglobinopathies. As a result, over 4 million newborns in the U.S. each year are tested for genetic blood disorders, some of which have significant long term clinical implications. Demand is currently served by a small number of market participants. Trinity will market the GeneSys(TM) system to the 38 U.S. State laboratories that participate in the national neonatal screening program immediately upon its release.
"The Trinity GeneSys(TM) platform provides a number of competitive advantages to the alternative products currently used by U.S. State laboratories. It is the only platform to provide truly automated sample handling. It provides screening results in two minutes and it enables confirmatory tests to be run on the same instrument," commented Brendan K. Farrell CEO of Trinity Biotech. "Trinity Biotech is particularly pleased to bring our market leading technology from the adult haemoglobinopathy market to the neonatal market. We believe that the product will prove an immediate success and this week are shipping our first two instruments to a State laboratory."
Source: Marketwire (April 2, 2008 - 12:00 PM EDT)
Gerade gefunden.Vielleicht interessiert es noch jemanden, ansonsten ist es hier ja ziemlich tot.


Trinity Biotech downgraded to "reduce"
03/27/08 - Goodbody Stockbrokers
NEW YORK, March 27 (newratings.com) - Analyst Ian Hunter of Goodbody Stockbrokers downgrades Trinity Biotech Plc (TRIB) from "add" to "reduce," while reducing his estimates for the company. The target price is set to $4.50.
In a research note published this morning, the analyst mentions that the company has reported its 4Q07 results marginally short of expectations. Although Trinity Biotech delivered robust growth in FY07, it has provided a cautious guidance for FY08 due to low visibility into its Point-of-Care revenues. The EPS estimates for FY08 and FY09 have been reduced by 21% to 34.5¢ and by 16% to 42.6¢, respectively.


Trinity Biotech downgraded to "reduce"
03/27/08 - Goodbody Stockbrokers
NEW YORK, March 27 (newratings.com) - Analyst Ian Hunter of Goodbody Stockbrokers downgrades Trinity Biotech Plc (TRIB) from "add" to "reduce," while reducing his estimates for the company. The target price is set to $4.50.
In a research note published this morning, the analyst mentions that the company has reported its 4Q07 results marginally short of expectations. Although Trinity Biotech delivered robust growth in FY07, it has provided a cautious guidance for FY08 due to low visibility into its Point-of-Care revenues. The EPS estimates for FY08 and FY09 have been reduced by 21% to 34.5¢ and by 16% to 42.6¢, respectively.
Und noch eine " schöne" Nachricht. Die geben Aktien aus, wie die FED US-$ druckt. 
Next Dow Jones Article
Trinity Biotech To Place $7.1 Million ADS4-8-08 9:56 AM EDT | E-mail Article | Print ArticleEdited Press Release
LONDON -(Dow Jones)- Trinity Biotech announced Tuesday that it has obtained commitments from a select group of institutional investors in the USA to purchase approximately 1.8 million American Depositary Shares at a price of $ 3.92 per ADS in a registered public offering.
This represents a discount of 15% to the closing price on NASDAQ on April 7, 2008.
Investors also will receive 5 year warrants to purchase an aggregate of approximately 545,000 American Depositary Shares at an exercise price of $5.54 per share, representing a 41% premium to the placement price. These warrants are callable by the Company if the share price exceeds $11.08 for 20 consecutive trading days.
The closing is expected to take place on Friday, April 11, 2008, subject to the satisfaction of customary closing conditions.
Trinity Biotech intends to use these funds for general corporate purposes and the repayment of the final deferred consideration from the 2006 bioMerieux acquisition.

Next Dow Jones Article
Trinity Biotech To Place $7.1 Million ADS4-8-08 9:56 AM EDT | E-mail Article | Print ArticleEdited Press Release
LONDON -(Dow Jones)- Trinity Biotech announced Tuesday that it has obtained commitments from a select group of institutional investors in the USA to purchase approximately 1.8 million American Depositary Shares at a price of $ 3.92 per ADS in a registered public offering.
This represents a discount of 15% to the closing price on NASDAQ on April 7, 2008.
Investors also will receive 5 year warrants to purchase an aggregate of approximately 545,000 American Depositary Shares at an exercise price of $5.54 per share, representing a 41% premium to the placement price. These warrants are callable by the Company if the share price exceeds $11.08 for 20 consecutive trading days.
The closing is expected to take place on Friday, April 11, 2008, subject to the satisfaction of customary closing conditions.
Trinity Biotech intends to use these funds for general corporate purposes and the repayment of the final deferred consideration from the 2006 bioMerieux acquisition.
Hallo hallo,
bin mal just dazu gestossen und wuerde gerne mal wissen ob noch wer hier vetreten ist ?
Habe mich ein wenig informiert und denke das hier noch was geht.
Resultate des 1. quartals sind ok ( denke ich ) und auf dem allzeit chart ist definitiv bei diesem Preis die unterstuetzung vertreten.
Hoffe das sich eventuell mal jemand meldet und hier wieder etwas leben rein kommt.
n01
bin mal just dazu gestossen und wuerde gerne mal wissen ob noch wer hier vetreten ist ?
Habe mich ein wenig informiert und denke das hier noch was geht.
Resultate des 1. quartals sind ok ( denke ich ) und auf dem allzeit chart ist definitiv bei diesem Preis die unterstuetzung vertreten.
Hoffe das sich eventuell mal jemand meldet und hier wieder etwas leben rein kommt.
n01
Tach,
desweiteren denke ich das die Aktien zum Preis von 3.92 zeigen das hier noch was moeglich sein muss.
Auch wenn die weitere Aktien ausgeben und es verwaessert wird heisst es dennoch jemand ist interessiert und ist einen Gedanken weiter.
Leider habe ich jetzt nach 2 Tagen intensiver Suche im Netz nicht allzuviel ueber die Firma rausbekommen und meist ist es eh alter Kram.
Dennoch bin ich ich sehr ueberzeugt das hier was gehen muss. Die gestrige Stueckzahl in USA zeigt auch das leben drinn ist in dieser Aktie und nicht nur ein paar Zocker am Werk sind.
Die meisten news sind ja nunmal etwas aelter und auch nicht wirklich aussage kraeftig in bezug auf die firma ( besides Q1 results ) und somit denke ich das mit der platzierung und der momentanen stille auf kurze sicht was ansteht. CEO hat definitiv ein zeichen gegeben mit dem erwerb seiner mehr als 250 tausend stueck erworbenen Aktien.
Jemand ne meinung zum ganzen ?
Wuerd mich mal freuen was zu lesen hier das zur sache gehoert.
n01
desweiteren denke ich das die Aktien zum Preis von 3.92 zeigen das hier noch was moeglich sein muss.
Auch wenn die weitere Aktien ausgeben und es verwaessert wird heisst es dennoch jemand ist interessiert und ist einen Gedanken weiter.
Leider habe ich jetzt nach 2 Tagen intensiver Suche im Netz nicht allzuviel ueber die Firma rausbekommen und meist ist es eh alter Kram.
Dennoch bin ich ich sehr ueberzeugt das hier was gehen muss. Die gestrige Stueckzahl in USA zeigt auch das leben drinn ist in dieser Aktie und nicht nur ein paar Zocker am Werk sind.
Die meisten news sind ja nunmal etwas aelter und auch nicht wirklich aussage kraeftig in bezug auf die firma ( besides Q1 results ) und somit denke ich das mit der platzierung und der momentanen stille auf kurze sicht was ansteht. CEO hat definitiv ein zeichen gegeben mit dem erwerb seiner mehr als 250 tausend stueck erworbenen Aktien.
Jemand ne meinung zum ganzen ?
Wuerd mich mal freuen was zu lesen hier das zur sache gehoert.
n01
Antwort auf Beitrag Nr.: 34.115.406 von Neela01 am 17.05.08 05:17:33Hallo!!
Finde es ja ganz schön, dass noch ein bißchen Interesse an Trinity besteht, allerdings macht das Management es den Anlegern nicht leicht, bei der Stange zu bleiben.
Bin schon ziemlich lange in dieser Firma investiert( mal mehr, mal weniger)und finde die Story und Produkte immer noch ganz gut. Allerdings werden positive Ansätze ( Neuzulassungen und Umsatzsteigerungen) immer wieder durch überproportionale Kostensteigerungen kaputt gemacht.
Und dann diese ständige Kapitalerhöhungen um Liquidität zu schaffen, sind der Tod eines jeden Kurses. Wenn es wenigstens noch einen Investor gäbe, der die Aktien abnimmt, aber nein, immer wieder überdie Börse, das schafft echtkein Vertrauen.Also, esist wirklich nicht zwingend hier investiert zu sein, es wird nix überraschendes geben und das Management muß bei Aktionären erstmal wieder durch Kontinuität Vertrauen gewinnen.
Ciao, schöne WE
Lubus
Finde es ja ganz schön, dass noch ein bißchen Interesse an Trinity besteht, allerdings macht das Management es den Anlegern nicht leicht, bei der Stange zu bleiben.

Bin schon ziemlich lange in dieser Firma investiert( mal mehr, mal weniger)und finde die Story und Produkte immer noch ganz gut. Allerdings werden positive Ansätze ( Neuzulassungen und Umsatzsteigerungen) immer wieder durch überproportionale Kostensteigerungen kaputt gemacht.
Und dann diese ständige Kapitalerhöhungen um Liquidität zu schaffen, sind der Tod eines jeden Kurses. Wenn es wenigstens noch einen Investor gäbe, der die Aktien abnimmt, aber nein, immer wieder überdie Börse, das schafft echtkein Vertrauen.Also, esist wirklich nicht zwingend hier investiert zu sein, es wird nix überraschendes geben und das Management muß bei Aktionären erstmal wieder durch Kontinuität Vertrauen gewinnen.
Ciao, schöne WE
Lubus
Man beachte bitte den Kauf eines Riesenpaketes unmittelbar vor Handelsschluss am gestrigen Freitag. Ich habe mich bereits am Donnerstag platziert. Mal sehen, was passiert.
Antwort auf Beitrag Nr.: 34.117.170 von honorar am 17.05.08 17:01:24Tach auch und hallo,
sorry aber bei dem Riesen paket weisss ich nicht wovon du sprichst.
Auf dem chart den du reinstellst ist das fuer mich die tagesmenge an gehandelten shares und nicht ein riesen paket.
Soweit ich das gesehen habe wurden in US als letztes ca. 30 tausend gehandelt, oder meinst du das mit riesen paket.
Sorry aber vielleicht verstehe ich auch was verkehrt bei dem chart.
Gehandelte menge in US am Freitag ca. 390 tausend und als letztes halt diese ca. 30 tausend.
Nun denn, es freut mich zu sehen das hier noch leute sind und sich kuemmern und nicht nur still mitlesen.
Ich gebe Lubus vollkommen recht mit seiner stellungnahme und denke das eventuell die uebernahme in 2006 dieser franzosen firma, der name faellt mir just nicht ein, vielleicht ueber deren verhaeltnisse war wenn die jetzt wieder shares ausgeben und sagen es sei zur refinanzierung der uebernahme !
Ich persoenlich denke das es wieder auf geht und Q2 gut ausfaellt und die institutionellen anleger dieser shares ja auch was vom kuchen haben wollen, bei einem preis von 3.92.
Sehe ich das recht oder ist bei ca. 3.95 bis 4 dollar nen wiederstand vorhanden, da trib oefters schon jetzt nicht drueber kommt?
Nun denn, ich muss weiterarbeiten und werfe spaeter wieder nen blick rein. Meiner einer ist nicht in Deutschland und bin 6 stunden vorraus.
Neela
sorry aber bei dem Riesen paket weisss ich nicht wovon du sprichst.
Auf dem chart den du reinstellst ist das fuer mich die tagesmenge an gehandelten shares und nicht ein riesen paket.
Soweit ich das gesehen habe wurden in US als letztes ca. 30 tausend gehandelt, oder meinst du das mit riesen paket.
Sorry aber vielleicht verstehe ich auch was verkehrt bei dem chart.
Gehandelte menge in US am Freitag ca. 390 tausend und als letztes halt diese ca. 30 tausend.
Nun denn, es freut mich zu sehen das hier noch leute sind und sich kuemmern und nicht nur still mitlesen.
Ich gebe Lubus vollkommen recht mit seiner stellungnahme und denke das eventuell die uebernahme in 2006 dieser franzosen firma, der name faellt mir just nicht ein, vielleicht ueber deren verhaeltnisse war wenn die jetzt wieder shares ausgeben und sagen es sei zur refinanzierung der uebernahme !
Ich persoenlich denke das es wieder auf geht und Q2 gut ausfaellt und die institutionellen anleger dieser shares ja auch was vom kuchen haben wollen, bei einem preis von 3.92.
Sehe ich das recht oder ist bei ca. 3.95 bis 4 dollar nen wiederstand vorhanden, da trib oefters schon jetzt nicht drueber kommt?
Nun denn, ich muss weiterarbeiten und werfe spaeter wieder nen blick rein. Meiner einer ist nicht in Deutschland und bin 6 stunden vorraus.
Neela
Was ein geiles Bid !
Antwort auf Beitrag Nr.: 34.122.419 von Neela01 am 19.05.08 10:20:59von welchem Bild sprichst Du bitte?

Antwort auf Beitrag Nr.: 34.123.012 von honorar am 19.05.08 11:27:49Hallo,
eine klasse Seite, habe ich noch nie vorher gesehen. Vielen Dank, das hilft mir einiges weiter und einiges wird jetzt schoen deutlich.
Kann ich mir wenigstens die zeit nu nen bissl vetreiben hier.
Danke fuers reinstellen.
Welches Bid ? das von gestern morgen in Fra., die 2.25 meinte ich.
eine klasse Seite, habe ich noch nie vorher gesehen. Vielen Dank, das hilft mir einiges weiter und einiges wird jetzt schoen deutlich.
Kann ich mir wenigstens die zeit nu nen bissl vetreiben hier.
Danke fuers reinstellen.
Welches Bid ? das von gestern morgen in Fra., die 2.25 meinte ich.
Da ich per BM gefragt worden bin, auch hier die Antwort:
Trinity Biotech trades its Ordinary Shares on the Irish Stock Exchange and ADR's on Nasdaq. Each ADR represents four Ordinary Shares. For further information please see the company's website: www.trinitybiotech.com.
Trinity Biotech trades its Ordinary Shares on the Irish Stock Exchange and ADR's on Nasdaq. Each ADR represents four Ordinary Shares. For further information please see the company's website: www.trinitybiotech.com.
American Depositary Receipts (ADR)
Als American Depositary Receipts werden auf Dollar lautende, von US-amerikanischen Depotbanken (depositary banks) in den USA ausgegebene Aktienzertifikate bezeichnet, die eine bestimmte Anzahl hinterlegter Aktien eines ausländischen Unternehmens verkörpern und an ihrer Stelle am US-Kapitalmarkt wie Aktien gehandelt werden. Ein ADR repräsentiert in der Regel einen Bruchteil einer Aktie, kann aber auch einer vollen Aktie entsprechen. Eine Übertragung von ADR erfolgt durch Indossament und Übergabe. Der jeweilige Inhaber eines ADR kann jederzeit gegen Rückübertragung des Zertifikats an die Depotbank die Herausgabe der bei einer ausländischen Hinterlegungsbank (custodian bank), in der Regel der Zweigstelle der Depotbank im Sitzstaat des ausländischen Unternehmens verwahrten Aktien bzw. deren Verkauf an der ausländischen Börse verlangen.
Ein wichtiger Grund für die Konstruktion von ADR-Programmen liegt darin, daß bestimmte amerikanische institutionelle Investoren, wie staatliche Pensionsfonds, Lebensversicherungsgesellschaften oder Kreditinstitute, Beschränkungen hinsichtlich deren Investment in ausländische Wertpapiere unterliegen. Da ADR wie amerikanische Aktien behandelt werden, kann so ohne die Emission von Aktien auf dem US-Kapitalmarkt dieser von ausländischen Unternehmen genutzt werden.
Eine Ausgabe von ADR kann grundsätzlich in der Form von unsponsored oder sponsored ADR-Programmen erfolgen. Bei unsponsored ADR-Programmen geht die Initiative allein von einer amerikanischen Depotbank oder einem Händler aus. Die Kosten für das ADR-Programm sind regelmäßig von den Investoren zu tragen. Darüber hinaus ist die Depotbank aufgrund des fehlenden Depotvertrages mit dem ausländischen Unternehmen nicht verpflichtet, Informationen des Unternehmens an die Investoren weiterzuleiten. Unsponsored ADR-Programme werden an vielen Börsen nicht zum Handel zugelassen, so daß nur geringe praktische Bedeutung zukommt.
Bei sponsored ADR-Programmen geht die Initiative vom Emittenten aus und wird von diesem in Zusammenarbeit mit der Depotbank realisiert. Dabei wird ein Depotvertrag (depositary agreement) geschlossen, der die Depotbank verpflichtet, die Ausgabe und Rücknahme von Zertifikaten, die Ausübung von Stimmrechten durch den US-Investor, die Weitergabe von Dividenden und Unternehmensinformationen und die Programmpflege zu übernehmen. Der Großteil der entstehenden Kosten bei sponsored ADR-Programmen wird von dem emittierenden Unternehmen getragen. Neben einer Privatplazierung gibt es drei verschiedene Ausprägungen für sponsored ADR-Programme:
Level I: Durch ein Level I-Programm kann für bereits existente Aktien eines Unternehmens ein Handel in den USA auf dem Over the Counter (OTC)-Markt initiiert werden. Hiermit kann jedoch weder neues Kapital aufgenommen noch das ADR an einer US-Börse notiert werden.
Ein Level I-Programm muß mit dem Formular Form F-6 bei der SEC gemäß den Bestimmungen des Securities Act (SA) angemeldet werden. Form F-6 verlangt nur Informationen über das ADR-Programm selbst, jedoch nicht über den dahinterstehenden ausländischen Emittenten. Eine Registrierung der hinterlegten Aktien nach dem Securities Exchange Act (SEA) ist aufgrund der Ausnahmeregelung der Rule 12g3-2(b) im Regelfall nicht erforderlich, da die ADR eines Level I-Programms weder an einer US-Börse noch im NASDAQ-System gehandelt werden sollen. Bei der SEC müssen die im Heimatmarkt des Unternehmens veröffentlichten Informationen in einer englischen Übersetzung eingereicht werden. Der bedeutende Vorteil eines Level I-Programms gegenüber höhergradigen Programmen liegt in der fehlenden Verpflichtung zur Rechnungslegung nach US-GAAP.
Level II: Für eine Notierung an einer US-amerikanischen Börse oder im NASDAQ ist mindestens ein Level II-ADR-Programm aufzulegen. Hierbei müssen neben der Form F-6 auch die zugrundeliegenden Aktien nach den Bestimmungen des SEA bei der SEC registriert und die Berichts- und Offenlegungsanforderungen der jeweiligen Börse beachtet werden. Eine Registrierung der Aktien erfolgt bei der SEC mit Form 20-F, das umfangreiche Angaben über den Emittenten verlangt. Es sind Abschlüsse nach US-GAAP vorzulegen, wesentliche Anteilsinhaber zu nennen und Angaben über die Vorstands- u. Aufsichtsratsmitglieder zu machen. Die Registrierung zieht allerdings eine mindestens jährliche Berichtspflicht in vergleichbarem Umfang nach sich.
Level III: Für die Aufnahme von Kapital durch die Emission neuer Aktien im Sitzstaat des Emittenten und den Handel der zugehörigen ADR an einer US-Börse ist ein Level III-Programm erforderlich. Neben den Formblättern Form F-6 und Form 20-F ist das Formblatt F-1 einzureichen; die dort geforderten Pflichten zur Offenlegung und Aufstellung von Jahresabschlüssen nach US-GAAP entsprechen weitgehend denen nach Form 20-F.
Neben den an die Öffentlichkeit gerichteten Level I- bis Level III-Programmen besteht die Möglichkeit einer Privatplazierung nach Rule 144 A, die sich an spezielle institutionelle Käufer (Qualified Institutional Buyers) richtet. Eine Privatplazierung hat den Vorteil, daß kein aufwendiges Registrierungsverfahren notwendig und kein Abschluß nach US-GAAP erforderlich ist.
Als American Depositary Receipts werden auf Dollar lautende, von US-amerikanischen Depotbanken (depositary banks) in den USA ausgegebene Aktienzertifikate bezeichnet, die eine bestimmte Anzahl hinterlegter Aktien eines ausländischen Unternehmens verkörpern und an ihrer Stelle am US-Kapitalmarkt wie Aktien gehandelt werden. Ein ADR repräsentiert in der Regel einen Bruchteil einer Aktie, kann aber auch einer vollen Aktie entsprechen. Eine Übertragung von ADR erfolgt durch Indossament und Übergabe. Der jeweilige Inhaber eines ADR kann jederzeit gegen Rückübertragung des Zertifikats an die Depotbank die Herausgabe der bei einer ausländischen Hinterlegungsbank (custodian bank), in der Regel der Zweigstelle der Depotbank im Sitzstaat des ausländischen Unternehmens verwahrten Aktien bzw. deren Verkauf an der ausländischen Börse verlangen.
Ein wichtiger Grund für die Konstruktion von ADR-Programmen liegt darin, daß bestimmte amerikanische institutionelle Investoren, wie staatliche Pensionsfonds, Lebensversicherungsgesellschaften oder Kreditinstitute, Beschränkungen hinsichtlich deren Investment in ausländische Wertpapiere unterliegen. Da ADR wie amerikanische Aktien behandelt werden, kann so ohne die Emission von Aktien auf dem US-Kapitalmarkt dieser von ausländischen Unternehmen genutzt werden.
Eine Ausgabe von ADR kann grundsätzlich in der Form von unsponsored oder sponsored ADR-Programmen erfolgen. Bei unsponsored ADR-Programmen geht die Initiative allein von einer amerikanischen Depotbank oder einem Händler aus. Die Kosten für das ADR-Programm sind regelmäßig von den Investoren zu tragen. Darüber hinaus ist die Depotbank aufgrund des fehlenden Depotvertrages mit dem ausländischen Unternehmen nicht verpflichtet, Informationen des Unternehmens an die Investoren weiterzuleiten. Unsponsored ADR-Programme werden an vielen Börsen nicht zum Handel zugelassen, so daß nur geringe praktische Bedeutung zukommt.
Bei sponsored ADR-Programmen geht die Initiative vom Emittenten aus und wird von diesem in Zusammenarbeit mit der Depotbank realisiert. Dabei wird ein Depotvertrag (depositary agreement) geschlossen, der die Depotbank verpflichtet, die Ausgabe und Rücknahme von Zertifikaten, die Ausübung von Stimmrechten durch den US-Investor, die Weitergabe von Dividenden und Unternehmensinformationen und die Programmpflege zu übernehmen. Der Großteil der entstehenden Kosten bei sponsored ADR-Programmen wird von dem emittierenden Unternehmen getragen. Neben einer Privatplazierung gibt es drei verschiedene Ausprägungen für sponsored ADR-Programme:
Level I: Durch ein Level I-Programm kann für bereits existente Aktien eines Unternehmens ein Handel in den USA auf dem Over the Counter (OTC)-Markt initiiert werden. Hiermit kann jedoch weder neues Kapital aufgenommen noch das ADR an einer US-Börse notiert werden.
Ein Level I-Programm muß mit dem Formular Form F-6 bei der SEC gemäß den Bestimmungen des Securities Act (SA) angemeldet werden. Form F-6 verlangt nur Informationen über das ADR-Programm selbst, jedoch nicht über den dahinterstehenden ausländischen Emittenten. Eine Registrierung der hinterlegten Aktien nach dem Securities Exchange Act (SEA) ist aufgrund der Ausnahmeregelung der Rule 12g3-2(b) im Regelfall nicht erforderlich, da die ADR eines Level I-Programms weder an einer US-Börse noch im NASDAQ-System gehandelt werden sollen. Bei der SEC müssen die im Heimatmarkt des Unternehmens veröffentlichten Informationen in einer englischen Übersetzung eingereicht werden. Der bedeutende Vorteil eines Level I-Programms gegenüber höhergradigen Programmen liegt in der fehlenden Verpflichtung zur Rechnungslegung nach US-GAAP.
Level II: Für eine Notierung an einer US-amerikanischen Börse oder im NASDAQ ist mindestens ein Level II-ADR-Programm aufzulegen. Hierbei müssen neben der Form F-6 auch die zugrundeliegenden Aktien nach den Bestimmungen des SEA bei der SEC registriert und die Berichts- und Offenlegungsanforderungen der jeweiligen Börse beachtet werden. Eine Registrierung der Aktien erfolgt bei der SEC mit Form 20-F, das umfangreiche Angaben über den Emittenten verlangt. Es sind Abschlüsse nach US-GAAP vorzulegen, wesentliche Anteilsinhaber zu nennen und Angaben über die Vorstands- u. Aufsichtsratsmitglieder zu machen. Die Registrierung zieht allerdings eine mindestens jährliche Berichtspflicht in vergleichbarem Umfang nach sich.
Level III: Für die Aufnahme von Kapital durch die Emission neuer Aktien im Sitzstaat des Emittenten und den Handel der zugehörigen ADR an einer US-Börse ist ein Level III-Programm erforderlich. Neben den Formblättern Form F-6 und Form 20-F ist das Formblatt F-1 einzureichen; die dort geforderten Pflichten zur Offenlegung und Aufstellung von Jahresabschlüssen nach US-GAAP entsprechen weitgehend denen nach Form 20-F.
Neben den an die Öffentlichkeit gerichteten Level I- bis Level III-Programmen besteht die Möglichkeit einer Privatplazierung nach Rule 144 A, die sich an spezielle institutionelle Käufer (Qualified Institutional Buyers) richtet. Eine Privatplazierung hat den Vorteil, daß kein aufwendiges Registrierungsverfahren notwendig und kein Abschluß nach US-GAAP erforderlich ist.
Antwort auf Beitrag Nr.: 34.135.368 von honorar am 20.05.08 17:45:56Hallo honorar,
schön wieder von Dir zu lesen.
Da Du jetzt wieder investiert bist gehe ich davon aus das du trib wieder für unterbewertet hältst?
schön wieder von Dir zu lesen.
Da Du jetzt wieder investiert bist gehe ich davon aus das du trib wieder für unterbewertet hältst?
Antwort auf Beitrag Nr.: 34.147.055 von Vakataka am 21.05.08 22:10:17Ja, die Aktie befindet sich im Bereich des ATL. Für mich ist Trib nach wie vor eine Perle.
Hier klar zu sehen:
Hier noch einmal zur Erinnerung aus letztem Dezember. Das dürfte sich bald positiv auf die Zahlen auswirken:
Trinity Biotech Cuts Jobs, Products
Friday December 7, 12:52 pm ET
Trinity Biotech to Cut Work Force, Close Manufacturing Plant in Major Restructuring
NEW YORK (AP) -- Trinity Biotech PLC, a developer and manufacturer of diagnostic products, said Friday it is reducing its work force to save $5 million annually, shuttering its Swedish manufacturing plant and discontinuing products as part of a major restructuring plan.
ADVERTISEMENT
The Irish company said the restructuring plan will reduce complexities in the business and allow it to target research and development.
As a result of the restructuring, the company will incur a one-time write-off of about $39.5 million before taxes in the fourth quarter. The company said except for about $2 million for redundancy costs and the closure of the Swedish plant, the write-off has no cash impact.
Trinity decided to discontinue 106 hemostasis and 69 infectious-disease products. The company offers hemostasis test kits for the detection of blood disorders. The company said the halting of the hemostasis products will be negligible on revenue. Eliminating the infectious-disease products will have less than a 1 percent impact on the company's revenue, Trinity said.
In addition, Trinity is suspending development of a number of ongoing projects, the two most significant being an HIV over-the-counter product and the development of an HIV confirmatory test. The project suspensions will allow the company to focus on a small number of key projects in each division, Trinity said.
Meanwhile, the closing of the plant in Umea, Sweden, will result in an overall net savings of about $500,000 a year, Trinity said. The company will benefit from some of these savings in 2008, with the full impact in 2009.
The company is also recognizing an impairment of about $20.2 million against its intangible assets, primarily goodwill.
Trinity did not disclose in its press release the number of positions it is cutting and could not be reached by the Associated Press.
Shares of Trinity fell 30 cents, or 3.6 percent, to $8.15 in afternoon trading. The stock has ranged between $7.93 and $11.85 over the past year.
Trinity Biotech Cuts Jobs, Products
Friday December 7, 12:52 pm ET
Trinity Biotech to Cut Work Force, Close Manufacturing Plant in Major Restructuring
NEW YORK (AP) -- Trinity Biotech PLC, a developer and manufacturer of diagnostic products, said Friday it is reducing its work force to save $5 million annually, shuttering its Swedish manufacturing plant and discontinuing products as part of a major restructuring plan.
ADVERTISEMENT
The Irish company said the restructuring plan will reduce complexities in the business and allow it to target research and development.
As a result of the restructuring, the company will incur a one-time write-off of about $39.5 million before taxes in the fourth quarter. The company said except for about $2 million for redundancy costs and the closure of the Swedish plant, the write-off has no cash impact.
Trinity decided to discontinue 106 hemostasis and 69 infectious-disease products. The company offers hemostasis test kits for the detection of blood disorders. The company said the halting of the hemostasis products will be negligible on revenue. Eliminating the infectious-disease products will have less than a 1 percent impact on the company's revenue, Trinity said.
In addition, Trinity is suspending development of a number of ongoing projects, the two most significant being an HIV over-the-counter product and the development of an HIV confirmatory test. The project suspensions will allow the company to focus on a small number of key projects in each division, Trinity said.
Meanwhile, the closing of the plant in Umea, Sweden, will result in an overall net savings of about $500,000 a year, Trinity said. The company will benefit from some of these savings in 2008, with the full impact in 2009.
The company is also recognizing an impairment of about $20.2 million against its intangible assets, primarily goodwill.
Trinity did not disclose in its press release the number of positions it is cutting and could not be reached by the Associated Press.
Shares of Trinity fell 30 cents, or 3.6 percent, to $8.15 in afternoon trading. The stock has ranged between $7.93 and $11.85 over the past year.
Technisch sieht es allerdings leider bescheiden aus:
http://quotes.barchart.com/texpert.asp?sym=trib&what=opinion
http://quotes.barchart.com/texpert.asp?sym=trib&what=opinion
Antwort auf Beitrag Nr.: 34.182.700 von honorar am 27.05.08 20:44:43abwarten. Ich seh das anders.
na also, geht doch 

Ich würde auf die Charttechnik nicht all zu viel setzen.
Der Kursanstieg jetzt kann Montag schon wieder Makulatur sein.
Wichtig ist, das sie den Laden endlich vernünftig führen.
Das scheint sich jetzt langsam abzuzeichnen.
Es gibt da noch ein paar heikle Themen, aber das wird schon
Der Kursanstieg jetzt kann Montag schon wieder Makulatur sein.
Wichtig ist, das sie den Laden endlich vernünftig führen.
Das scheint sich jetzt langsam abzuzeichnen.
Es gibt da noch ein paar heikle Themen, aber das wird schon

Antwort auf Beitrag Nr.: 34.208.365 von Vakataka am 30.05.08 21:31:52Ich bin ja positiv gestimmt, sonst hätte ich sie nicht gekauft, aber es gehört für mich dazu, auch die andere Seite zu zeigen.
Ich bin bereits gut im grünen Bereich und bleibe dabei.
Ich gehöre nicht zu der Familie der Pusher oder Basher, darum habe ich mir erlaubt, 2 Seiten hier zu posten.
Ich bin bereits gut im grünen Bereich und bleibe dabei.
Ich gehöre nicht zu der Familie der Pusher oder Basher, darum habe ich mir erlaubt, 2 Seiten hier zu posten.

Wenn man hier auf Aktualisieren klickt, hat man RT 

und wer sich den Link speicher will:
http://www.advfn.com/p.php?pid=staticchart&s=N%5ETRIB&p=0&t=…
Da ist mein Wochenendgeschenk

und wer sich den Link speicher will:
http://www.advfn.com/p.php?pid=staticchart&s=N%5ETRIB&p=0&t=…
Da ist mein Wochenendgeschenk

Antwort auf Beitrag Nr.: 34.208.601 von honorar am 30.05.08 22:04:21tja, was montag ist werden wir sehen, aber ich bin gespannt wie FRA eroeffnet. Mal sehen obs nen grosses Gap gibt.
Ich freu mich und laechel weiter bis Montag.














Sascha
Ich freu mich und laechel weiter bis Montag.














Sascha
http://quotes.barchart.com/texpert.asp?sym=trib&what=opinion
sieht schon anders aus als die Tage.
Meiner Meinung nach geht noch was bis 4.10 und dann abwarten obs drueber geht, aber drunter wirds nimma.
Schon interessasnt wie man shares ausgibt und paar tage spaeter die news rauskommen. Well well well
sieht schon anders aus als die Tage.
Meiner Meinung nach geht noch was bis 4.10 und dann abwarten obs drueber geht, aber drunter wirds nimma.
Schon interessasnt wie man shares ausgibt und paar tage spaeter die news rauskommen. Well well well
Moin zusammen,
habe ich zwar etwas daneben gelegen aber die Richtung nach oben stimmt auf jeden fall.
Bin etwas ueber die Stille hier ueberrascht denn mitlesen tun ja doch einige aber sagen keiner.
Nun denn, ich freu mich weiter denn so oder so es geht auf.
Sascha
habe ich zwar etwas daneben gelegen aber die Richtung nach oben stimmt auf jeden fall.
Bin etwas ueber die Stille hier ueberrascht denn mitlesen tun ja doch einige aber sagen keiner.
Nun denn, ich freu mich weiter denn so oder so es geht auf.
Sascha
Antwort auf Beitrag Nr.: 34.241.419 von Neela01 am 05.06.08 04:41:34Moinsen,
Du kannst Dir sicher sein, dass hier in D nur eine Handvoll Leute den Wert kennen. Die Musik spielt drüben und hier passt sich der Kurs nur an.
Du kannst Dir sicher sein, dass hier in D nur eine Handvoll Leute den Wert kennen. Die Musik spielt drüben und hier passt sich der Kurs nur an.
Antwort auf Beitrag Nr.: 34.251.798 von honorar am 06.06.08 09:50:52Tach auch,
ja da hast Du klar recht, die musik wird drueben gemacht aber es ist teilweise schon ne frechheit was fuer Gaps es gibt zwischen hier und drueben. Haben wir ja die letzten tage gesehen, ein unterschied von mehr als 5 cent pro aktie.
Nun denn, ich bin sicher und man sieht es ja das es auf geht. Heute eventuell wieder bissl ab aber am montag dann wird wieder gelaechelt.
Nach ewiger recherche ueber diesen wert sollte man im klaren sein das die platzierung bei instis auf kurz oder lang nach oben fuehren wird.
Allzeittief war in naehe oder auch erreicht worden und nun der sagen wir rebound. Schliessung des werkes in Skandinavien wird positiv auf die Q2 zahlen sein spaetestens auf die zahlen Q3 einwirken.
Das Modell dieser Firma ist klar und ausbaufaehig und der markt ist auf nachfrage positioniert meiner meinung nach.
Haette ich mehr geld uebrig wuerde ich voll reingehen in diesen wert aber leider nunja ist dem nicht so.
Es geht auf, ganz klar zu sehen an den seiten die honorar reingestellt hat und die taeglich upgedated werden.
Sascha
ja da hast Du klar recht, die musik wird drueben gemacht aber es ist teilweise schon ne frechheit was fuer Gaps es gibt zwischen hier und drueben. Haben wir ja die letzten tage gesehen, ein unterschied von mehr als 5 cent pro aktie.
Nun denn, ich bin sicher und man sieht es ja das es auf geht. Heute eventuell wieder bissl ab aber am montag dann wird wieder gelaechelt.
Nach ewiger recherche ueber diesen wert sollte man im klaren sein das die platzierung bei instis auf kurz oder lang nach oben fuehren wird.
Allzeittief war in naehe oder auch erreicht worden und nun der sagen wir rebound. Schliessung des werkes in Skandinavien wird positiv auf die Q2 zahlen sein spaetestens auf die zahlen Q3 einwirken.
Das Modell dieser Firma ist klar und ausbaufaehig und der markt ist auf nachfrage positioniert meiner meinung nach.
Haette ich mehr geld uebrig wuerde ich voll reingehen in diesen wert aber leider nunja ist dem nicht so.
Es geht auf, ganz klar zu sehen an den seiten die honorar reingestellt hat und die taeglich upgedated werden.
Sascha
Hier mal was Sachliches von letzter Woche:
Trinity Biotech Announces Important Performance Findings on the Uni-Gold Recombigen Rapid HIV Test
DUBLIN, IRELAND -- (MARKET WIRE) -- 05/28/08 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced important findings on the performance of the Uni-Gold Recombigen HIV test, as published in the April issue of the Journal of Clinical Microbiology.
The study carried out assessed the sensitivities of current, FDA-approved and CLIA-waived, rapid HIV tests and found significant variability with respect to early HIV detection capability in recently infected individuals. In this study, Uni-Gold demonstrated far superior performance than all currently available FDA-approved, CLIA-waived rapid HIV tests and outperformed even an FDA-approved, "moderately complex" rapid HIV test available.
Brendan Farrell, Chief Executive Officer of Trinity Biotech, commented, "Accurately diagnosing HIV infections is central to stemming the spread of the disease. The Uni-Gold Recombigen Rapid HIV test is the only FDA-approved, CLIA-waived rapid HIV test available with 100 percent sensitivity, or the ability to determine a patient's HIV positive status. As was noted in this important study, of all the FDA-approved rapid HIV tests available today, only the Uni-Gold Recombigen HIV test could potentially detect both IgG and IgM, using a patented, third generation, sandwich-based capture and detection system. This is why we firmly believe Uni-Gold is ideally suited as the screening test for acute care settings, such as hospital emergency rooms, labor and delivery settings, or stat labs where blood is being taken from the patient and 100 percent sensitivity and earlier detection is critical. Trinity wishes to commend researchers at the University of California, San Francisco and the San Francisco Department of Health for their important work assessing rapid HIV test performance in recently infected individuals."
For further information, please see: http://www.unigoldhiv.com/ or call 800.325.3424.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Contact :
Trinity Biotech plc
Brendan Farrell
(353)-1-2769800
E-mail: Email Contact
Trinity Biotech Announces Important Performance Findings on the Uni-Gold Recombigen Rapid HIV Test
DUBLIN, IRELAND -- (MARKET WIRE) -- 05/28/08 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced important findings on the performance of the Uni-Gold Recombigen HIV test, as published in the April issue of the Journal of Clinical Microbiology.
The study carried out assessed the sensitivities of current, FDA-approved and CLIA-waived, rapid HIV tests and found significant variability with respect to early HIV detection capability in recently infected individuals. In this study, Uni-Gold demonstrated far superior performance than all currently available FDA-approved, CLIA-waived rapid HIV tests and outperformed even an FDA-approved, "moderately complex" rapid HIV test available.
Brendan Farrell, Chief Executive Officer of Trinity Biotech, commented, "Accurately diagnosing HIV infections is central to stemming the spread of the disease. The Uni-Gold Recombigen Rapid HIV test is the only FDA-approved, CLIA-waived rapid HIV test available with 100 percent sensitivity, or the ability to determine a patient's HIV positive status. As was noted in this important study, of all the FDA-approved rapid HIV tests available today, only the Uni-Gold Recombigen HIV test could potentially detect both IgG and IgM, using a patented, third generation, sandwich-based capture and detection system. This is why we firmly believe Uni-Gold is ideally suited as the screening test for acute care settings, such as hospital emergency rooms, labor and delivery settings, or stat labs where blood is being taken from the patient and 100 percent sensitivity and earlier detection is critical. Trinity wishes to commend researchers at the University of California, San Francisco and the San Francisco Department of Health for their important work assessing rapid HIV test performance in recently infected individuals."
For further information, please see: http://www.unigoldhiv.com/ or call 800.325.3424.
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Contact :
Trinity Biotech plc
Brendan Farrell
(353)-1-2769800
E-mail: Email Contact
Trinity Biotech Retains Lytham Partners, LLC to Conduct National Investor Relations Programme
DUBLIN, IRELAND -- (MARKET WIRE) -- 05/29/08 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, announced that it has retained Lytham Partners, LLC to conduct a full-scale national investor relations programme.
Brendan Farrell, Chief Executive Officer, said, "We have made substantial progress in recent quarters to reposition the company for improved operational performance. We believe, as do the managing partners at Lytham Partners, that operational and financial performance are the predominant attributes by which a company should be judged and valued. Having made the changes necessary to enhance our performance, it is now important that the investment community in our largest market, the United States, fully understands the strategic direction and the investment opportunity that Trinity Biotech represents.
"We believe the efforts of Lytham Partners can substantially improve the potential for enhanced shareholder value as we continue to successfully execute our strategic business plan."
Joe Diaz, partner at Lytham Partners, commented, "We look forward to the opportunity of representing Trinity Biotech to the national investment community. We believe that the diagnostic testing technology that Trinity Biotech brings to the market has the potential to dramatically improve the ability of medical professionals to rapidly diagnose, treat, and improve quality of life for their patients worldwide. We look forward to working with the entire Trinity Biotech team."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Lytham Partners is a national investor relations firm representing small-cap companies that feature leading-edge products, services and/or technology. In addition to a client roster of innovative domestically based companies, the firm has extensive experience working with international clients looking to expand their visibility in the U.S. investment markets.
Contact :
Trinity Biotech plc
Brendan Farrell
(353)-1-2769800
E-mail: Email ContactLytham Partners, LLC
Joe Diaz
Joe Dorame
Robert Blum
(602) 889-9700
DUBLIN, IRELAND -- (MARKET WIRE) -- 05/29/08 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, announced that it has retained Lytham Partners, LLC to conduct a full-scale national investor relations programme.
Brendan Farrell, Chief Executive Officer, said, "We have made substantial progress in recent quarters to reposition the company for improved operational performance. We believe, as do the managing partners at Lytham Partners, that operational and financial performance are the predominant attributes by which a company should be judged and valued. Having made the changes necessary to enhance our performance, it is now important that the investment community in our largest market, the United States, fully understands the strategic direction and the investment opportunity that Trinity Biotech represents.
"We believe the efforts of Lytham Partners can substantially improve the potential for enhanced shareholder value as we continue to successfully execute our strategic business plan."
Joe Diaz, partner at Lytham Partners, commented, "We look forward to the opportunity of representing Trinity Biotech to the national investment community. We believe that the diagnostic testing technology that Trinity Biotech brings to the market has the potential to dramatically improve the ability of medical professionals to rapidly diagnose, treat, and improve quality of life for their patients worldwide. We look forward to working with the entire Trinity Biotech team."
Forward-looking statements in this release are made pursuant to the "safe harbor" provision of the Private Securities Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialization and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and blood coagulation disorders, and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
Lytham Partners is a national investor relations firm representing small-cap companies that feature leading-edge products, services and/or technology. In addition to a client roster of innovative domestically based companies, the firm has extensive experience working with international clients looking to expand their visibility in the U.S. investment markets.
Contact :
Trinity Biotech plc
Brendan Farrell
(353)-1-2769800
E-mail: Email ContactLytham Partners, LLC
Joe Diaz
Joe Dorame
Robert Blum
(602) 889-9700
Antwort auf Beitrag Nr.: 34.253.274 von honorar am 06.06.08 12:19:47












achja, auch von mir mal was sachliches, hehehehe.
1+1 ist 2
schoenes WE












achja, auch von mir mal was sachliches, hehehehe.
1+1 ist 2
schoenes WE
ok, war nen scherz. Bloss nicht aufregen.
Freut euch lieber und am Montag geht es weiter.


Freut euch lieber und am Montag geht es weiter.


Tach,
scheint sich abzuzeichnen das hier kaum einer posted.
An Vakataka mal die Frage von welchen heiklen Themen er spricht bei Trib ? In einem vorherigen Posting steht da was von heiklen Themen, lass uns doch mal was das fuer welche sind.
Find ich sehr schade das hier alles so schreibfaul ist und alles nur mitliest. Nen kleines hallo waere doch auch mal nett von den anwesenden Mitlesern. Waere doch schoen mal wieder etwas Leben hier in den thread zu bringen.
Der eine mag mich fuer nen dummschwaetzer halten und der andere fuer nen Trottel aber in anderen threads funktioniert das ganz gut mit der kommunikation und dort kann man auch mal nebenbei spass haben ohne schlechtes Gewissen.
schoenen Tag euch
scheint sich abzuzeichnen das hier kaum einer posted.
An Vakataka mal die Frage von welchen heiklen Themen er spricht bei Trib ? In einem vorherigen Posting steht da was von heiklen Themen, lass uns doch mal was das fuer welche sind.
Find ich sehr schade das hier alles so schreibfaul ist und alles nur mitliest. Nen kleines hallo waere doch auch mal nett von den anwesenden Mitlesern. Waere doch schoen mal wieder etwas Leben hier in den thread zu bringen.
Der eine mag mich fuer nen dummschwaetzer halten und der andere fuer nen Trottel aber in anderen threads funktioniert das ganz gut mit der kommunikation und dort kann man auch mal nebenbei spass haben ohne schlechtes Gewissen.
schoenen Tag euch
Mal nen kleiner Tip von mir,
es geht bis 4.55 hoch !! Wetten ! Kleines auf und ab aber die alte marke von vor April anfang april wird wieder erreicht.
Desweiteren find ich, das mit kleinen Umsaetzen in USA der kurs hochgeht sehr interessasnt und angenehm. Findet also kein abverkauf statt.
Vielleicht hat ja dazu mal jemand ne meinung.
es geht bis 4.55 hoch !! Wetten ! Kleines auf und ab aber die alte marke von vor April anfang april wird wieder erreicht.
Desweiteren find ich, das mit kleinen Umsaetzen in USA der kurs hochgeht sehr interessasnt und angenehm. Findet also kein abverkauf statt.
Vielleicht hat ja dazu mal jemand ne meinung.
Antwort auf Beitrag Nr.: 34.271.078 von Neela01 am 10.06.08 07:48:01also ist bin sehr skeptisch, ob die ständig steigenden Kosten von TRib in den Griff bekommen werden. In den letzten 3 Jahren sind die Kosten immer stärker gestiegen als der Umsatz und die Übernahmen haben dieses Bild noch verstärkt. Aus meiner Sicht sollte das Management ausgetauscht werden.
Na mal sehen, ob die Schließung der Fabrik ab dem dritten Quartal endlich zu sinkenden kosten und steigenden Gewinnen führt. Von Charttechnik halte ich bei Trib nicht viel. Es wurden ja auch die 25 US-D ausgerufen, die alle vier Jahre im Chart zu sehen sind.
Ich bin maßlos enttäuscht von Trib. Gute Produkte aber schlechtes Management!
Na mal sehen, ob die Schließung der Fabrik ab dem dritten Quartal endlich zu sinkenden kosten und steigenden Gewinnen führt. Von Charttechnik halte ich bei Trib nicht viel. Es wurden ja auch die 25 US-D ausgerufen, die alle vier Jahre im Chart zu sehen sind.
Ich bin maßlos enttäuscht von Trib. Gute Produkte aber schlechtes Management!
Jau genauso ist es, eigentlich wirklich gute Produkte!!!, aber es kommt einfach nichts rüber, die bekommen es leider nicht auf die Reihe was aus sich zu machen!!!! Nur so am Rande ich bin damals bei 16 EU eingestiegen  und bin von Ihnen überzeugt, aber es geht einfach leider gar nichts
und bin von Ihnen überzeugt, aber es geht einfach leider gar nichts  die könnten vieeel mehr aus sich machen schadr drum!!
die könnten vieeel mehr aus sich machen schadr drum!!
 und bin von Ihnen überzeugt, aber es geht einfach leider gar nichts
und bin von Ihnen überzeugt, aber es geht einfach leider gar nichts  die könnten vieeel mehr aus sich machen schadr drum!!
die könnten vieeel mehr aus sich machen schadr drum!!
Alles anschnallen und festhalten, es geht wieder runter.


Moin,
ich sollt mich selbststaendig machen mit Prognosen, da lieg ich ja ganz gut mit.




ich sollt mich selbststaendig machen mit Prognosen, da lieg ich ja ganz gut mit.




Trinity Biotech to Demonstrate Destiny Max at MLTD Conference in Athens
Monday June 23, 8:00 am ET
http://biz.yahoo.com/iw/080623/0409387.html
Trinity Biotech Announces the Launch of New HIV Incidence Assay
Tuesday June 24, 8:00 am ET
http://biz.yahoo.com/iw/080624/0409897.html
Monday June 23, 8:00 am ET
http://biz.yahoo.com/iw/080623/0409387.html
Trinity Biotech Announces the Launch of New HIV Incidence Assay
Tuesday June 24, 8:00 am ET
http://biz.yahoo.com/iw/080624/0409897.html
Freitag 31.10. kommen Zahlen, mal sehen, womit sie diesmal kommen 

Der Destiny Max wirds bringen. mehr kann ich nicht sagen 

Trinity Biotech Announces First Quarter 2009 Financial Results
EPS Increases to $0.12 From $0.055. Revenues Decrease 3.5% on a Constant Currency Basis
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
EPS Increases to $0.12 From $0.055. Revenues Decrease 3.5% on a Constant Currency Basis
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
Antwort auf Beitrag Nr.: 37.064.679 von Vakataka am 29.04.09 17:50:02Detailed Quote for Trinity Biotech plc (TRIB)
$ 2.30 0.65 (+39.39%) Volume: 265.16 k 11:45 EDT 29.04.2009
$ 2.30 0.65 (+39.39%) Volume: 265.16 k 11:45 EDT 29.04.2009
Antwort auf Beitrag Nr.: 37.064.679 von Vakataka am 29.04.09 17:50:02ein EPS von 0,12 US-D ist endlich mal eine Positive Überraschung, auf die wir schon lange gewartet haben.
Hallo Vaka,
besitzt TRIB auch Testsysteme bzw. -streifen für Schweinepest? Wenn ja, dann sollte das zweite Quartal ähnlich erfolgreich verlaufen, wie das erste Quartal. Dann sollte ein Kurs in der Region 5 US-D möglich sein.
bye Danger
Hallo Vaka,
besitzt TRIB auch Testsysteme bzw. -streifen für Schweinepest? Wenn ja, dann sollte das zweite Quartal ähnlich erfolgreich verlaufen, wie das erste Quartal. Dann sollte ein Kurs in der Region 5 US-D möglich sein.
bye Danger
Antwort auf Beitrag Nr.: 37.069.645 von Dangerous am 30.04.09 09:19:28Hallo Danger,
wenn wir für 2009 bei einem EPS von 0,45 - 0,50$ landen wäre das doch was, warten wir mal ab
Von einem Schweinepest Test ist mir nichts bekannt, die sind ja nicht in der Tiermedizin aktiv.
Trib hat einen Test gegen Influenza A,B und Parainfluenza von Bartels.
http://www.trinitybiotech.com/data/Products/Insert/B1029-86B…
Für Umsatz und Gewinn sind aber andere Dinge wichtiger.
vaka
wenn wir für 2009 bei einem EPS von 0,45 - 0,50$ landen wäre das doch was, warten wir mal ab

Von einem Schweinepest Test ist mir nichts bekannt, die sind ja nicht in der Tiermedizin aktiv.
Trib hat einen Test gegen Influenza A,B und Parainfluenza von Bartels.
http://www.trinitybiotech.com/data/Products/Insert/B1029-86B…
Für Umsatz und Gewinn sind aber andere Dinge wichtiger.
vaka
Antwort auf Beitrag Nr.: 37.070.593 von Vakataka am 30.04.09 10:39:52Tageshoch bisher 3,62$, plus 40%
Eine FDA Zulassung wird doch jetzt noch nicht erwartet??

Eine FDA Zulassung wird doch jetzt noch nicht erwartet??
Hallo Vaka,
vielen Dank für deine fundierte Antwort. Bin jetzt so lange schon bei Trib dabei. Die Hoffnung stirbt zuletzt. Endlich mal prositiver Newsflow
vielen Dank für deine fundierte Antwort. Bin jetzt so lange schon bei Trib dabei. Die Hoffnung stirbt zuletzt. Endlich mal prositiver Newsflow

Antwort auf Beitrag Nr.: 37.202.225 von Vakataka am 18.05.09 19:32:20Da liegt was in der Luft?
$ 3.76 0.5999 (+18.98%) Volume: 174.49 k 13:21 EDT 10.06.2009
$ 3.76 0.5999 (+18.98%) Volume: 174.49 k 13:21 EDT 10.06.2009
riesige Umsätze! Erst deutlich nach oben. Jetzt wird schon wieder abverkauft! Gibt es News?
Antwort auf Beitrag Nr.: 37.528.188 von Dangerous am 07.07.09 18:45:51 Trinity Biotech Receives FDA Approval for Destiny Max in the USA
Trinity Biotech Receives FDA Approval for Destiny Max in the USA
DUBLIN, IRELAND -- (Marketwire) -- 07/07/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced the FDA approval and US launch of its high throughput haemostasis analyzer, the Destiny Max.
Trinity has today obtained FDA approval of its Destiny Max analyzer and is now launching the instrument in the U.S. market with immediate effect. The target market for the instrument includes university hospitals, high throughput general hospitals, high volume commercial laboratories and reference laboratories.
Key features of the Destiny Max include:
-- being the only high throughput instrument on the worldwide market that
allows simultaneous and automated measurement of mechanical and optical
clot detection, chromogenic and immuno-turbidimetric assays;
-- best in class graphical user interface and touch screen technology;
-- the most reliable and novel cap piercing solution on the market.
As part of its U.S. launch Trinity will be showcasing the Destiny Max at the ISTH Congress in Boston on 11-16 July, 2009 and at the AACC Clinical Lab Expo in Chicago on 21-23 July, 2009.
Commenting on the approval Ronan O'Caoimh, CEO said, "I am delighted to announce FDA approval of the Destiny Max analyzer for the U.S. market. This follows the successful launch of Destiny Max in Europe and other international markets, following CE mark approval in December, 2008. The FDA approval and U.S. launch of Destiny Max represents a key strategic milestone for Trinity. This completes the roll out of Destiny Max in all major worldwide markets thus providing Trinity with access to the high throughput haemostasis market, estimated to be US$500 million per annum.
This approval by the FDA significantly improves the marketability of the analyzer and represents a clear endorsement of Trinity's ability to integrate hardware, software, reagents and consumables into a new state-of-the-art instrument platform that will exceed customer expectations. In the few short months since its international launch we have already enjoyed considerable success with the Destiny Max globally with sales in Japan, China, Italy, Ireland, the Netherlands, Germany, Australia, Turkey and the United Kingdom. Based on the market reaction we have received to date, I am more convinced than ever that we now have the best haemostasis instrument available on the market today.
The combination of Destiny Max with our mid-throughput Destiny Plus and other instruments, in conjunction with our comprehensive reagent portfolio and strong service commitment means that Trinity is now in a leading position to offer a complete and best in class heamostasis product range to all customers in the market. This will undoubtedly act as a key driver for the growth of our haemostasis business going forward, which we expect will result in a significant increase in our market share."
Trinity Biotech Receives FDA Approval for Destiny Max in the USA
DUBLIN, IRELAND -- (Marketwire) -- 07/07/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced the FDA approval and US launch of its high throughput haemostasis analyzer, the Destiny Max.
Trinity has today obtained FDA approval of its Destiny Max analyzer and is now launching the instrument in the U.S. market with immediate effect. The target market for the instrument includes university hospitals, high throughput general hospitals, high volume commercial laboratories and reference laboratories.
Key features of the Destiny Max include:
-- being the only high throughput instrument on the worldwide market that
allows simultaneous and automated measurement of mechanical and optical
clot detection, chromogenic and immuno-turbidimetric assays;
-- best in class graphical user interface and touch screen technology;
-- the most reliable and novel cap piercing solution on the market.
As part of its U.S. launch Trinity will be showcasing the Destiny Max at the ISTH Congress in Boston on 11-16 July, 2009 and at the AACC Clinical Lab Expo in Chicago on 21-23 July, 2009.
Commenting on the approval Ronan O'Caoimh, CEO said, "I am delighted to announce FDA approval of the Destiny Max analyzer for the U.S. market. This follows the successful launch of Destiny Max in Europe and other international markets, following CE mark approval in December, 2008. The FDA approval and U.S. launch of Destiny Max represents a key strategic milestone for Trinity. This completes the roll out of Destiny Max in all major worldwide markets thus providing Trinity with access to the high throughput haemostasis market, estimated to be US$500 million per annum.
This approval by the FDA significantly improves the marketability of the analyzer and represents a clear endorsement of Trinity's ability to integrate hardware, software, reagents and consumables into a new state-of-the-art instrument platform that will exceed customer expectations. In the few short months since its international launch we have already enjoyed considerable success with the Destiny Max globally with sales in Japan, China, Italy, Ireland, the Netherlands, Germany, Australia, Turkey and the United Kingdom. Based on the market reaction we have received to date, I am more convinced than ever that we now have the best haemostasis instrument available on the market today.
The combination of Destiny Max with our mid-throughput Destiny Plus and other instruments, in conjunction with our comprehensive reagent portfolio and strong service commitment means that Trinity is now in a leading position to offer a complete and best in class heamostasis product range to all customers in the market. This will undoubtedly act as a key driver for the growth of our haemostasis business going forward, which we expect will result in a significant increase in our market share."
seit dem Tief steigt die Aktien immer in Sprüngen an. Jetzt ist aktuell wieder ein Ausbruch gelungen. Dieser könnte wieder 1-1,5 UD-$ nach oben laufen. Nächstes Kursziel ca. 6 US-D. Hoffentlich bringen die Quartalszahlen keine Ernüchterung.
Antwort auf Beitrag Nr.: 37.605.492 von Dangerous am 20.07.09 12:53:01
Trinity Biotech Announces Second Quarter 2009 Financial Results
EPS Increases to $0.14 From $0.07; Revenues Decrease 5.7% on a Constant Currency Basis
DUBLIN, IRELAND -- (Marketwire) -- 07/23/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2009.
On a constant currency basis, revenues for the quarter decreased to $32.3 million from $34.3 million, compared to the same period last year, representing a difference of 5.7%. Point-of-Care revenues increased substantially, growing by 50%, largely driven by increased HIV sales in the key markets of Africa and the USA. This was offset by a 13% decrease in Clinical Laboratory revenues, primarily attributable to a fall in haemostasis revenues in advance of the launch of Destiny Max and lower Fitzgerald revenues.
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
Trinity Biotech Announces Second Quarter 2009 Financial Results
EPS Increases to $0.14 From $0.07; Revenues Decrease 5.7% on a Constant Currency Basis
DUBLIN, IRELAND -- (Marketwire) -- 07/23/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended June 30, 2009.
On a constant currency basis, revenues for the quarter decreased to $32.3 million from $34.3 million, compared to the same period last year, representing a difference of 5.7%. Point-of-Care revenues increased substantially, growing by 50%, largely driven by increased HIV sales in the key markets of Africa and the USA. This was offset by a 13% decrease in Clinical Laboratory revenues, primarily attributable to a fall in haemostasis revenues in advance of the launch of Destiny Max and lower Fitzgerald revenues.
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
zum Glück sehr gute Nachrichten. Deutliche Gewinnsteigerung bei leicht rückläufigen Umsätzen. Warum der Kurs nachgibt, ist mir schleierhaft. Allerdings nachbörslich wird wieder ein Kurs von 5 US-D angezeigt. Wenn die Entwicklung so weiter geht, könnten wir einen Gewinn von ca. 0,50 US-D pro Jahr erzielen. In gute Zeiten wurde eine Bewertung mit einem KGV von 20 zugrunde gelegt. Das wäre ein möglicher Zielkurs von 10 US-D. Wenn jetzt der neue Test wieder für steigende Umsätze sorgt, dann könnten wir vielleicht Ende des Jahres dort landen. Wee will see
Antwort auf Beitrag Nr.: 37.605.492 von Dangerous am 20.07.09 12:53:01leider hat der Kurs die falsche Richtung eingeschlagen

starker Kurs heute

Antwort auf Beitrag Nr.: 37.949.746 von Vakataka am 09.09.09 21:02:01Das könnte der Grund für den heutigen Kursanstieg sein
http://seekingalpha.com/article/160651-trinity-biotech-irish…
http://seekingalpha.com/article/160651-trinity-biotech-irish…
DUBLIN, IRELAND, Oct. 14, 2009 (Marketwire) --
DUBLIN, IRELAND -- (Marketwire) -- 10/14/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will report financial results for the third quarter of 2009 on Wednesday October 21, 2009. The Company has scheduled a conference call for that same day, Wednesday, October 21, 2009, at 11:00am EDT (4:00pm BST) to discuss the results of the quarter.
Interested parties can access the call by dialing:
USA: 1-800-860-2442
International: 1-412-858-4600
Conference ID #: 434757
A simultaneous webcast of the call can be accessed at: http://www.videonewswire.com/event.asp?id=62977
A replay of the call can be accessed until October 23, 2009 by dialing:
USA: 1-877-344-7529
International: 1-412-317-0088
Conference ID #: 434757
DUBLIN, IRELAND -- (Marketwire) -- 10/14/09 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, will report financial results for the third quarter of 2009 on Wednesday October 21, 2009. The Company has scheduled a conference call for that same day, Wednesday, October 21, 2009, at 11:00am EDT (4:00pm BST) to discuss the results of the quarter.
Interested parties can access the call by dialing:
USA: 1-800-860-2442
International: 1-412-858-4600
Conference ID #: 434757
A simultaneous webcast of the call can be accessed at: http://www.videonewswire.com/event.asp?id=62977
A replay of the call can be accessed until October 23, 2009 by dialing:
USA: 1-877-344-7529
International: 1-412-317-0088
Conference ID #: 434757
Gewinn fast verdoppelt trotz Umsatzrückgang. Die Performance der Aktie ist sehr enttäuschend und nicht nachvollziehbar.
Antwort auf Beitrag Nr.: 38.228.941 von Dangerous am 22.10.09 09:53:04Wir gehen auf 55-60 US cent für 2009.
Die Aktie wird noch anziehen.
Viel Glück uns allen.
Die Aktie wird noch anziehen.
Viel Glück uns allen.
Die nächsten Wochen mal wieder auf Trib achten

Antwort auf Beitrag Nr.: 38.995.800 von Vakataka am 23.02.10 17:47:15warum? 

Antwort auf Beitrag Nr.: 38.996.284 von ehrenwort am 23.02.10 18:36:47Im März kommen die Q4 Zahlen raus, bis dahin sollte der Kurs zulegen. Wenn 2009 so ausfällt wie ich in #787 geschrieben habe kann sich jeder den Kurs hochrechnen.
Wenn die Erwartungen verfehlt werden geht es wieder böse abwärts
Bis dann
Wenn die Erwartungen verfehlt werden geht es wieder böse abwärts

Bis dann

Antwort auf Beitrag Nr.: 38.997.879 von Vakataka am 23.02.10 21:37:41$ 4.52-- 0.32 (+7.62%) Volume: 107.08 k
Hoffen wir mal, das dies nur der Anfang war.
Hoffen wir mal, das dies nur der Anfang war.
Antwort auf Beitrag Nr.: 38.998.332 von Vakataka am 23.02.10 22:27:22$ 5.07 0.32 (+6.74%) Volume: 199.77 k
So langsam macht die Aktie wieder Freude.
Sieht so aus, als ob die Q Zahlen gut ausfallen, zudem ist die Tristat Zulassung überfällig
So langsam macht die Aktie wieder Freude.
Sieht so aus, als ob die Q Zahlen gut ausfallen, zudem ist die Tristat Zulassung überfällig

Antwort auf Beitrag Nr.: 39.024.689 von Vakataka am 26.02.10 22:22:41Und weiter geht es
$ 5.48 0.41 (+8.09%) Volume: 140.08 k 12:10 EST 01.03.2010

$ 5.48 0.41 (+8.09%) Volume: 140.08 k 12:10 EST 01.03.2010
Antwort auf Beitrag Nr.: 39.035.095 von Vakataka am 01.03.10 18:26:45Hallo Vakata.
Vielen Dank, dass Du den Thread ein bißchen am Leben erhälst.
Das Ganze sieht ja schon fast nach einer Ralley aus. Weißt Du, wann die Quartalszahlen kommen sollen und vor allem was erwartet wird?
Gruß
Lubus104
Vielen Dank, dass Du den Thread ein bißchen am Leben erhälst.
Das Ganze sieht ja schon fast nach einer Ralley aus. Weißt Du, wann die Quartalszahlen kommen sollen und vor allem was erwartet wird?
Gruß
Lubus104
Antwort auf Beitrag Nr.: 39.035.604 von Lubus104 am 01.03.10 19:23:41Hallo Lubus,
die Q4 Zahlen wurden in der Vergangenheit um den 10 . März
bekannt gegeben. Bis zum 12.3 sollten die raus sein.
Erwartet werden um die 55 cent.
Die Daten für das 1. Quartal kommen Ende April.
Die Zulassung für Tristat ist überfällig..
Gruß
Vaka
die Q4 Zahlen wurden in der Vergangenheit um den 10 . März
bekannt gegeben. Bis zum 12.3 sollten die raus sein.
Erwartet werden um die 55 cent.
Die Daten für das 1. Quartal kommen Ende April.
Die Zulassung für Tristat ist überfällig..
Gruß
Vaka
Antwort auf Beitrag Nr.: 39.035.953 von Vakataka am 01.03.10 20:00:43Sorry,
erwartet werden für das Gesamtjahr um die 55 cent.
Bisher sind wir bei 41 cent ( 12-14,4-14,6 ).
erwartet werden für das Gesamtjahr um die 55 cent.
Bisher sind wir bei 41 cent ( 12-14,4-14,6 ).
Antwort auf Beitrag Nr.: 39.036.345 von Vakataka am 01.03.10 20:52:55Vielen Dank.
Das wäre dann ja ein KGV von 10, wenn es sich um US-Cent handelt, € - Cent mag ich ja gar nicht glauben.
Was genau ist Tristat, eine Weiterentwicklung des HIV-Tests?
Jedenfalls macht es wieder ein bißchen Freude, die Aktie anzuschauen. Bin schon seit 4 Jahren investiert und konnte gar nicht glauben, dass die so abgestürzt ist. Der Laden macht doch eigentlich gar nicht so einen schlechten Eindruck.
Das wäre dann ja ein KGV von 10, wenn es sich um US-Cent handelt, € - Cent mag ich ja gar nicht glauben.
Was genau ist Tristat, eine Weiterentwicklung des HIV-Tests?
Jedenfalls macht es wieder ein bißchen Freude, die Aktie anzuschauen. Bin schon seit 4 Jahren investiert und konnte gar nicht glauben, dass die so abgestürzt ist. Der Laden macht doch eigentlich gar nicht so einen schlechten Eindruck.
Achtung, anschnallen!
Das geht heute noch richtig ab.
Die Q4 Zahlen sind raus.
15,5 cent.
Aber der Hammer ist folgende Meldung:
Disposal of the Coagulation business
Trinity Biotech has entered into a binding agreement for the sale of its worldwide Coagulation business to the Stago Group for $90m. Of the consideration, $67.5m will be paid on closing, $11.25m on the first anniversary of closing and the remaining $11.25m on the second anniversary of closing. No conditions or earn out provisions will apply to this deferred element of the consideration which is supported by a bank guarantee. The transaction is expected to close during quarter 2, 2010. A further $4m will be released to working capital following the collection of existing accounts receivables.
In total, 320 Trinity employees will transfer to Stago and all their contractual rights and benefits under their existing employment arrangements will be honoured. Stago has committed to continue manufacturing coagulation reagents in Bray, Ireland and will invest in upgrading this facility. They will also take over the German factory where they will continue to manufacture the Destiny range of instruments. In addition, a number of Trinity sales and marketing personnel in the USA, UK, Germany and France will transfer to Stago. Consequently, the active contracts with customers and distributors will be assigned to Stago under their existing terms and arrangements.
Although our Coagulation revenues have decreased over the past 3 years, with the launch of the new Destiny Max instrument, that level of decrease had reduced during the past year and we were confident that we could succeed in growing our market share over the coming years. However, we felt that the price, which represents over 100% of our average market capitalisation over the past 3 months was a good one and represented excellent shareholder value.
Following this transaction, which will reduce revenues by approximately 40%, Trinity expects annualised revenues of $72m. Our goal is to achieve EPS of between 90% and 100% of existing levels.
Ronan O'Caoimh CEO of Trinity Biotech stated "While we were committed to Coagulation and believe we would have been successful in significantly increasing our market share, the offer received from Stago makes sense for our shareholders and employees. Moreover, Stago's expertise and commitment in this domain, will allow a more rapid market penetration of the coagulation franchise we developed over time.
Following this transaction we are confident of immediately recommencing on the path of revenue growth. Our goal is to immediately achieve EPS of between 90% and 100% of existing levels and then aggressively grow earnings from that point onwards.
The company will now focus on developing its point-of-care business (POC). Our focus in the POC area will be on Infectious Diseases, HbA1c and Coagulation, which all have double digit growth rates and each have a market size exceeding $300m.
Das geht heute noch richtig ab.
Die Q4 Zahlen sind raus.
15,5 cent.
Aber der Hammer ist folgende Meldung:
Disposal of the Coagulation business
Trinity Biotech has entered into a binding agreement for the sale of its worldwide Coagulation business to the Stago Group for $90m. Of the consideration, $67.5m will be paid on closing, $11.25m on the first anniversary of closing and the remaining $11.25m on the second anniversary of closing. No conditions or earn out provisions will apply to this deferred element of the consideration which is supported by a bank guarantee. The transaction is expected to close during quarter 2, 2010. A further $4m will be released to working capital following the collection of existing accounts receivables.
In total, 320 Trinity employees will transfer to Stago and all their contractual rights and benefits under their existing employment arrangements will be honoured. Stago has committed to continue manufacturing coagulation reagents in Bray, Ireland and will invest in upgrading this facility. They will also take over the German factory where they will continue to manufacture the Destiny range of instruments. In addition, a number of Trinity sales and marketing personnel in the USA, UK, Germany and France will transfer to Stago. Consequently, the active contracts with customers and distributors will be assigned to Stago under their existing terms and arrangements.
Although our Coagulation revenues have decreased over the past 3 years, with the launch of the new Destiny Max instrument, that level of decrease had reduced during the past year and we were confident that we could succeed in growing our market share over the coming years. However, we felt that the price, which represents over 100% of our average market capitalisation over the past 3 months was a good one and represented excellent shareholder value.
Following this transaction, which will reduce revenues by approximately 40%, Trinity expects annualised revenues of $72m. Our goal is to achieve EPS of between 90% and 100% of existing levels.
Ronan O'Caoimh CEO of Trinity Biotech stated "While we were committed to Coagulation and believe we would have been successful in significantly increasing our market share, the offer received from Stago makes sense for our shareholders and employees. Moreover, Stago's expertise and commitment in this domain, will allow a more rapid market penetration of the coagulation franchise we developed over time.
Following this transaction we are confident of immediately recommencing on the path of revenue growth. Our goal is to immediately achieve EPS of between 90% and 100% of existing levels and then aggressively grow earnings from that point onwards.
The company will now focus on developing its point-of-care business (POC). Our focus in the POC area will be on Infectious Diseases, HbA1c and Coagulation, which all have double digit growth rates and each have a market size exceeding $300m.
BID 5,90 ASK 6,29 an der Nasdaq,
das kann ein langer Abend werden.
Ich rechne mit Rekordumsätzen, muss schon mal meine Tan Liste rausholen
das kann ein langer Abend werden.
Ich rechne mit Rekordumsätzen, muss schon mal meine Tan Liste rausholen

Antwort auf Beitrag Nr.: 39.114.658 von Vakataka am 11.03.10 15:34:47Tan Liste wieder weggesteckt.
Starker Umsatz, Kurs miserabel.
Hoffe jetzt noch auf die FDA Zulassung.
@ Lubus
Das ist ein Diabetes Test, der wird mehr Umsatz bringen wie der Unigold.
Starker Umsatz, Kurs miserabel.
Hoffe jetzt noch auf die FDA Zulassung.
@ Lubus
Das ist ein Diabetes Test, der wird mehr Umsatz bringen wie der Unigold.
Antwort auf Beitrag Nr.: 39.114.595 von Vakataka am 11.03.10 15:29:22Hallo Vaka! Vielen Dank, dass du uns weiterhin auf dem Laufenden hälst. Welcher Teilbereich wird denn verkauft? Wenn sich durch den Deal die Umsätze nur um 40 % reduzieren, dann haben sie einen Superkaufpreis erzielt. Mal sehen, wie dieser neu investiert wird. Was bedeutet eigentlich Point of Sale in dieser Branche. Im Einzelhandel und im Bankwesen spricht man von Zahlungsverkehrsterminals. Sie werden also zukünftig nur noch Test und Medikamente verkaufen? Keine mehr selbst produzieren und entwickeln?
Antwort auf Beitrag Nr.: 39.122.653 von Dangerous am 12.03.10 11:49:28Hallo Danger
Steht alles in der PR.
Die komplette Coag Sparte ist verkauft.
Ich sehe das auch so, ist ein guter Verkaufpreis. Wenn Trib bisher für die Sparte 65 Mio ausgegeben hat sind 90 Mios schon ne Hausnummer.
Wenn man dann noch bedenkt das die Coag Sparte kaum zum Gewinn beigetragen hat..
Trib macht demnächst 40% weniger Umsatz, aber noch 90% der Gewinne bei halbierter Belegschaft.
Durch organisches Wachstum von 8-12% pro Jahr soll der alte Umsatz wieder erreicht werden.
Hierbei wird der Diabetes Test eine große Rolle spielen, könnte ein echter Blockbuster werden.
Die Verbindlichkeiten von ca 36 M können getilgt werden und man hat noch eine volle Kasse
von 50 M abzgl der anfallenden Steuer.
Man wird wieder kleine Firmen mit interessanten Produkten kaufen und die zur Marktreife entwickeln und zur Zulassung bringen.
Trib hat da wohl schon einige Lizenzen und Produkte im Visier.
Das ganze sieht für mich positiv aus, allerdings haben die Irren in der Vergangenheit bei Aquisitionen manchmal daneben gegriffen.
Im Moment sehen wir wohl sell on good news, das sollte sich spätestens ändern wenn der Deal vollzogen ist.
Ich warte jetzt auf die FDA Zulassung und hoffe das es ordentlich nach oben geht, dann bin ich erstmal raus.
Viel Glück uns allen
Vaka
Steht alles in der PR.
Die komplette Coag Sparte ist verkauft.
Ich sehe das auch so, ist ein guter Verkaufpreis. Wenn Trib bisher für die Sparte 65 Mio ausgegeben hat sind 90 Mios schon ne Hausnummer.
Wenn man dann noch bedenkt das die Coag Sparte kaum zum Gewinn beigetragen hat..
Trib macht demnächst 40% weniger Umsatz, aber noch 90% der Gewinne bei halbierter Belegschaft.
Durch organisches Wachstum von 8-12% pro Jahr soll der alte Umsatz wieder erreicht werden.
Hierbei wird der Diabetes Test eine große Rolle spielen, könnte ein echter Blockbuster werden.
Die Verbindlichkeiten von ca 36 M können getilgt werden und man hat noch eine volle Kasse
von 50 M abzgl der anfallenden Steuer.
Man wird wieder kleine Firmen mit interessanten Produkten kaufen und die zur Marktreife entwickeln und zur Zulassung bringen.
Trib hat da wohl schon einige Lizenzen und Produkte im Visier.
Das ganze sieht für mich positiv aus, allerdings haben die Irren in der Vergangenheit bei Aquisitionen manchmal daneben gegriffen.
Im Moment sehen wir wohl sell on good news, das sollte sich spätestens ändern wenn der Deal vollzogen ist.
Ich warte jetzt auf die FDA Zulassung und hoffe das es ordentlich nach oben geht, dann bin ich erstmal raus.
Viel Glück uns allen
Vaka
Hallo Vaka,
vielen Dank. Seit einem Jahr good news und der Kurs kommt kaum von der Stelle. Vielleicht wird ein Aktienrückkauf beschlossen. Das würde den Kurs auf Trapp bringen und könnte gut als Akquise-Währung eingesetzt werden. Bin schon solange dabei. Vielleicht sehe ich meinen Einstandskurs in 1-2 Jahren mal wieder. Die PR müsste verbessert werden und die Investorengespräche verstärkt werden.
Ciao
vielen Dank. Seit einem Jahr good news und der Kurs kommt kaum von der Stelle. Vielleicht wird ein Aktienrückkauf beschlossen. Das würde den Kurs auf Trapp bringen und könnte gut als Akquise-Währung eingesetzt werden. Bin schon solange dabei. Vielleicht sehe ich meinen Einstandskurs in 1-2 Jahren mal wieder. Die PR müsste verbessert werden und die Investorengespräche verstärkt werden.
Ciao
Antwort auf Beitrag Nr.: 39.157.107 von Dangerous am 17.03.10 11:38:52Der Deal ist durch, bin gespannt ob das im Kurs eingepreist ist,
oder geht es gleich richtig ab
Trinity Biotech Closes Deal to Sell Its Worldwide Coagulation Business Line to Stago
DUBLIN, IRELAND -- 05/05/10 --
Posted : Wed, 05 May 2010 11:00:16 GMT
Author : Trinity Biotech
Category : Press Release
DUBLIN, IRELAND -- 05/05/10 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it had closed its deal to dispose of its coagulation business to the Stago Group.
Trinity Biotech has sold its worldwide Coagulation business to the Stago Group for $90m of which $67.5m was paid on closing. A further $11.25m will be paid on the first anniversary of closing and the remaining $11.25m on the second anniversary of closing. No conditions or earnout provisions will apply to this deferred element of the consideration which is supported by a bank guarantee. A further $4m will be released to working capital following the collection of existing accounts receivables. Trinity has used the first tranche of the proceeds to repay all of its bank debts and to boost its cash reserves to in excess of $45m.
In total, 320 Trinity employees have transferred to Stago and all their contractual rights and benefits under their existing employment arrangements will be honoured. Stago will continue manufacturing coagulation reagents in Bray, Ireland and will invest in upgrading this facility. They have also taken over Trinity's manufacturing facility in Germany where they will manufacture the Destiny range of instruments. In addition, a number of Trinity sales and marketing personnel in the USA, UK, Germany and France have transferred to Stago. All active contracts with customers and distributors have been assigned to Stago under their existing terms and arrangements.
Given that coagulation revenues had been decreasing over the past number of years it was felt that the price, which represents over 100% of Trinity's average market capitalisation over the 3 months prior to the deal, was a good one and represented excellent shareholder value. Following the transaction, which will reduce revenues by approximately 40%, Trinity expects annualised revenues to be $72m whilst EPS will continue to be 90% to 100% of existing levels (51-57 cent EPS).
Trinity will now concentrate on developing its point-of-care business (POC) where the focus will be on Infectious Diseases, HbA1c and Coagulation, which all have double digit growth rates and each have a market size exceeding $300m.
Infectious diseases POC
Our concentration will be on developing qualitative tests in the sexually transmitted disease, enteric and respiratory fields utilising lateral flow technology for which we hold the requisite licences. We are well experienced in this area and currently have in excess of 20% of the HIV POC market worldwide.
Following the announcement of this transaction we significantly increased our point-of-care R&D activity in Ireland and have opened a new point-of-care R&D facility in our Carlsbad, San Diego facility.
HbA1c POC
Our Tri-stat Diabetes HbA1c rapid system has been FDA approved and is currently awaiting a CLIA waiver. The combination of Tri-stat and the new PDX instrument positions us strongly in this high growth market.
Coagulation POC
Under our agreement with the Stago Group we are free to participate in the POC segment of the coagulation market. We intend to develop a range of coagulation tests and will immediately commence the development of a lateral flow assay for D-dimer.
Ronan O'Caoimh, CEO of Trinity Biotech stated "We are very pleased to announce that we have closed the deal to sell our worldwide coagulation business to Stago from a number of perspectives
* we believe that the price of $90m represents excellent value for our shareholders;
* we have now eliminated all bank debt and have moved from a net debt position of $1 per share to a positive cash position of $3.50 per share;
* we will continue to be strongly profitable and will earn at least 51 cent per share in the next 12 months; and
* we now have the resources to aggressively implement our point of care business development strategy.
We would also like to wish Stago and their many new employees every success in the future."
http://www.earthtimes.org/articles/show/trinity-biotech-clos…
oder geht es gleich richtig ab

Trinity Biotech Closes Deal to Sell Its Worldwide Coagulation Business Line to Stago
DUBLIN, IRELAND -- 05/05/10 --
Posted : Wed, 05 May 2010 11:00:16 GMT
Author : Trinity Biotech
Category : Press Release
DUBLIN, IRELAND -- 05/05/10 -- Trinity Biotech plc (NASDAQ: TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it had closed its deal to dispose of its coagulation business to the Stago Group.
Trinity Biotech has sold its worldwide Coagulation business to the Stago Group for $90m of which $67.5m was paid on closing. A further $11.25m will be paid on the first anniversary of closing and the remaining $11.25m on the second anniversary of closing. No conditions or earnout provisions will apply to this deferred element of the consideration which is supported by a bank guarantee. A further $4m will be released to working capital following the collection of existing accounts receivables. Trinity has used the first tranche of the proceeds to repay all of its bank debts and to boost its cash reserves to in excess of $45m.
In total, 320 Trinity employees have transferred to Stago and all their contractual rights and benefits under their existing employment arrangements will be honoured. Stago will continue manufacturing coagulation reagents in Bray, Ireland and will invest in upgrading this facility. They have also taken over Trinity's manufacturing facility in Germany where they will manufacture the Destiny range of instruments. In addition, a number of Trinity sales and marketing personnel in the USA, UK, Germany and France have transferred to Stago. All active contracts with customers and distributors have been assigned to Stago under their existing terms and arrangements.
Given that coagulation revenues had been decreasing over the past number of years it was felt that the price, which represents over 100% of Trinity's average market capitalisation over the 3 months prior to the deal, was a good one and represented excellent shareholder value. Following the transaction, which will reduce revenues by approximately 40%, Trinity expects annualised revenues to be $72m whilst EPS will continue to be 90% to 100% of existing levels (51-57 cent EPS).
Trinity will now concentrate on developing its point-of-care business (POC) where the focus will be on Infectious Diseases, HbA1c and Coagulation, which all have double digit growth rates and each have a market size exceeding $300m.
Infectious diseases POC
Our concentration will be on developing qualitative tests in the sexually transmitted disease, enteric and respiratory fields utilising lateral flow technology for which we hold the requisite licences. We are well experienced in this area and currently have in excess of 20% of the HIV POC market worldwide.
Following the announcement of this transaction we significantly increased our point-of-care R&D activity in Ireland and have opened a new point-of-care R&D facility in our Carlsbad, San Diego facility.
HbA1c POC
Our Tri-stat Diabetes HbA1c rapid system has been FDA approved and is currently awaiting a CLIA waiver. The combination of Tri-stat and the new PDX instrument positions us strongly in this high growth market.
Coagulation POC
Under our agreement with the Stago Group we are free to participate in the POC segment of the coagulation market. We intend to develop a range of coagulation tests and will immediately commence the development of a lateral flow assay for D-dimer.
Ronan O'Caoimh, CEO of Trinity Biotech stated "We are very pleased to announce that we have closed the deal to sell our worldwide coagulation business to Stago from a number of perspectives
* we believe that the price of $90m represents excellent value for our shareholders;
* we have now eliminated all bank debt and have moved from a net debt position of $1 per share to a positive cash position of $3.50 per share;
* we will continue to be strongly profitable and will earn at least 51 cent per share in the next 12 months; and
* we now have the resources to aggressively implement our point of care business development strategy.
We would also like to wish Stago and their many new employees every success in the future."
http://www.earthtimes.org/articles/show/trinity-biotech-clos…
Antwort auf Beitrag Nr.: 39.459.933 von Vakataka am 05.05.10 13:17:26Die Übernahme kann eigentlich nicht eingepreist sein.
Trib ist jetzt Schuldenfrei und hat 45 Mio cash.
Die Zulassung für den Tri-stat Diabetes HbA1c kann täglich kommen.
Man lese die PR aufmerksam.
Der Makler in Stuttgart hat BID/Ask schon angehoben.
Viel Glück uns allen
Trib ist jetzt Schuldenfrei und hat 45 Mio cash.
Die Zulassung für den Tri-stat Diabetes HbA1c kann täglich kommen.
Man lese die PR aufmerksam.
Der Makler in Stuttgart hat BID/Ask schon angehoben.
Viel Glück uns allen
Nach dem guten Ergebnis geht es heute bestimmt wieder abwärts
DUBLIN, IRELAND, May 11, 2010 (Marketwire) --
Trinity Biotech Announces Quarter 1 Financial Results ;
EPS Increases by 25% to 15 Cent; Cash From Operations Increases 122%
Quarter 1 Results
Total revenues for the quarter were $29.0m which compares to $31.1m in quarter 1, 2009, a decrease of 6.7%.
Point-of-care revenues for the quarter decreased by 6.6% when compared to quarter 1, 2009. This decline is still largely attributable to the company's decision to restrict shipments to a major HIV customer due to credit related issues. This was partly offset by the continued growth of HIV sales in the USA which increased by 6% quarter on quarter.
Continuing clinical laboratory (i.e. excluding coagulation) revenues were $13.3m which represents a decrease of 3.5% when compared to $13.8m in quarter 1 2009. This decrease does not reflect an underlying reduction in business levels but rather that lyme sales, which are very seasonal in nature, are lower this quarter than in the corresponding quarter last year.
Coagulation revenues fell from $12.7m in quarter 1, 2009 to $11.4m in quarter 1, 2010, a decrease of 10.3%.

DUBLIN, IRELAND, May 11, 2010 (Marketwire) --
Trinity Biotech Announces Quarter 1 Financial Results ;
EPS Increases by 25% to 15 Cent; Cash From Operations Increases 122%
Quarter 1 Results
Total revenues for the quarter were $29.0m which compares to $31.1m in quarter 1, 2009, a decrease of 6.7%.
Point-of-care revenues for the quarter decreased by 6.6% when compared to quarter 1, 2009. This decline is still largely attributable to the company's decision to restrict shipments to a major HIV customer due to credit related issues. This was partly offset by the continued growth of HIV sales in the USA which increased by 6% quarter on quarter.
Continuing clinical laboratory (i.e. excluding coagulation) revenues were $13.3m which represents a decrease of 3.5% when compared to $13.8m in quarter 1 2009. This decrease does not reflect an underlying reduction in business levels but rather that lyme sales, which are very seasonal in nature, are lower this quarter than in the corresponding quarter last year.
Coagulation revenues fell from $12.7m in quarter 1, 2009 to $11.4m in quarter 1, 2010, a decrease of 10.3%.
Antwort auf Beitrag Nr.: 39.499.812 von Vakataka am 11.05.10 14:54:03Hallo zusammen, bin mal wieder seit Jahren im Board.
Scheint sich nicht viel verändert zu haben.
PR ist nicht gut.
Verwässerung durch immer neue Aktien.
Wer da glaubt, das ein Aktienrückkaufprogramm folgt, der irrt meiner Meinung nach. Ich habe diverse Jahre auf die HIV Zulassung gewartet und bin dan raus.
Gruß bibos
Scheint sich nicht viel verändert zu haben.
PR ist nicht gut.
Verwässerung durch immer neue Aktien.
Wer da glaubt, das ein Aktienrückkaufprogramm folgt, der irrt meiner Meinung nach. Ich habe diverse Jahre auf die HIV Zulassung gewartet und bin dan raus.
Gruß bibos
wurde eigentlich schon die Quartalszahlen besprochen?
mal sehen, wie die Quartalszahlen ausfallen. Sollen wohl heute kommen.
Antwort auf Beitrag Nr.: 40.357.032 von Dangerous am 20.10.10 13:42:25
Nicht schlecht
EPS of 16.5 Cent per Share -- An Increase of 13%; Free Cash Flows of $3.8M
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
Nicht schlecht
EPS of 16.5 Cent per Share -- An Increase of 13%; Free Cash Flows of $3.8M
http://app.quotemedia.com/quotetools/newsStoryPopup.go?story…
Antwort auf Beitrag Nr.: 40.358.097 von Vakataka am 20.10.10 15:16:00Da geht noch mehr
Hier der aktuelle Kurs
http://www.nasdaq.com/aspx/nasdaqlastsale.aspx?symbol=TRIB&s…

Hier der aktuelle Kurs
http://www.nasdaq.com/aspx/nasdaqlastsale.aspx?symbol=TRIB&s…
Antwort auf Beitrag Nr.: 40.484.659 von Vakataka am 09.11.10 16:12:52DUBLIN, IRELAND--(Marketwire - 02/02/11) - Trinity Biotech plc (NASDAQ:TRIB - News), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced the acquisition of the Toronto based diagnostics company, Phoenix Biotech Corp, for US$2.5 million...
http://finance.yahoo.com/news/Trinity-Biotech-Announces-the-…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-the-…
Antwort auf Beitrag Nr.: 40.970.828 von Vakataka am 02.02.11 19:32:26Hi Vaka,
der Kurs dreht langsam nach oben. Der MACD hat gerade ein Kaufsignal generiert und der RSI hat noch reichlich Luft nach oben. Hoffentlich knacken wir jetzt die 9 US-D Marke. Dann wäre Luft bis fast 12 US-D.
Wann kommen denn die Quartalszahlen? Diese sollten dem Kurs doch Flügel verleihen?
Ciao Danger
der Kurs dreht langsam nach oben. Der MACD hat gerade ein Kaufsignal generiert und der RSI hat noch reichlich Luft nach oben. Hoffentlich knacken wir jetzt die 9 US-D Marke. Dann wäre Luft bis fast 12 US-D.
Wann kommen denn die Quartalszahlen? Diese sollten dem Kurs doch Flügel verleihen?
Ciao Danger
Antwort auf Beitrag Nr.: 41.041.134 von Dangerous am 15.02.11 11:20:26geht doch. Übernahme und Ausdehnung des Aktienrückkaufprogramms
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-8…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-8…
Antwort auf Beitrag Nr.: 41.041.134 von Dangerous am 15.02.11 11:20:26Hallo Danger,
Share Buy-Back ist geil, haben wir alle lange warten müssen,
bedeutet aber wohl auch das die nichts besseres mit der Kohle
anfangen können...
Inwieweit der Vertrag mit Menarini Diagnostics zu den Umsätzen
beitragen wird bleibt abzuwarten.
Die Q4 kommen normalerweise in der 2. Märzwoche.
Die Q1 in der 2. Maiwoche.
Im April sollte Stago noch 12 Mio überweisen und wenn ich mich
richtig erinnere warten wir immer noch auf eine FDA Zulassung.
Ich denke auch das die Aktie langsam Richtung 12$ läuft.
1/3 habe ich im Dezember vertickt, bei 12 kommt der Rest weg.
Viel Glück uns allen
vaka
Share Buy-Back ist geil, haben wir alle lange warten müssen,
bedeutet aber wohl auch das die nichts besseres mit der Kohle
anfangen können...
Inwieweit der Vertrag mit Menarini Diagnostics zu den Umsätzen
beitragen wird bleibt abzuwarten.
Die Q4 kommen normalerweise in der 2. Märzwoche.
Die Q1 in der 2. Maiwoche.
Im April sollte Stago noch 12 Mio überweisen und wenn ich mich
richtig erinnere warten wir immer noch auf eine FDA Zulassung.
Ich denke auch das die Aktie langsam Richtung 12$ läuft.
1/3 habe ich im Dezember vertickt, bei 12 kommt der Rest weg.
Viel Glück uns allen
vaka
Quartalszahlen sehen gut aus und der Kurs hat den Widerstand bei 8 US-D genommen. Jetzt könnte es bis 11-11,50 US-D hoch gehen. Da lauert der nächste größere Widerstand. Hoffentlich kommt dann mal wieder einen größerer Ausbruch der ja in aller Regelmäßigkeit alle 4-5 Jahre passiert
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-1…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-1…
Antwort auf Beitrag Nr.: 41.142.758 von Dangerous am 03.03.11 18:33:29Sieht wirklich gut aus und eine Dividende wollen die auch zahlen.
Ich lach mich schlapp, hätte ich nie für möglich gehalten.
Wenn jetzt noch die FDA Zulassung kommt...
Du meinst den Wiederstand bei 9$ ?
Vielleicht knacken wir heute noch die 10$
http://www.nasdaq.com/aspx/nasdaqlastsale.aspx?symbol=TRIB&s…
viel Glück uns allen
vaka
Ich lach mich schlapp, hätte ich nie für möglich gehalten.
Wenn jetzt noch die FDA Zulassung kommt...
Du meinst den Wiederstand bei 9$ ?
Vielleicht knacken wir heute noch die 10$

http://www.nasdaq.com/aspx/nasdaqlastsale.aspx?symbol=TRIB&s…
viel Glück uns allen
vaka
Antwort auf Beitrag Nr.: 41.142.987 von Vakataka am 03.03.11 19:04:53Hallo Vaka,
ja natürlich 9 US-D. Den haben wir ja nun genommen. Sieht gut aus. Endlich mal. Dann sollten eigentlich deutlich höhere Kurse drin sein.
Ciao Danger
ja natürlich 9 US-D. Den haben wir ja nun genommen. Sieht gut aus. Endlich mal. Dann sollten eigentlich deutlich höhere Kurse drin sein.
Ciao Danger
Antwort auf Beitrag Nr.: 41.151.053 von Dangerous am 04.03.11 20:58:49Hier nochmal alles interessante über Trinity in kompakter Zusammenfassung,
liest sich gut.
http://seekingalpha.com/article/256971-trinity-biotech-share…
liest sich gut.
http://seekingalpha.com/article/256971-trinity-biotech-share…
Antwort auf Beitrag Nr.: 41.166.759 von Vakataka am 08.03.11 18:54:14Trinity Biotech Announces CE Marking of Premier Hb9210
http://finance.yahoo.com/news/Trinity-Biotech-Announces-CE-i…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-CE-i…
Antwort auf Beitrag Nr.: 41.454.319 von Vakataka am 04.05.11 21:23:52http://seekingalpha.com/article/275895-best-healthcare-secto…
Antwort auf Beitrag Nr.: 41.682.993 von Dangerous am 22.06.11 13:05:13Quartalszahlen
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-3…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-iw-3…
Antwort auf Beitrag Nr.: 41.824.907 von Dangerous am 21.07.11 16:31:50aktuelle Quartalszahlen. Mein Gewinnziel für 2012 liegt bei 0,80 Dollar und mein Kursziel bei 12,00 Dollar (keine Kaufempfehlung). Charttechnisch haben wir jetzt die 10 Dollar wieder geknackt. Der Weg Richtung 11-12 Dollar ist frei. Hoffentlich gibt es noch einen interessanten Kauf, der nochmal zusätzlich Gewinn für 2012 bringt, denn sonst ist nicht soviel Fantasie im Kurs.
http://finance.yahoo.com/news/Trinity-Biotech-Announces-pz-3…
http://finance.yahoo.com/news/Trinity-Biotech-Announces-pz-3…
Trinity Biotech Announces FDA Approval of Premier Hb9210
DUBLIN, Ireland, Dec. 6, 2011 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has obtained FDA approval for the Premier Hb9210.
The Premier instrument is a fully automated system that measures HbA1c in high and low throughput clinical laboratories. HbA1c, also known as glycated hemoglobin, is a measure of a patient's average blood sugar control over the last two to three months and is a key tool in the diagnosis and monitoring of diabetes. The current worldwide prevalence of diabetes is estimated to be 250 million and it is expected to reach 380 million people by 2025. Within the USA it is estimated that 12.9% of persons over the age of 20 have diabetes, 40% of whom are undiagnosed. The worldwide costs of diabetes in 2007 were approximately $232 billion and this is expected to increase to $302 billion by 2025.
FDA approval enables the instrument to be launched and sold in the United States and will greatly facilitate sales in other international jurisdictions. The instrument has already been CE marked for sale in Europe and was formally launched in Quarter 2, 2011.
In the USA, Trinity will market and sell the Premier through a combination of direct selling through our existing US salesforce and through Fisher Healthcare's extensive distribution network. Fisher Healthcare is one of the leading distributors of HbA1c products in the USA with an installed base of approximately 1,000 A1c instruments. Fisher has recently signed a distribution agreement with Trinity and will be exclusively selling the Premier instrument in the USA.
Trinity also recently announced an agreement to exclusively supply Menarini Diagnostics with the new Premier instrument for distribution in European territories. As one of Europe's leading pharmaceutical and diagnostics companies, Menarini, with a turnover of €2.6 billion and 12,000 employees, is the market leader in HbA1c measurement in Europe. Menarini has a market share of 40% in the European HbA1c market, a large installed base of equipment and over 20 years experience in HbA1c measurement.
We are also making significant progress in obtaining the necessary approvals in other key markets. In June we submitted our application to the Chinese regulatory authorities and we expect to receive approval by the end of 2012. This will represent an excellent opportunity to build on our existing installed base in China.
The Premier analyser is a best in class instrument with the following key advantages :
Results available in 1 minute, making it the fastest test in the market;
State of the art software using touchscreen technology to facilitate ease of use for operators;
Patented exclusive boronate affinity technology, therefore eliminating interference from hemoglobin variants;
Modular instrument which will significantly reduce the cost of on-site maintenance.
Commenting on the announcement, Trinity Biotech's CEO, Mr Ronan O'Caoimh, said, "HbA1c testing is already a US$300 million global market with enormous growth potential. We have now the fastest, most user friendly and state of the art HbA1c analyser in the world. FDA approval of the instrument, in conjunction with its recent CE marking, now gives us access to the US and European markets, the two most important individual markets in the world, and will accelerate the approval process in other key markets such as Brazil and China. Together with our partners, Menarini in Europe and Fisher in the USA, we are ideally positioned to become a significant and serious player in the global HbA1c market in the coming years."
Source: Globe Newswire (December 6, 2011 - 8:00 AM EST)
DUBLIN, Ireland, Dec. 6, 2011 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced that it has obtained FDA approval for the Premier Hb9210.
The Premier instrument is a fully automated system that measures HbA1c in high and low throughput clinical laboratories. HbA1c, also known as glycated hemoglobin, is a measure of a patient's average blood sugar control over the last two to three months and is a key tool in the diagnosis and monitoring of diabetes. The current worldwide prevalence of diabetes is estimated to be 250 million and it is expected to reach 380 million people by 2025. Within the USA it is estimated that 12.9% of persons over the age of 20 have diabetes, 40% of whom are undiagnosed. The worldwide costs of diabetes in 2007 were approximately $232 billion and this is expected to increase to $302 billion by 2025.
FDA approval enables the instrument to be launched and sold in the United States and will greatly facilitate sales in other international jurisdictions. The instrument has already been CE marked for sale in Europe and was formally launched in Quarter 2, 2011.
In the USA, Trinity will market and sell the Premier through a combination of direct selling through our existing US salesforce and through Fisher Healthcare's extensive distribution network. Fisher Healthcare is one of the leading distributors of HbA1c products in the USA with an installed base of approximately 1,000 A1c instruments. Fisher has recently signed a distribution agreement with Trinity and will be exclusively selling the Premier instrument in the USA.
Trinity also recently announced an agreement to exclusively supply Menarini Diagnostics with the new Premier instrument for distribution in European territories. As one of Europe's leading pharmaceutical and diagnostics companies, Menarini, with a turnover of €2.6 billion and 12,000 employees, is the market leader in HbA1c measurement in Europe. Menarini has a market share of 40% in the European HbA1c market, a large installed base of equipment and over 20 years experience in HbA1c measurement.
We are also making significant progress in obtaining the necessary approvals in other key markets. In June we submitted our application to the Chinese regulatory authorities and we expect to receive approval by the end of 2012. This will represent an excellent opportunity to build on our existing installed base in China.
The Premier analyser is a best in class instrument with the following key advantages :
Results available in 1 minute, making it the fastest test in the market;
State of the art software using touchscreen technology to facilitate ease of use for operators;
Patented exclusive boronate affinity technology, therefore eliminating interference from hemoglobin variants;
Modular instrument which will significantly reduce the cost of on-site maintenance.
Commenting on the announcement, Trinity Biotech's CEO, Mr Ronan O'Caoimh, said, "HbA1c testing is already a US$300 million global market with enormous growth potential. We have now the fastest, most user friendly and state of the art HbA1c analyser in the world. FDA approval of the instrument, in conjunction with its recent CE marking, now gives us access to the US and European markets, the two most important individual markets in the world, and will accelerate the approval process in other key markets such as Brazil and China. Together with our partners, Menarini in Europe and Fisher in the USA, we are ideally positioned to become a significant and serious player in the global HbA1c market in the coming years."
Source: Globe Newswire (December 6, 2011 - 8:00 AM EST)
Antwort auf Beitrag Nr.: 42.447.313 von Vakataka am 06.12.11 23:00:09Trinity Biotech Announces Quarter 4 Financial Results
EPS Increases by 11.7% to 19.1 Cents Per ADR
DUBLIN, March 5, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended December 31, 2011.
Quarter 4 Results
Total revenues for Q4, 2011 were $20.0m which compares to $19.2m in Q4, 2010, representing an increase of 4%. This was primarily attributable to higher HIV sales in our two key markets of Africa and USA.
Point-of-care revenues for Q4, 2011 were $3.9m which is 12.4% higher than Q4, 2010. Clinical Laboratory revenues increased from $15.7m to $16.1m, which represents an increase of 2.1% compared to Q4, 2010. However, excluding Fitzgerald revenues, which fell by 12% in the quarter, the increase in our core diabetes/infectious diseases revenues was 6%.......
http://www.nasdaq.com/article/trinity-biotech-announces-quar…
EPS Increases by 11.7% to 19.1 Cents Per ADR
DUBLIN, March 5, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended December 31, 2011.
Quarter 4 Results
Total revenues for Q4, 2011 were $20.0m which compares to $19.2m in Q4, 2010, representing an increase of 4%. This was primarily attributable to higher HIV sales in our two key markets of Africa and USA.
Point-of-care revenues for Q4, 2011 were $3.9m which is 12.4% higher than Q4, 2010. Clinical Laboratory revenues increased from $15.7m to $16.1m, which represents an increase of 2.1% compared to Q4, 2010. However, excluding Fitzgerald revenues, which fell by 12% in the quarter, the increase in our core diabetes/infectious diseases revenues was 6%.......
http://www.nasdaq.com/article/trinity-biotech-announces-quar…
Antwort auf Beitrag Nr.: 42.854.514 von Vakataka am 05.03.12 22:31:15Moin!
Also, da muß ich mal sagen, dass mich dieser Jahresabschluß doch recht erfreut.
Das erste Mal seit langer Zeit habe ich das Gefühl, bei Trinity in einem Unternehmen investiert zu sein, was eine solide Perspektive hat. Hoffe, dass das Produktspektrum nun endlich gefunden wurde und das Wachstum, spez. im Profit noch ein bißchen beschleunigt wird.
Vielleicht hat sich der lange Atem ja doch gelohnt.
Also, da muß ich mal sagen, dass mich dieser Jahresabschluß doch recht erfreut.
Das erste Mal seit langer Zeit habe ich das Gefühl, bei Trinity in einem Unternehmen investiert zu sein, was eine solide Perspektive hat. Hoffe, dass das Produktspektrum nun endlich gefunden wurde und das Wachstum, spez. im Profit noch ein bißchen beschleunigt wird.
Vielleicht hat sich der lange Atem ja doch gelohnt.
Antwort auf Beitrag Nr.: 42.854.514 von Vakataka am 05.03.12 22:31:15Weil es so gut ist, kopiere ich das mal komplett rein
Trinity Biotech Announces Quarter 1 Financial Results
EPS of 19.4 Cents Per ADR - An Increase of 11%
DUBLIN, April 19, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended March 31, 2012.
Quarter 1 Results
Total revenues for Q1, 2012 were $20.0m which compares to $18.7m in Q1, 2011, an increase of 7.4%.
Point-Of-Care revenues for Q1, 2012 increased by 13.3% when compared to Q1, 2011. This increase was mainly attributable to increased HIV sales in Africa.
Clinical Laboratory revenues increased from $14.1m to $14.9m, which represents an increase of 5.5% compared to Q1, 2011. This included increased revenues from sales of the new Premier instrument, including the first placements in the USA.
Revenues for Q1, 2012 by key product area were as follows:
2011
Quarter 1 2012
Quarter 1
Increase
US$'000 US$'000 %
Point-of-Care 4,521 5,121 13.3%
Clinical Laboratory 14,133 14,905 5.5%
Total 18,654 20,026 7.4%
Gross profit for Q1, 2012 amounted to $10.3m, representing a gross margin of 51.6% which represents an improvement from the 51.2% for the same period in 2011. This improvement was due to increased Point-Of-Care sales, which traditionally have higher gross margins. However, this was partially offset by lower margins on increased sales of Premier instruments.
Research and Development expenses increased from $0.7m to $0.8m, representing an increase of 23%. Meanwhile Selling, General and Administrative (SG&A) expenses increased from $5.0m to $5.2m compared to Q1, 2011. This was mainly due to professional fees related to the acquisition of Fiomi Diagnostics AB during the quarter.
Operating Profit increased from $3.7m to over $4.1m for Q1, 2012 which was an increase of 11.7%. In the same period, operating margin increased from 19.8% to 20.6%.
Net financial income decreased slightly from $0.6m in Q1, 2011 to $0.5m this quarter.
Profit After Tax increased by almost 10% to $4.1m from $3.8m in the comparative period last year. Meanwhile, EPS for Q1, 2012 increased by 11% from 17.5 cents to 19.4 cents. The tax charge for Q1, 2012 was in line with the comparative period and represented an effective tax rate of 12.1%.
Free Cash Flows for the quarter were $1.4m and were impacted by the timing of revenue receipts and the anticipated increased working capital associated with moving to full production of the Premier instrument. Other significant cash movements in the quarter included cash payments of $5.6m related to the acquisition of Fiomi Diagnostics and share repurchases of $1m. The total cash balance at the end of the quarter was $65.5m and this will be increased by a further $11.25m following the receipt of the final deferred consideration payment from the Stago Group on April 30, 2012.
Recent Developments
The Company continued to sell its new Premier instrument, with sales during the quarter in Europe, Turkey, South America as well as the first placements in the USA. Overall, the launch of Premier is now gathering significant momentum. Having shipped 12 instruments in Q4, 2011, this increased to 31 instruments this quarter. With the increasing pace of sales in Europe and the USA, the number of instruments sold in Q2 can be expected to increase substantially. Further increases will be achieved when Chinese registration is obtained in Q4, 2012.
The Company announced it had obtained CE Marking and filed for FDA approval for its new point-of-care Uni-Gold™ Giardia test. This is the first of a range of new point-of-care tests to be developed at the Company's San Diego facility and will be followed by tests for C Difficile, Cryptosporidium, Syphilis, Strep pneumonia and Herpes by the end of 2012.
The Company continued its share buyback program during the quarter, repurchasing 97,000 ADRs at a cost of $1m.
During the quarter, Trinity completed the acquisition of Fiomi Diagnostics AB for $13.1m. This consisted of an upfront cash payment of $5.6m, 408,000 ADRs in Trinity Biotech and contingent consideration of $3.4m. Fiomi is at an advanced stage of developing a point-of-care test for Troponin I and other cardiac markers. The technology, which uses a micro-pillar flow technique, is capable of providing extremely sensitive, highly reproducible, quantitative, multiplexed results which give more accurate results than the current established point-of-care tests in the $900m cardiac market.
Comments
Commenting on the results, Kevin Tansley, Chief Financial Officer, said, "This quarter we have continued our growth trajectory in terms of both revenues and earnings. Revenues grew by 7% largely driven by increased HIV revenues and sales of our new A1c analyzer, the Premier. Meanwhile, profit after tax for the quarter increased to over $4m, thus resulting in an improvement of 11% in EPS to 19.4 cents."
Ronan O'Caoimh, CEO, stated, "We have had an extremely strong start to 2012. From a financial perspective we have continued to perform very well with the main highlights being the 13% growth in HIV revenues and the placement of our first Premier instruments in the USA.
"During the quarter, we acquired Fiomi Diagnostics AB, a Swedish company which is at an advanced stage in the development of a highly sensitive Troponin I test. This marks Trinity's entry into the highly lucrative and growing $900m cardiac market. We expect to be selling this product in Europe in the second half of 2013 and in the USA by the end of Q2, 2014, once FDA approval has been obtained. This Troponin I test will be followed by a test for BNP and a multiplex cardiac panel, thus giving a complete suite of cardiac products. The technology also has a broad range of non-cardiac applications.
"Also in the first quarter, we successfully launched the first of our new point-of-care products, a test for Giardia. This test was developed at our San Diego facility and will be followed by the launch of tests for C Difficile, Cryptosporidium, Syphilis, Strep pneumonia and Herpes by the end of the year."
Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
The Trinity Biotech plc logo is available at http://www.globenewswire.com/newsroom/prs/?pkgid=10602
Trinity Biotech plc
Consolidated Income Statements
(US$000's except share data)
Three Months Ended
March 31, 2012 Three Months Ended
March 31, 2011
(unaudited) (unaudited)
Revenues 20,026 18,654
Cost of sales (9,683) (9,097)
Gross profit 10,343 9,557
Gross profit % 51.6% 51.2%
Other operating income 175 297
Research & development expenses (845) (687)
Selling, general and administrative expenses (5,204) (5,046)
Indirect share based payments (337) (422)
Operating profit 4,132 3,699
Financial income 546 642
Financial expenses (1) (4)
Net financing income 545 638
Profit before tax 4,677 4,337
Income tax expense (567) (585)
Profit for the period 4,110 3,752
Earnings per ADR (US cents) 19.4 17.5
Diluted earnings per ADR (US cents) 18.6 16.9
Weighted average no. of ADRs used in computing basic earnings per ADR 21,217,683 21,388,026
Weighted average no. of ADRs used in computing diluted earnings per ADR 22,154,641 22,191,689
The above financial statements have been prepared in accordance with the principles of International Financial Reporting Standards and the Company's accounting policies but do not constitute an interim financial report as defined in IAS 34 (Interim Financial Reporting).
Trinity Biotech plc
Consolidated Balance Sheets
March 31,
2012
US$ '000
(unaudited) Dec 31,
2011
US$ '000
(audited)
ASSETS
Non-current assets
Property, plant and equipment 7,823 7,626
Goodwill and intangible assets 59,832 45,390
Deferred tax assets 3,034 2,977
Other assets 528 493
Total non-current assets 71,217 56,486
Current assets
Inventories 19,301 19,838
Trade and other receivables 25,677 23,973
Income tax receivable 271 117
Cash and cash equivalents 65,499 71,085
Total current assets 110,748 115,013
TOTAL ASSETS 181,965 171,499
EQUITY AND LIABILITIES
Equity attributable to the equity holders of the parent
Share capital 1,109 1,106
Share premium 3,086 2,736
Accumulated surplus 151,082 143,482
Other reserves 4,021 4,008
Total equity 159,298 151,332
Current liabilities
Interest-bearing loans and borrowings 70 108
Income tax payable 1,879 1,582
Trade and other payables 10,104 11,589
Provisions 50 50
Total current liabilities 12,103 13,329
Non-current liabilities
Other payables 3,273 10
Deferred tax liabilities 7,291 6,828
Total non-current liabilities 10,564 6,838
TOTAL LIABILITIES 22,667 20,167
TOTAL EQUITY AND LIABILITIES 181,965 171,499
The above financial statements have been prepared in accordance with the principles of International Financial Reporting Standards and the Company's accounting policies but do not constitute an interim financial report as defined in IAS 34 (Interim Financial Reporting).
Trinity Biotech plc
Consolidated Statement of Cash Flows
(US$000's) Three Months Ended March 31, 2012
(unaudited) Three Months Ended March 31, 2011
(unaudited)
Cash and cash equivalents at beginning of period 71,085 58,002
Operating cash flows before changes in working capital 5,115 4,773
Changes in working capital (1,821) 980
Cash generated from operations 3,294 5,753
Net Interest and Income taxes received 475 238
Capital Expenditure & Financing (net) (2,387) (2,105)
Free cash flow 1,382 3,886
Cash paid to acquire Phoenix Bio-tech (333) (1,000)
Cash paid to acquire Fiomi Diagnostics (5,624) --
Repurchase of own company shares (1,011) (1,070)
Cash and cash equivalents at end of period 65,499 59,818
CONTACT: Trinity Biotech plc
Kevin Tansley
(353)-1-2769800
E-mail: kevin.tansley@trinitybiotech.com
Lytham Partners LLC
Joe Diaz, Joe Dorame & Robert Blum
602-889-9700
Trinity Biotech PLC Logo
Source: Globe Newswire (April 19, 2012 - 7:36 AM EDT)
News by QuoteMedia

Trinity Biotech Announces Quarter 1 Financial Results
EPS of 19.4 Cents Per ADR - An Increase of 11%
DUBLIN, April 19, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced results for the quarter ended March 31, 2012.
Quarter 1 Results
Total revenues for Q1, 2012 were $20.0m which compares to $18.7m in Q1, 2011, an increase of 7.4%.
Point-Of-Care revenues for Q1, 2012 increased by 13.3% when compared to Q1, 2011. This increase was mainly attributable to increased HIV sales in Africa.
Clinical Laboratory revenues increased from $14.1m to $14.9m, which represents an increase of 5.5% compared to Q1, 2011. This included increased revenues from sales of the new Premier instrument, including the first placements in the USA.
Revenues for Q1, 2012 by key product area were as follows:
2011
Quarter 1 2012
Quarter 1
Increase
US$'000 US$'000 %
Point-of-Care 4,521 5,121 13.3%
Clinical Laboratory 14,133 14,905 5.5%
Total 18,654 20,026 7.4%
Gross profit for Q1, 2012 amounted to $10.3m, representing a gross margin of 51.6% which represents an improvement from the 51.2% for the same period in 2011. This improvement was due to increased Point-Of-Care sales, which traditionally have higher gross margins. However, this was partially offset by lower margins on increased sales of Premier instruments.
Research and Development expenses increased from $0.7m to $0.8m, representing an increase of 23%. Meanwhile Selling, General and Administrative (SG&A) expenses increased from $5.0m to $5.2m compared to Q1, 2011. This was mainly due to professional fees related to the acquisition of Fiomi Diagnostics AB during the quarter.
Operating Profit increased from $3.7m to over $4.1m for Q1, 2012 which was an increase of 11.7%. In the same period, operating margin increased from 19.8% to 20.6%.
Net financial income decreased slightly from $0.6m in Q1, 2011 to $0.5m this quarter.
Profit After Tax increased by almost 10% to $4.1m from $3.8m in the comparative period last year. Meanwhile, EPS for Q1, 2012 increased by 11% from 17.5 cents to 19.4 cents. The tax charge for Q1, 2012 was in line with the comparative period and represented an effective tax rate of 12.1%.
Free Cash Flows for the quarter were $1.4m and were impacted by the timing of revenue receipts and the anticipated increased working capital associated with moving to full production of the Premier instrument. Other significant cash movements in the quarter included cash payments of $5.6m related to the acquisition of Fiomi Diagnostics and share repurchases of $1m. The total cash balance at the end of the quarter was $65.5m and this will be increased by a further $11.25m following the receipt of the final deferred consideration payment from the Stago Group on April 30, 2012.
Recent Developments
The Company continued to sell its new Premier instrument, with sales during the quarter in Europe, Turkey, South America as well as the first placements in the USA. Overall, the launch of Premier is now gathering significant momentum. Having shipped 12 instruments in Q4, 2011, this increased to 31 instruments this quarter. With the increasing pace of sales in Europe and the USA, the number of instruments sold in Q2 can be expected to increase substantially. Further increases will be achieved when Chinese registration is obtained in Q4, 2012.
The Company announced it had obtained CE Marking and filed for FDA approval for its new point-of-care Uni-Gold™ Giardia test. This is the first of a range of new point-of-care tests to be developed at the Company's San Diego facility and will be followed by tests for C Difficile, Cryptosporidium, Syphilis, Strep pneumonia and Herpes by the end of 2012.
The Company continued its share buyback program during the quarter, repurchasing 97,000 ADRs at a cost of $1m.
During the quarter, Trinity completed the acquisition of Fiomi Diagnostics AB for $13.1m. This consisted of an upfront cash payment of $5.6m, 408,000 ADRs in Trinity Biotech and contingent consideration of $3.4m. Fiomi is at an advanced stage of developing a point-of-care test for Troponin I and other cardiac markers. The technology, which uses a micro-pillar flow technique, is capable of providing extremely sensitive, highly reproducible, quantitative, multiplexed results which give more accurate results than the current established point-of-care tests in the $900m cardiac market.
Comments
Commenting on the results, Kevin Tansley, Chief Financial Officer, said, "This quarter we have continued our growth trajectory in terms of both revenues and earnings. Revenues grew by 7% largely driven by increased HIV revenues and sales of our new A1c analyzer, the Premier. Meanwhile, profit after tax for the quarter increased to over $4m, thus resulting in an improvement of 11% in EPS to 19.4 cents."
Ronan O'Caoimh, CEO, stated, "We have had an extremely strong start to 2012. From a financial perspective we have continued to perform very well with the main highlights being the 13% growth in HIV revenues and the placement of our first Premier instruments in the USA.
"During the quarter, we acquired Fiomi Diagnostics AB, a Swedish company which is at an advanced stage in the development of a highly sensitive Troponin I test. This marks Trinity's entry into the highly lucrative and growing $900m cardiac market. We expect to be selling this product in Europe in the second half of 2013 and in the USA by the end of Q2, 2014, once FDA approval has been obtained. This Troponin I test will be followed by a test for BNP and a multiplex cardiac panel, thus giving a complete suite of cardiac products. The technology also has a broad range of non-cardiac applications.
"Also in the first quarter, we successfully launched the first of our new point-of-care products, a test for Giardia. This test was developed at our San Diego facility and will be followed by the launch of tests for C Difficile, Cryptosporidium, Syphilis, Strep pneumonia and Herpes by the end of the year."
Litigation Reform Act of 1995. Investors are cautioned that such forward-looking statements involve risks and uncertainties including, but not limited to, the results of research and development efforts, the effect of regulation by the United States Food and Drug Administration and other agencies, the impact of competitive products, product development commercialisation and technological difficulties, and other risks detailed in the Company's periodic reports filed with the Securities and Exchange Commission.
Trinity Biotech develops, acquires, manufactures and markets diagnostic systems, including both reagents and instrumentation, for the point-of-care and clinical laboratory segments of the diagnostic market. The products are used to detect infectious diseases and to quantify the level of Haemoglobin A1c and other chemistry parameters in serum, plasma and whole blood. Trinity Biotech sells direct in the United States, Germany, France and the U.K. and through a network of international distributors and strategic partners in over 75 countries worldwide. For further information please see the Company's website: www.trinitybiotech.com.
The Trinity Biotech plc logo is available at http://www.globenewswire.com/newsroom/prs/?pkgid=10602
Trinity Biotech plc
Consolidated Income Statements
(US$000's except share data)
Three Months Ended
March 31, 2012 Three Months Ended
March 31, 2011
(unaudited) (unaudited)
Revenues 20,026 18,654
Cost of sales (9,683) (9,097)
Gross profit 10,343 9,557
Gross profit % 51.6% 51.2%
Other operating income 175 297
Research & development expenses (845) (687)
Selling, general and administrative expenses (5,204) (5,046)
Indirect share based payments (337) (422)
Operating profit 4,132 3,699
Financial income 546 642
Financial expenses (1) (4)
Net financing income 545 638
Profit before tax 4,677 4,337
Income tax expense (567) (585)
Profit for the period 4,110 3,752
Earnings per ADR (US cents) 19.4 17.5
Diluted earnings per ADR (US cents) 18.6 16.9
Weighted average no. of ADRs used in computing basic earnings per ADR 21,217,683 21,388,026
Weighted average no. of ADRs used in computing diluted earnings per ADR 22,154,641 22,191,689
The above financial statements have been prepared in accordance with the principles of International Financial Reporting Standards and the Company's accounting policies but do not constitute an interim financial report as defined in IAS 34 (Interim Financial Reporting).
Trinity Biotech plc
Consolidated Balance Sheets
March 31,
2012
US$ '000
(unaudited) Dec 31,
2011
US$ '000
(audited)
ASSETS
Non-current assets
Property, plant and equipment 7,823 7,626
Goodwill and intangible assets 59,832 45,390
Deferred tax assets 3,034 2,977
Other assets 528 493
Total non-current assets 71,217 56,486
Current assets
Inventories 19,301 19,838
Trade and other receivables 25,677 23,973
Income tax receivable 271 117
Cash and cash equivalents 65,499 71,085
Total current assets 110,748 115,013
TOTAL ASSETS 181,965 171,499
EQUITY AND LIABILITIES
Equity attributable to the equity holders of the parent
Share capital 1,109 1,106
Share premium 3,086 2,736
Accumulated surplus 151,082 143,482
Other reserves 4,021 4,008
Total equity 159,298 151,332
Current liabilities
Interest-bearing loans and borrowings 70 108
Income tax payable 1,879 1,582
Trade and other payables 10,104 11,589
Provisions 50 50
Total current liabilities 12,103 13,329
Non-current liabilities
Other payables 3,273 10
Deferred tax liabilities 7,291 6,828
Total non-current liabilities 10,564 6,838
TOTAL LIABILITIES 22,667 20,167
TOTAL EQUITY AND LIABILITIES 181,965 171,499
The above financial statements have been prepared in accordance with the principles of International Financial Reporting Standards and the Company's accounting policies but do not constitute an interim financial report as defined in IAS 34 (Interim Financial Reporting).
Trinity Biotech plc
Consolidated Statement of Cash Flows
(US$000's) Three Months Ended March 31, 2012
(unaudited) Three Months Ended March 31, 2011
(unaudited)
Cash and cash equivalents at beginning of period 71,085 58,002
Operating cash flows before changes in working capital 5,115 4,773
Changes in working capital (1,821) 980
Cash generated from operations 3,294 5,753
Net Interest and Income taxes received 475 238
Capital Expenditure & Financing (net) (2,387) (2,105)
Free cash flow 1,382 3,886
Cash paid to acquire Phoenix Bio-tech (333) (1,000)
Cash paid to acquire Fiomi Diagnostics (5,624) --
Repurchase of own company shares (1,011) (1,070)
Cash and cash equivalents at end of period 65,499 59,818
CONTACT: Trinity Biotech plc
Kevin Tansley
(353)-1-2769800
E-mail: kevin.tansley@trinitybiotech.com
Lytham Partners LLC
Joe Diaz, Joe Dorame & Robert Blum
602-889-9700
Trinity Biotech PLC Logo
Source: Globe Newswire (April 19, 2012 - 7:36 AM EDT)
News by QuoteMedia
Antwort auf Beitrag Nr.: 43.062.274 von Vakataka am 19.04.12 18:47:21Da kann man ja schon mal zufrieden sein, mit dem ersten Quartal
Jedes Quartal 11% drauf und der Kurs geht dahin wo ich ihn gerne hätte.

Jedes Quartal 11% drauf und der Kurs geht dahin wo ich ihn gerne hätte.

Antwort auf Beitrag Nr.: 43.064.917 von Lubus104 am 20.04.12 11:16:31hoffentlich fliegt der Deckel bei 12 US-D auch bald. The sky is open.

Antwort auf Beitrag Nr.: 43.075.671 von Dangerous am 23.04.12 15:19:22
Trinity Biotech Announces an Annual Dividend of 15 Cents Per ADR -- an Increase of 50%
DUBLIN, Ireland, April 25, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced a proposed dividend of 15 cents per ADR.
In 2011, Trinity announced the commencement of an annual dividend policy and paid a dividend of 10 cents per ADR. This year the Company is proposing a dividend of 15 cents per ADR, representing an increase of 50%. The payment of this dividend is subject to shareholder approval, which will be sought at the Company's forthcoming AGM to be held on May 25, 2012. Subject to this approval being granted, the record date will be June 8, 2012 and payment will follow 2 weeks later.

Trinity Biotech Announces an Annual Dividend of 15 Cents Per ADR -- an Increase of 50%
DUBLIN, Ireland, April 25, 2012 (GLOBE NEWSWIRE) -- Trinity Biotech plc (Nasdaq:TRIB), a leading developer and manufacturer of diagnostic products for the point-of-care and clinical laboratory markets, today announced a proposed dividend of 15 cents per ADR.
In 2011, Trinity announced the commencement of an annual dividend policy and paid a dividend of 10 cents per ADR. This year the Company is proposing a dividend of 15 cents per ADR, representing an increase of 50%. The payment of this dividend is subject to shareholder approval, which will be sought at the Company's forthcoming AGM to be held on May 25, 2012. Subject to this approval being granted, the record date will be June 8, 2012 and payment will follow 2 weeks later.
Antwort auf Beitrag Nr.: 43.092.886 von Vakataka am 26.04.12 17:43:27Quartalzahlen folgen diese Woche und der Kurs hat schon mal an der 12 US-D Marke gekratzt. Hoffentlich fällt diese Hürde bald.
http://finance.yahoo.com/news/trinity-biotech-plc-announce-s…
http://finance.yahoo.com/news/trinity-biotech-plc-announce-s…
Antwort auf Beitrag Nr.: 43.365.714 von Dangerous am 09.07.12 12:31:08Umsätze weiter gesteigert. Der Gewinn ist auf 0,20 US-D für das Q2 gestiegen. Halbjahresgewinn 0,39 US-D. Damit sollte meine Schätzung von 0,80 US-D Jahresgewinn übertroffen werden. Der nächste charttecnischer Widerstand liegt bei 15 US-D.
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Antwort auf Beitrag Nr.: 43.382.117 von Dangerous am 13.07.12 11:00:53neuer Test geht an den Start. Der Kurs ist endlich nach oben ausgebrochen!!
Antwort auf Beitrag Nr.: 43.382.117 von Dangerous am 13.07.12 11:00:53Erzähl mir mal was über das Unternehmen. War auf der Site. Habe Analystenmeinungen eingeholt.
Scheint so zu sein, dass man mit Glück 16 bis 18 Dollar erwarten kann. Ist natürlich etwas wenig und setzt Gewinne in der von Dir genannten Größenordnung voraus. Gehe ich rein, dann mit einer ganzen Menge und deshalb hätte ich es gerne, wenn die Phantasie auch 30 Dollar hergeben würde und entsprechende Umsätze und Gewinne generiert werden könnten. Also, was ist an dem Unternhemen so besonders. Mich hat der Chart aufmerksam gemacht, sonst ersteinmal nichts. Im Moment geht der Pharmasektor, nur meiner Meinung nach ist es übertrieben.
Scheint so zu sein, dass man mit Glück 16 bis 18 Dollar erwarten kann. Ist natürlich etwas wenig und setzt Gewinne in der von Dir genannten Größenordnung voraus. Gehe ich rein, dann mit einer ganzen Menge und deshalb hätte ich es gerne, wenn die Phantasie auch 30 Dollar hergeben würde und entsprechende Umsätze und Gewinne generiert werden könnten. Also, was ist an dem Unternhemen so besonders. Mich hat der Chart aufmerksam gemacht, sonst ersteinmal nichts. Im Moment geht der Pharmasektor, nur meiner Meinung nach ist es übertrieben.
Morgen kommen die Q4-Zahlen. Der Kurs ist schon sehr gut gelaufen. In der Vergangeheit gab es immer ein Rücksetzer mit Bekanntgabe der Q-Zahlen, wenn der Kurs im Vorfeld gut gelaufen ist. Mal sehen, wie das Ergebnis aussieht und welche Fanatasien geweckt werden.
@sukraman
Ich bin kein Analyst. Seit 10 Jahren halte ich die Aktie und habe alle Höhen und Tiefen mitgemacht. So langsam rückt die Gewinnzone in greifbare Nähe. Die Dividende versüsst jetzt das lange warten.
@sukraman
Ich bin kein Analyst. Seit 10 Jahren halte ich die Aktie und habe alle Höhen und Tiefen mitgemacht. So langsam rückt die Gewinnzone in greifbare Nähe. Die Dividende versüsst jetzt das lange warten.
Mir geht es genau so. Aber so langsam gibt es doch ein wenig Licht am Ende des Tunnels. Diese Analyse macht doch auch Hoffnung:
http://www.trinitybiotech.com/Pages/News.aspx?NewsID=40
http://www.trinitybiotech.com/Pages/News.aspx?NewsID=40
Antwort auf Beitrag Nr.: 44.217.067 von westix am 05.03.13 18:08:04sehr interessante Analyse. Sie sollten die Liquidität nutzen und weitere Aktien zurückkaufen. Dann steigt automatisch das EPS. Kursziel 24 Dollar klingt gut.
Na . das fängt ja an Spaß zu machen. Aktienrückkauf und Bereinigung des Portefolio haben ja was gebracht. 0,81 Cent/ADR, damit haben wir das 20fache an Bewertung, finde das geht noch bei dem Geschäft und den Aussichten. Werde die Aktie weiterhalten und traue ihr 25 Usd zu. Was mir gefällt, dass bei Rücksetzern des Nasdaq (zugegeben, gabs schon lange nicht mehr) die Aktie nicht mitgeht.
in den letzten 2 Handelstagen gelang endlich ein deutlicher Move nach oben. Die Indikatoren weisen schon eine deutliche Überhitzung an. Etwas Luft ist noch vorhanden. Knacken wir jetzt das Zwischenhoch aus März 2013 bei 19 US-D? Die Bandbreite 19-20 US-D stellt ein massiver Widerstand dar. Darüber könnte es zum fünften Berg seit 1993 kommen? Am Freitag kommen die Zahlen. Die müssen dann schon sehr gut ausfallen, damit der Kurs über 20 US-D ausbrechen kann. Dividende ist vor kurzen auch geflossen. Zahlt sich mein Investment nach 10 Jahren endlich aus? Es wäre schön.
Termin für die Q-Zahlen wurde auf yahoo auf den 26.07. geändert. Leider hat die Aktie exakt unter 19 US-D den Rückwärtsgang eingelegt. Hoffentlich können die Q-Zahlen überzeugen, so dass die Aktien den Widerstand überwindet? We will see
bei deutlich anziehenden Umsätzen wurde der Widerstand bei 19 US-D geknackt. Mal sehen, ob die 20 US-D Marke auch in den nächsten Tagen fällt. Ich bin auf die Q-Zahlen gespannt!
jetzt sollen die Zahlen erst am 30.07. kommen!
http://finance.yahoo.com/news/trinity-biotech-plc-announce-s…
Gestern wurde die 20 US-D Marke übersprungen. Nachbörslich leider auf 19,80 gefallen. Wo geht die Reise hin?
http://finance.yahoo.com/news/trinity-biotech-plc-announce-s…
Gestern wurde die 20 US-D Marke übersprungen. Nachbörslich leider auf 19,80 gefallen. Wo geht die Reise hin?
leichter Umsatzanstieg, Gewinn rückläufig u.a. aufgrund der Akquisation von Immco Diagnostics Inc, davon hatte ich bis dato garnichts gehört :-( Die Zahlen wecken erstmal keine Fantasie, um die 20 US-D zu knacken. Vielleicht folgen im Conferenc Call noch Infos zur Übernahme und den weiteren Perspektiven?
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Antwort auf Beitrag Nr.: 45.143.941 von Dangerous am 30.07.13 17:19:20geschafft! Gestern 21 US-D. Damit rückt der nächste Widerstand bei 23-24 US-D in Reichweite. Mal sehen, wie es weitergeht.
Antwort auf Beitrag Nr.: 45.204.289 von Dangerous am 08.08.13 09:03:32Ja, der Kurs zeigt eine Super-Entwicklung! Aber eine Frage habe ich an dich, wie kommst du auf die Widerstände. Nach meiner Kenntnis sind wir charttechnisch doch im Niemandsland, sprich Alltime-High.
Beste Grüße
Beste Grüße
Antwort auf Beitrag Nr.: 45.204.333 von Lubus104 am 08.08.13 09:08:51bitte auf comdirect gehen und Trinity Biotech bei der Kurssuche eingeben. Als Börsenplatz bitte Nasdaq auswählen und beim Chart auf das unendlich Symbol klicken. All-Time-High liegt bei 30 US-D
Antwort auf Beitrag Nr.: 45.214.239 von Dangerous am 09.08.13 09:21:06Tatsache! im Jahr 2000 (während des Neue Markt Hypes?) war der Preis tatsächlich bei fast 30$.
Aktie war aber anscheinend noch nicht in D platziert.
Vielen Dank für die Info.
Vielleicht erreichen wir ja die 30$ noch, schließlich ist Trinity in der Zeit kräftig gewachsen und auf Einkaufstour gewesen.
Aktie war aber anscheinend noch nicht in D platziert.
Vielen Dank für die Info.

Vielleicht erreichen wir ja die 30$ noch, schließlich ist Trinity in der Zeit kräftig gewachsen und auf Einkaufstour gewesen.
Antwort auf Beitrag Nr.: 45.215.509 von Lubus104 am 09.08.13 11:28:47mal sehen, was die Quartalszahlen bringen. Geht es weiter nach oben? Knacken wir die 23 US-D dann ist der Weg bis 25 US-D frei. Anschließend wäre noch das All-Time-High bei 30 US-D offen!
Antwort auf Beitrag Nr.: 45.642.835 von Dangerous am 17.10.13 09:29:1615 % Umsatzwachstum. Die Übernahme Q2 trägt bereits zum Wachstum bei. Mit den Quartalszahlen wurde eine weitere Übernahme gemeldet. Durch die Kaufpreiszahlung ist ein Verlust im aktuellen Quartal entstanden. Die neuen Produkte bieten Potential. Allerdings finde ich den Kaufpreis für die Übernahme zu hoch. Q-Zahlen wurden aber vom Markt gut aufgenommen. 25 US-D rücken in Sichtweite.
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Antwort auf Beitrag Nr.: 45.650.315 von Dangerous am 18.10.13 08:26:34der Kurs ist schon sehr gut gelaufen. Hoffentlich geht es über 25 US-D! Der Kurs ist schon stark überkauft, aber die Faktoren sind noch nicht ganz ausgereizt. Wenn man sich die Peaks der letzten 15 Jahre ansieht, dann wäre noch etwas Platz nach oben. We will see
geschafft. Der Kurs ist gerade auf 25,05 US-D gestiegen. Entscheidend wird der Schlußkurs sein. Beim nachhaltigen Bruch ist der Weg zum All-Time-High bis 30 US-D offen. Dann werde ich wahrscheinlich nach 10 Jahren aussteigen. Ich warte sicher noch die Ergebnisse des Q4 ab und entscheide dann. Das KGV auf Basis meiner Schätzung für 2014 von 0,85-0,95 beträgt aktuell 29-25 bei einem aktuellen Umsatzwachstum von 15 % etwas zu hoch. Wie das Umsatzwachstum durch die weiteren Übernahmen im Q4 ausfällt, bleibt abzuwarten. Durch die Übernahme wurde ja im Q3 ein Verlust generiert. Daher kann das Gewinnwachstum nicht eingeschätzt werden.
Schlußkurs genau 25,00 US-D. Hoffentlich springen wir drüber. Der Kurs ist schon sehr stark überkauft. Ich habe mir noch einmal die einzelnen Kommentierungen zum letzten Quartalsbericht durchgelesen. Diese sind doch sehr vielversprechend HV-Test 2. Hier wird von einer Verdoppelung der Umsätze 2014 ausgegangen. Die Aussichten im Bereich Laboratorium sind auch sehr gut. Es wird von einem Wachstum von fast 50 % ausgegangen. Auch der neue Malaria Test und weitere zugekaufte Umsätze versprechen ein starkes Wachstum 2014. Wahrscheinlich ist meine Schätzung für den Gewinn 2014 viel zu konservativ. Ich warte auf jeden Fall das Q-Erbnis Q4 ab. Mal sehen, wie dann das Umsatz- und Gewinnwachstum aussieht.
Antwort auf Beitrag Nr.: 45.740.909 von Dangerous am 01.11.13 13:07:04Danke, dass du immer mal wieder etwas Aktuelles zur Aktie schreibst.
Kursverlauf ist doch sehr vielversprechend und ich könnte mir vorstellen, dass die Aktie erstmal weiterläuft, auch wenn das KGV schon an die 30 reicht.
Aber es kommt doch mehr und mehr Fantasie in den Wert und die Aufmerksamkeit steigt.
Kursverlauf ist doch sehr vielversprechend und ich könnte mir vorstellen, dass die Aktie erstmal weiterläuft, auch wenn das KGV schon an die 30 reicht.
Aber es kommt doch mehr und mehr Fantasie in den Wert und die Aufmerksamkeit steigt.
Antwort auf Beitrag Nr.: 45.753.187 von Lubus104 am 04.11.13 10:23:39noch lassen die Chartindikatoren einen weiteren Anstieg zu, obwohl die Aktie schon stark überkauft ist. Wenn jetzt keine Konsolidierung folgt, dann könnte ich mir auch ein schnelles ansteigen bis 30 US-D vorstellen. Mal sehen, wie es weitergeht.
Antwort auf Beitrag Nr.: 45.755.705 von Dangerous am 04.11.13 15:23:55Mal etwas Anderes, an welcher Börse würdest du die Aktie handeln?
In D sind die Umsätze ja eher null
In D sind die Umsätze ja eher null

Antwort auf Beitrag Nr.: 45.756.767 von Lubus104 am 04.11.13 17:08:38da ich vor 10 Jahren gekauft habe und bisher ein Verkauf aifgrund des Kursverlaufs nicht in Frage kam, habe ich mich damit nicht beschäftgt. Der Handelsumsatz in Frankfurt ist noch am Größten. Mit einem direkten Kauf/Verkauf an der Nasdaq habe ich mich noch nicht befasst.
der MACD steht kurz vor einem Verkaufssignal. Auch alle anderen Indikatoren laufen bereits leicht nach unten. Da der Kurs nicht nachhaltig über 25 US-D springt, zeigt auch eine möglichen Konsolidierung an. Hoffentlich bewegt sich der Kurs nur seitwärts!
Antwort auf Beitrag Nr.: 45.792.048 von Dangerous am 08.11.13 13:20:11bleibt es bei der kleinen Konsolidierung oder geht es weiter runter? Wenn der Deckel bei 25,50 UD-D fliegt, dann könnte der Kurs auf 30 US-D steigen. Derzeit zeigt der Kurs leider im Verhältnis zum Gesamtmarkt einige Schwächen auf.
Antwort auf Beitrag Nr.: 45.894.688 von Dangerous am 22.11.13 11:43:20Ich denke ja, dass der allgemeine Börsenverlauf nur beschränkte Auswirkungen auf die Kursentwicklung von Trinity hat, da es sich um einen Nebenwert handelt.
Vielleicht gibt es den nächsten Schub erst, wenn die großen Werte etwas zurückgehen. We`ll see.
Aber ein bißchen Anlauf braucht sie wohl um durch die 25 US-D zu gehen.
Vielleicht gibt es den nächsten Schub erst, wenn die großen Werte etwas zurückgehen. We`ll see.
Aber ein bißchen Anlauf braucht sie wohl um durch die 25 US-D zu gehen.
Antwort auf Beitrag Nr.: 45.898.324 von Lubus104 am 22.11.13 17:25:51leider wurden die 25 US-D nach unten gebrochen. Jetzt sollte eine weitere Konsolidierung folgen. Der RSI ist bereits relativ tief (40) - MehrjahresTiefs bei 30 und Ausnahme 20. Der MACD und verschiedene Oscillatoren zeigen weiteres begrenztes Abwärtspotential an. Bei 23 US-D ist eine gute Auffanglinie vorhanden. Mal sehen, wie es weiter geht?
Antwort auf Beitrag Nr.: 45.984.710 von Dangerous am 05.12.13 11:40:01der Kurs ist unter den unteren Rand des Bollinger-Bandes gefallen. Es könnte eine kleine Zwischenerholung folgen. Ansonsten hat auch der Aroon Oscillator in den negativen Bereich gedreht. Die Konsolidierung sollte erstmal tendenziell Bestand haben. Vielleicht kommt auch mal wieder etwas Fundamentales vom Unternehmen als Adhoc zwischendurch (nicht nur das Quartalsergebnis)!
Antwort auf Beitrag Nr.: 46.008.996 von Dangerous am 09.12.13 10:33:54gestern Intraday bereits fast die 23 US-D angetestet. Noch ist die Bodenbildung nicht in Sicht, aber die Indikatoren laufen jetzt stark in den überverkauften Bereich.
Antwort auf Beitrag Nr.: 46.016.548 von Dangerous am 10.12.13 08:35:55leider ist der Kurs unter 23 US-D gefallen. Es scheint sich jetzt ein Tief anzudeuten (mögliche W-Formation). Der Kurs sollte allerdings nicht unter das Oktober-Tief bei etwa 21,50 US-D fallen. Dann könnte sich eine SKS-Formation herausbilden und der Kurs weiter fallen. Ich hoffe auf eine Stabilisierung. Sicher hängt auch einiges von der morgigen FED-Entscheidung ab. Sollte das Tapering geringer als erwartet ausfallen, dann könnte es wieder aufwärts gehen. We will see. Die Aussichten für TRIB für das Jahr 2014 sind sehr gut.
Antwort auf Beitrag Nr.: 46.064.208 von Dangerous am 17.12.13 13:20:34die W-Formation ist abgeschlossen und der Kurs deutlich über 23 US-D gesprungen. Es sieht charttechnisch wieder besser aus.
Antwort auf Beitrag Nr.: 46.082.056 von Dangerous am 19.12.13 13:23:25die Charttechnik sieht vielversprechend aus. Der MACD hat ein Kaufsignal generiert. Der RSI ist auch wieder nach oben gedreht und hat noch weiter Luft nach oben. Gestern wurde ein neues 52 Wochen-Hoch erreicht. Der Weg nach oben ist momentan wieder frei. Mal sehen, wie weit der Kurs noch läuft? Das All-Time-High bei 30 US-D wäre die nächste Barriere für die nächsten Monate. Die Quartalszahlen folgen erst Anfang März. Bis dahin wird es wohl keine fundamentalen News geben. Dies ist nicht als Kaufempfehlung zu verstehen. Nur meine Gedanke! Meine Tendenz geht nach 11 Jahren Investment Richtung Verkauf.
Antwort auf Beitrag Nr.: 46.171.755 von Dangerous am 07.01.14 12:32:13die Rakete zündet heute. Ich bereite meinen Ausstieg vor :-)
Antwort auf Beitrag Nr.: 46.193.613 von Dangerous am 09.01.14 16:52:36Leider habe ich mein Verkaufslimit zu hoch angesetzt (21 EUR). Der US-D Chart hat leider auch ein Verkaufssignal ausgelöst (MACD). Dann hoffe ich, dass die FED nächste Woche nur einen kleinen Tapering-Schritt verkündet. Dann sollte es in New York wieder aufwärts gehen. Die Performance im Januar sieht bei den US Indezes nicht so gut aus. Die sogenannte Januar-Regel ist ja recht verlässlich. Bei einem negativen Januar gibt es auch ein negatives Aktienjahr. Zumeist drehen die US Börsen allerdings Ende Januar nach oben. Na mal sehen, wie es weiter geht. Auf fundamentale Daten von Trib müssen wir noch etwas warten.
mittlerweile ist der Kurs auf 25 US-D gefallen. Dort befindet sich auch die 38/50 Tage-Linie. Der RSI ist auch bereits recht tief. Auch der Dow Jones ist bereits stark gefallen. Endet morgen die Korrektur mit der FED-Sitzung oder geht diese weiter? We will see
endlich fundamentale Nachrichten. Kurs ist gleich gestiegen. Unter 25 US-D würde sich das Chartbild deutlich eintrüben:
http://finance.yahoo.com/news/trinity-biotech-announces-euro…
http://finance.yahoo.com/news/trinity-biotech-announces-euro…
die charttechnische Situation hat sich verbessert. Sollte der Kurs über 26 US-D springen, dann wäre die Konsolidierung mit einer klassischen W-Formation abgeschlossen.
Quartalszahlen sind raus. Umsatzentwicklung Q 4 sehr gut!
http://finance.yahoo.com/news/trinity-biotech-announces-resu…
http://finance.yahoo.com/news/trinity-biotech-announces-resu…
im Rahmen der Konsolidierung hat die Unterstützung bei 22 US-D gehalten. Der Widerstand bei 25 US-D wurde heute angetestet. Morgen folgen die Quartalszahlen. Mal sehen, ob mit den Quartalszahlen ein Sprung darüber möglich ist. Zumeist wurde in der Vergangeheit nach einem Kursanstieg vor den Quartalszahlen, die Aktie am Tag der Veröffentlichung abverkauft. Sell the facts. Hoffentlich fällt die Kursreaktion morgen anders aus. Dann rückt wieder die Marke 29-30 US-D in den Fokus. We will see
gutes Umsatzwachstum, der Gewinn enttäuscht, wahrscheinlich wird der Gewinn erst im Q3 steigen
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Roth Capital hat das Rating auf kaufen mit Kursziel 30 US-D erhöht
Charttechnisch sieht es gut aus, wenn der Widerstand bei 25 US-D nachhaltig überwunden wird.
Charttechnisch sieht es gut aus, wenn der Widerstand bei 25 US-D nachhaltig überwunden wird.
Antwort auf Beitrag Nr.: 46.909.656 von Dangerous am 02.05.14 10:17:51charttechnisch wird es jetzt eng. Wenn die 50 Tage Linie die 200 Tage Linie kreuzt, spricht man wohl vom Todeskreuz. Sollten der Kurs nicht endlich die Marke von 24,50-25,00 US-D brechen, dann steige ich aus. Fundamental sieht es eigentlich gut aus.
sah es letzten Donnerstag charttechnisch sehr gut aus. Alle Indikatoren drehten und lösten Kaufsignale aus. Das Bollinger-Band wurde leicht nach oben verlassen, haben 2 Minus-Tage die Charttechnik wieder ins Gegenteil verkehrt. Hoffentlich stabilisiert sich der Kurs bei 23 US-D. Ich hoffe, dass mit den Quartalszahlen Ende Juli etwas Schwung in die Aktie kommt. Die Fundamental-Daten sollten gut ausfallen.
der Kurs hält sich bei ca. 23 US-D. Ich hoffe, dass die Quartalszahlen morgen überzeugen können
Umsatzwachstum sehr gut +22 % Gewinn +13 % (Kosten für Akquisationen und Marketing), gute Aussichten für den Roll-out weitere Produkte, vorbörslich mit 2,5 % im Plus, charttechnische Widerstände ca. 23,70 US-D oberer Rand des Bollinger Bandes, 24,55 US-D 200 Tage-Linie sowie 25 US-D oberer Rand des Seitwärtstrends seit März
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
ein Teil der Gewinne wurde gestern wieder abverkauft. Die Indikatoren stehen alle kurz vor einem Kaufsignal. Mal sehen, wie der Markt auf die FED-Sitzung reagiert. Dann entscheidet sich der weitere Trend. Tapering wird sicher fortgesetzt (Reduzierung auf 25 MRD US-D). Entscheidend wird sicher die Information zu möglichen Zinserhöhungen in 2015 sein. Wenn die FED die Märkte beruhigt, dass Sie noch die Zinsen geraume Zeit niedrig lassen wird, dann wird der Markt seine Seitwärts/Aufwärtsbewegung fortsetzen. Das sollte dann auch gut für TRIB sein.
so FED im Rahmen der Erwartungen. Mit dem Zusatz, dass sich die Inflation an das obere Ziel annähert, wurde die Befürchtung früher steigender Zinsen genähert. So dann kam noch die Argentinien-Pleite und schwache Unternehmensberichte aus DT hinaus. Der Markt rauscht runter und oh Wunder TRIB hält sich sehr gut. Ich habe jetzt eine Verkaufsorder zu 17 EUR platziert. Ansonsten sind die fundamentalen Aussichten bei TRIB gut.
die Marke von 23 US-D wackelt. Mal sehen, ob die Märkte diese Woche eine kleine Erholung starten und wie sich Trib dabei schlägt
in der Zwischenzeit wurde die Aktie heftig abverkauft. Die Quartalszahlen gestern kamen nicht gut an. Der Quartalsgewinn war leicht rückläufig. Der Neumonatsgewinn liegt nur noch leicht über dem Vorjahr. Die Umsatzsteigerung ist sehr gut. Jetzt wäre es an der Zeit, dass die Gewinne auch wieder steigen. Die Übernahmen sollten ja auch langsam verdaut sein. Hier die Meldung:
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Die Probleme bei der klinischen Studien für Troponin lesen sich nicht gut. Das Problem lag bei einem Zulieferer. Das Problem wurde erkannt und abgestellt. Der Lieferant für den chemischen Grundstoff liefert jetzt wieder die ursprüngliche Zusammensetzung.
Data compilation and cardiologist adjudication will be completed during the months of June and July and it is anticipated that a final submission will be made in August, 2015. We confidently believe that the product will receive FDA clearance thereafter.
Bereits im Q3 kamen dann auch Umsätze aus der Übernahme der Imco (Augentest):
Following a successful partial launch in a number of test markets earlier this year, it received its nationwide launch late in June, 2014 thus making Q3, 2014 effectively the first full quarter of sales. To date the test has been well received in the market and revenues to date have been exceeding expectations.
Durch Schließeung der Blutbank in England und den Marketingkosten für den Augentest fielen höhere Kosten an:
The impact of these factors during the quarter was approximately $0.4m, which if they were to be excluded would have resulted in profits of $4.8m and EPS of 21 cents".
Das Q4 sollte deutlich höhere Gewinne ausweisen:
Ronan O'Caoimh, CEO of Trinity said "From an operations point of view we had a very solid quarter. We shipped a record number of Premier instruments (120) this quarter and remain on course to meet our target of 460 units for 2014. We closed our two blood bank screening facilities in the UK and transferred production to our other facilities in Jamestown, New York and Bray, Ireland on schedule. We will benefit from the resultant operating efficiencies from next quarter onwards. Meanwhile, in conjunction with our partner, Nicox, we have now rolled-out our test for Sjogren's syndrome nationwide in the USA. Prior to this, it had only been sold in a limited number of test markets and already revenues are exceeding expectations.
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Die Probleme bei der klinischen Studien für Troponin lesen sich nicht gut. Das Problem lag bei einem Zulieferer. Das Problem wurde erkannt und abgestellt. Der Lieferant für den chemischen Grundstoff liefert jetzt wieder die ursprüngliche Zusammensetzung.
Data compilation and cardiologist adjudication will be completed during the months of June and July and it is anticipated that a final submission will be made in August, 2015. We confidently believe that the product will receive FDA clearance thereafter.
Bereits im Q3 kamen dann auch Umsätze aus der Übernahme der Imco (Augentest):
Following a successful partial launch in a number of test markets earlier this year, it received its nationwide launch late in June, 2014 thus making Q3, 2014 effectively the first full quarter of sales. To date the test has been well received in the market and revenues to date have been exceeding expectations.
Durch Schließeung der Blutbank in England und den Marketingkosten für den Augentest fielen höhere Kosten an:
The impact of these factors during the quarter was approximately $0.4m, which if they were to be excluded would have resulted in profits of $4.8m and EPS of 21 cents".
Das Q4 sollte deutlich höhere Gewinne ausweisen:
Ronan O'Caoimh, CEO of Trinity said "From an operations point of view we had a very solid quarter. We shipped a record number of Premier instruments (120) this quarter and remain on course to meet our target of 460 units for 2014. We closed our two blood bank screening facilities in the UK and transferred production to our other facilities in Jamestown, New York and Bray, Ireland on schedule. We will benefit from the resultant operating efficiencies from next quarter onwards. Meanwhile, in conjunction with our partner, Nicox, we have now rolled-out our test for Sjogren's syndrome nationwide in the USA. Prior to this, it had only been sold in a limited number of test markets and already revenues are exceeding expectations.
aktuell kratzen wir an der Marke bei 18,20 US-D. Sollte wir den Widerstand knacken, dann wäre die Konsolidierung abgeschlossen. Es könnte dann perspektivisch bis zum Frühjahr auf 22 US-D hoch gehen. We will see
Leider sind wir wieder unter 18 Dollar zurückgefallen. Charttechnisch kein gutes Zeichen. Hoffentlich nehmen wir den Widerstand bald wieder. In der ersten März Woche folgen die Quartalszahlen. Da Trinity zum Teil in Irland unter Euro-Bedingungen produziert und in US-Dollar verkauft, sollten Sie im letzten Quartal vom gestiegenen US-Dollar produzieren. Lassen wir uns überraschen, wie die Quartalszahlen ausfallen. Durch die Schließung der Labore in UK sollte auch dei Kostenseite entlastet werden.
nächste Woche sollten dann die Quartalszahlen folgen. Hier eine positive Nachricht zum Troponin Test:
http://finance.yahoo.com/news/trinity-biotech-announces-reco…
http://finance.yahoo.com/news/trinity-biotech-announces-reco…
am 03.03. werden die Q-Zahlen veröffentlicht. Hoffentlich nehmen wir den Widerstand bei 18 US-D.
das Wachstum hat sich im Q4 deutlich abgeschwächt. So richtig Fantasie kann man aus den Zahlen nicht ziehen. Erst wenn der Troponin Test von der FDA zugelassen wird, dann würde hier wieder neues Potential entstehen:
http://finance.yahoo.com/news/trinity-biotech-announces-resu…
http://finance.yahoo.com/news/trinity-biotech-announces-resu…
aus meiner Sicht liegt das maximale Kursziel bei 22 UD-D. Basis KGV Gewinn 2014. Wie 2015 verlaufen wird? Leider gibt es keinen Ausblick.
die Begebung einer Anleihe von 100 Mio US-D sorgt für einen Kursturz von 9 %. Was wollen Sie mir soviel Liquidität? Steht eine größere Übernahme bevor? Wenn dies zu günstigen Konditionen erfolgt, dann wäre doch eigentlich ein Kurssprung angesagt. Oder verstehe ich die Meldung falsch?
http://finance.yahoo.com/news/trinity-biotech-plc-announces-…
Ende April kommen die Quartalszahlen. Mal sehen, ob es dann Infos zur Verwendung gibt.
http://finance.yahoo.com/news/trinity-biotech-plc-announces-…
Ende April kommen die Quartalszahlen. Mal sehen, ob es dann Infos zur Verwendung gibt.
am 30.04. folgen die Quartalszahlen. Hoffentlich gib es dann auc Infos, was mit der Liquidität aus der Anleihe geplant ist
Umsatz ist leicht gestiegen. Der steigende US-D hat sich hier negativ ausgewirkt. Der Gewinn ist auf 0,17 US-D zurückgegangen. Es wird eine Dividende von 0,22 US-D ausgeschüttet. Die Liquidität aus der Anleihe soll für Übernahmen verwendet werden:
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Das aktuelle KGV würde somit bei ca. 24 liegen, wenn die Quartalsergebnisse weiterhin bei 0,17 US-D stagnieren. Damit sollte der Kurs bei 16 US-D abgesichert sein. Dann hoffe wir auf baldige positive Nachrichten zu Firmenübernahmen, damit wieder zusätzliche Fantasie aufkommt.
http://finance.yahoo.com/news/trinity-biotech-announces-quar…
Das aktuelle KGV würde somit bei ca. 24 liegen, wenn die Quartalsergebnisse weiterhin bei 0,17 US-D stagnieren. Damit sollte der Kurs bei 16 US-D abgesichert sein. Dann hoffe wir auf baldige positive Nachrichten zu Firmenübernahmen, damit wieder zusätzliche Fantasie aufkommt.
der Kurs ist wieder über 16 US-D gesprungen. Es wäre wichtig, dass das Niveau hält, da es sich um den Tiefstkurs der letzten 12 Monate handelt. Da der US-D aktuell etwas schwächelt, sollte das Ergebnis im Q2 etwas besser ausfallen. Seit April ist der Euro-Kurs von 1,07 auf aktuell 1,12 gestiegen.
Der Widerstand bei 18 US-D ist umkämpft. Hoffentlich wird dieser bald genommen.
Earnings Date: Jul 27 - Jul 31
Earnings Date: Jul 27 - Jul 31
die 200 Tage Linie haben wir am Dienstag von unten nach oben durchbrochen. Ein weiteres charttechnisches Kaufsignal! Der RSI, die Stochastik und das Momentum sind aber leicht überkauft. Der MACD ist auch in die Waagerechte übergegangen. Wenn der Kurs nicht schnell Richtung 19 US-D steigt, dann ist wohl mit einer Konsolidierung Richtung 17 US-D zu rechnen.
die 50 Tage Linie schwenkt jetzt nach oben. Auch ein gutes Zeichen. Mal abwarten, wie es weiter geht.
So nach über 10 Jahren habe ich jetzt verkauft und verabschiede mich. Leider etwas zu früh verkauft. Allen Investierten weiterhin viel Erfolg.
uh die Quartalsergebnisse sind aber sehr schwach ausgefallen. Gut, dass ich verkauft habe. Die Zwischenkonsolidierung bei 16 US-D wird wohl nicht halten. Das Kursziel bei fallenden Gewinne sehe ich jetzt eher bei 10 US-D.
Beitrag zu dieser Diskussion schreiben
Zu dieser Diskussion können keine Beiträge mehr verfasst werden, da der letzte Beitrag vor mehr als zwei Jahren verfasst wurde und die Diskussion daraufhin archiviert wurde.
Bitte wenden Sie sich an feedback@wallstreet-online.de und erfragen Sie die Reaktivierung der Diskussion oder starten Sie eine neue Diskussion.
Investoren beobachten auch:
| Wertpapier | Perf. % |
|---|---|
| -0,34 | |
| +0,56 | |
| +0,61 | |
| +1,19 | |
| -2,63 | |
| +2,12 | |
| +2,01 | |
| +0,82 | |
| -0,27 | |
| +1,18 |
Meistdiskutiert
| Wertpapier | Beiträge | |
|---|---|---|
| 207 | ||
| 107 | ||
| 83 | ||
| 73 | ||
| 49 | ||
| 43 | ||
| 43 | ||
| 39 | ||
| 33 | ||
| 31 |
| Zeit | Titel |
|---|---|
| 06.07.23 |



















