Sellas Life Sciences Group (ehemals Galena Biopharma) (Seite 146)
eröffnet am 30.10.12 22:43:19 von
neuester Beitrag 05.05.24 11:51:48 von
neuester Beitrag 05.05.24 11:51:48 von
Beiträge: 2.940
ID: 1.177.530
ID: 1.177.530
Aufrufe heute: 51
Gesamt: 345.795
Gesamt: 345.795
Aktive User: 0
ISIN: US81642T2096 · WKN: A2PU3T · Symbol: RXK3
1,2220
EUR
-2,55 %
-0,0320 EUR
Letzter Kurs 03.05.24 Tradegate
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,2000 | +471,16 | |
| 13,110 | +38,44 | |
| 6,5000 | +27,45 | |
| 1,2100 | +21,00 | |
| 48,25 | +19,94 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 6,2600 | -14,25 | |
| 3,8500 | -14,45 | |
| 1,0011 | -15,88 | |
| 36,70 | -22,87 | |
| 2,5600 | -70,32 |
Beitrag zu dieser Diskussion schreiben
abwärts ist relaiv!

Antwort auf Beitrag Nr.: 50.635.731 von tancho am 16.09.15 12:33:28jetzt wo Du das sagst gehts abwärts 

ist meistens so wenn ich dabei bin

If the trend is really your friend ...
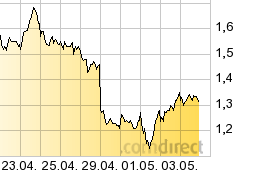
...whats this?

...whats this?


Antwort auf Beitrag Nr.: 50.629.600 von hardliner1A am 15.09.15 15:31:07Hi Hardliner, Augenmerk auf Gale-301 und die ESMO werfen
da bin ich voll & ganz bei dir ...
Vorallem bzgl. ESMO (european footprint & wieder ist man Roche in der Schweiz ganz nah bei , ich frag mich nur ob Schwartz gedenkt (Höchstpersönlich) hier in Erscheinung zu treten
, ich frag mich nur ob Schwartz gedenkt (Höchstpersönlich) hier in Erscheinung zu treten ...wäre -meines Erachtens -ein ganz starken Signalwirkung das positive Zwischendata demnächst im Anmarsch sei
...wäre -meines Erachtens -ein ganz starken Signalwirkung das positive Zwischendata demnächst im Anmarsch sei
da bin ich voll & ganz bei dir ...

Vorallem bzgl. ESMO (european footprint & wieder ist man Roche in der Schweiz ganz nah bei
 , ich frag mich nur ob Schwartz gedenkt (Höchstpersönlich) hier in Erscheinung zu treten
, ich frag mich nur ob Schwartz gedenkt (Höchstpersönlich) hier in Erscheinung zu treten ...wäre -meines Erachtens -ein ganz starken Signalwirkung das positive Zwischendata demnächst im Anmarsch sei
...wäre -meines Erachtens -ein ganz starken Signalwirkung das positive Zwischendata demnächst im Anmarsch sei
Antwort auf Beitrag Nr.: 50.629.247 von Growth2012 am 15.09.15 14:49:12Wenn die Insider jetzt auch noch kaufen würden, dann wäre uns allen gewiss, dass hier was großes im Busch ist 
Ich bin letzte Woche schon mal vorsorglich eingestiegen
Das FDA Meeting würde ich aber nicht gerade als Kurstreiber bezeichnen. Das findet ja erst zum Ende des Jahres statt.
Ich denke, wir sollten eher unser Augenmerk auf Gale-301 und die ESMO werfen.

Ich bin letzte Woche schon mal vorsorglich eingestiegen

Das FDA Meeting würde ich aber nicht gerade als Kurstreiber bezeichnen. Das findet ja erst zum Ende des Jahres statt.
Ich denke, wir sollten eher unser Augenmerk auf Gale-301 und die ESMO werfen.

Bin zurzeit sehr überzeugt vom Gale's Behmühungen Richtung Zulassung mit NeuVax, hier zwei postings stateside die diese Überzeügung "leicht unterstützten"...
Dr. Schwartz Dropped a BOMB on the Shorts - FDA Meeting for FBP
He also stated that 50% of Ovo/Endo patients suffer a recurrence in the FIRST 12 MONTHS -- This is very short time line - This Trial data is NOT BLIND - He sees the results and says he is "excited, very excited", add that up along with a significant unmet need + a Meeting with the FDA -- Do the math. GALE PPS Is on the way UP. Less
LOL James. The response of the share price to your post reminds me of the saying you can lead a cow to the water but you cannot make her drink. I have been jumping in and out of GALE for a few times in the past but I always keep 1 share in my portfolio to help me monitor it. When the G 301 result came out I could not believe what I saw. Even more amazing was the share price at that time. I jumped back in with both feet and hands. I have been buying non stop, even today. You cannot ask for more of an opportunity. A 1/15 vg vs 11/22 cg with a p=.035 in 9.8 months and a share price of 1.5. Are you kidding ? You don't need to be a medical scientist to understand it.
The only thing even more amazing is some people go short on GALE. Must be the lead in their drinking water. Less
http://finance.yahoo.com/mbview/threadview/?&bn=12a6b315-348…
The question for me is Partnership, or go it alone
Schwartz wäre nie so euphorisch wenn er keine vertiefte kenntnisse hätte (The Dream Team "Übertreibung days "lassen wieder Grüßen
This Trial data is NOT BLIND - He sees the results and says he is "excited, very excited", add that up along with a significant unmet need + a Meeting with the FDA


Dr. Schwartz Dropped a BOMB on the Shorts - FDA Meeting for FBP
He also stated that 50% of Ovo/Endo patients suffer a recurrence in the FIRST 12 MONTHS -- This is very short time line - This Trial data is NOT BLIND - He sees the results and says he is "excited, very excited", add that up along with a significant unmet need + a Meeting with the FDA -- Do the math. GALE PPS Is on the way UP. Less
LOL James. The response of the share price to your post reminds me of the saying you can lead a cow to the water but you cannot make her drink. I have been jumping in and out of GALE for a few times in the past but I always keep 1 share in my portfolio to help me monitor it. When the G 301 result came out I could not believe what I saw. Even more amazing was the share price at that time. I jumped back in with both feet and hands. I have been buying non stop, even today. You cannot ask for more of an opportunity. A 1/15 vg vs 11/22 cg with a p=.035 in 9.8 months and a share price of 1.5. Are you kidding ? You don't need to be a medical scientist to understand it.
The only thing even more amazing is some people go short on GALE. Must be the lead in their drinking water. Less
http://finance.yahoo.com/mbview/threadview/?&bn=12a6b315-348…
The question for me is Partnership, or go it alone

Schwartz wäre nie so euphorisch wenn er keine vertiefte kenntnisse hätte (The Dream Team "Übertreibung days "lassen wieder Grüßen

This Trial data is NOT BLIND - He sees the results and says he is "excited, very excited", add that up along with a significant unmet need + a Meeting with the FDA


The 2 year DFS estimate is 24.8% (CG) v 91.7% (1000 mcg pts) (p=0.02). OUTSTANDING RESULTS!!
Preliminary results of the phase I/IIa dose finding trial of a folate binding protein vaccine (E39+GM-CSF) in ovarian and endometrial cancer patients to prevent recurrence
Session title: Gynaecological Cancer
Session type: Poster Session
Track: Gynaecological Cancer
Abstract number: 2764
Abstract title:
Preliminary results of the phase I/IIa dose finding trial of a folate binding protein vaccine (E39+GM-CSF) in ovarian and endometrial cancer patients to prevent recurrence
J.M. Greene(1), E.J. Schneble(1), J.S. Berry(1), A.F. Trappey(1), G.T. Clifton(2), K. Darcy(3), J.C. Elkas(4), C.A. Hamilton(5), G.L. Maxwell(6), G.E. Peoples(7)
(1)San Antonio Military Medical Center, General Surgery, San Antonio, TX, USA
(2)MD Anderson Cancer Center, Surgical Oncology, Houston, TX, USA
(3)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Annandale, VA, USA
(4)Mid-Atlantic Gynecologic Oncology and Pelvic Surgical Associates, Gynecologic Oncology, Annandale, VA, USA
(5)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Bethesda, MD, USA
(6)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Falls Church, VA, USA
(7)Cancer Vaccine Development Program/Cancer Insight/Galena Consultant, Surgical Oncology, Bethesda, MD/San Antonio, TX, USA
Background: Folate Binding Protein (FBP) (aka Folate Receptor-a) is an immunogenic protein over-expressed in endometrial (EC) and ovarian cancer (OC). We are conducting a phase I/IIa trial with E39, an HLA-A2-restricted, FBP-derived peptide + GM-CSF vaccine. E39 is given adjuvantly to prevent clinical recurrences (CR) in disease-free high-risk, EC and OC patients (pts) after standard-of-care therapy (SOC). Here, we summarize toxicity, in vivo immunologic responses, and disease-free survival (DFS) in this dose-finding trial.
Materials and Methods: This trial began as a 3×3, dose-escalation (100, 500, 1000 mcg of E39), safety phase I trial and transitioned to a phase IIa comparing expanded dose cohorts. Disease-free EC and OC pts were enrolled after SOC. HLA-A2+ pts were vaccinated (VG), and HLA-A2- pts followed prospectively as a control group (CG). 6 monthly intradermal inoculations of E39 + 250 mcg GM-CSF are administered followed by 2 boosters every 6 months. Demographic, safety, immunologic, and CR data are being collected. In vivo immunologic response is measured by the orthogonal mean of a delayed type hypersensitivity reaction (DTH) to the E39 peptide without GM-CSF placed remote from the primary vaccination site. DFS is compared by Kaplan-Meir and logrank tests. Continuous variables are compared with analysis of variance and proportions with Fisher's exact test.
Results: 51 pts were enrolled; 29 in the VG (15 pts 1000mcg v 14 pts <1000mcg) and 22 in the CG. There are no differences in age, grade, stage, or histology between groups (all p=0.1). E39 is well-tolerated and safe with no >grade 3 toxicities and no differences in toxicities based on dose. DTH increased pre- to post-vaccination (5.9mm+1.5 v 11.7mm+3.2, p=0.08). DTH increase was larger in 1000 mcg pts (3.8mm v 17.8mm, p=0.06) v <1000 mcg pts (8mm v 11mm, p=0.56). With 9.3 months median follow-up, CR rate is 38% (VG) v 50% (CG), p=0.41. Among 1000 mcg pts, there is only one CR (6.7% v 50% CG, p=0.01). The 2 year DFS estimate is 24.8% (CG) v 91.7% (1000 mcg pts) (p=0.02).
Conclusions: This phase I/IIa trial reveals that E39 + GM-CSF is well-tolerated and elicits a strong and dose-dependent in vivo immune response. Early efficacy results are promising in the 1000 mcg dose cohort. This dose-finding study proves the safety and establishes the dose of E39 for a prospective, randomized, GM-CSF controlled, late stage trial in HLA-A2+ EC and OC patients to prevent recurrence.
Trial Number: NCT01580696
Conflict of interest: Other Substantive Relationships: George Peoples is a consultant to and stockholder of Galena. He holds partial patent rights and royalties to E39.
http://investorshub.advfn.com/boards/read_msg.aspx?message_i…
Preliminary results of the phase I/IIa dose finding trial of a folate binding protein vaccine (E39+GM-CSF) in ovarian and endometrial cancer patients to prevent recurrence
Session title: Gynaecological Cancer
Session type: Poster Session
Track: Gynaecological Cancer
Abstract number: 2764
Abstract title:
Preliminary results of the phase I/IIa dose finding trial of a folate binding protein vaccine (E39+GM-CSF) in ovarian and endometrial cancer patients to prevent recurrence
J.M. Greene(1), E.J. Schneble(1), J.S. Berry(1), A.F. Trappey(1), G.T. Clifton(2), K. Darcy(3), J.C. Elkas(4), C.A. Hamilton(5), G.L. Maxwell(6), G.E. Peoples(7)
(1)San Antonio Military Medical Center, General Surgery, San Antonio, TX, USA
(2)MD Anderson Cancer Center, Surgical Oncology, Houston, TX, USA
(3)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Annandale, VA, USA
(4)Mid-Atlantic Gynecologic Oncology and Pelvic Surgical Associates, Gynecologic Oncology, Annandale, VA, USA
(5)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Bethesda, MD, USA
(6)Department of Defense Gynecologic Cancer Center of Excellence Women's Health Integrated Research Center at Inova Health System/Inova Fairfax Hospital, Gynecologic Oncology, Falls Church, VA, USA
(7)Cancer Vaccine Development Program/Cancer Insight/Galena Consultant, Surgical Oncology, Bethesda, MD/San Antonio, TX, USA
Background: Folate Binding Protein (FBP) (aka Folate Receptor-a) is an immunogenic protein over-expressed in endometrial (EC) and ovarian cancer (OC). We are conducting a phase I/IIa trial with E39, an HLA-A2-restricted, FBP-derived peptide + GM-CSF vaccine. E39 is given adjuvantly to prevent clinical recurrences (CR) in disease-free high-risk, EC and OC patients (pts) after standard-of-care therapy (SOC). Here, we summarize toxicity, in vivo immunologic responses, and disease-free survival (DFS) in this dose-finding trial.
Materials and Methods: This trial began as a 3×3, dose-escalation (100, 500, 1000 mcg of E39), safety phase I trial and transitioned to a phase IIa comparing expanded dose cohorts. Disease-free EC and OC pts were enrolled after SOC. HLA-A2+ pts were vaccinated (VG), and HLA-A2- pts followed prospectively as a control group (CG). 6 monthly intradermal inoculations of E39 + 250 mcg GM-CSF are administered followed by 2 boosters every 6 months. Demographic, safety, immunologic, and CR data are being collected. In vivo immunologic response is measured by the orthogonal mean of a delayed type hypersensitivity reaction (DTH) to the E39 peptide without GM-CSF placed remote from the primary vaccination site. DFS is compared by Kaplan-Meir and logrank tests. Continuous variables are compared with analysis of variance and proportions with Fisher's exact test.
Results: 51 pts were enrolled; 29 in the VG (15 pts 1000mcg v 14 pts <1000mcg) and 22 in the CG. There are no differences in age, grade, stage, or histology between groups (all p=0.1). E39 is well-tolerated and safe with no >grade 3 toxicities and no differences in toxicities based on dose. DTH increased pre- to post-vaccination (5.9mm+1.5 v 11.7mm+3.2, p=0.08). DTH increase was larger in 1000 mcg pts (3.8mm v 17.8mm, p=0.06) v <1000 mcg pts (8mm v 11mm, p=0.56). With 9.3 months median follow-up, CR rate is 38% (VG) v 50% (CG), p=0.41. Among 1000 mcg pts, there is only one CR (6.7% v 50% CG, p=0.01). The 2 year DFS estimate is 24.8% (CG) v 91.7% (1000 mcg pts) (p=0.02).
Conclusions: This phase I/IIa trial reveals that E39 + GM-CSF is well-tolerated and elicits a strong and dose-dependent in vivo immune response. Early efficacy results are promising in the 1000 mcg dose cohort. This dose-finding study proves the safety and establishes the dose of E39 for a prospective, randomized, GM-CSF controlled, late stage trial in HLA-A2+ EC and OC patients to prevent recurrence.
Trial Number: NCT01580696
Conflict of interest: Other Substantive Relationships: George Peoples is a consultant to and stockholder of Galena. He holds partial patent rights and royalties to E39.
http://investorshub.advfn.com/boards/read_msg.aspx?message_i…
Hoffe sehr das GALE ein wesentliche Beitrag hiebei leisten wird....
http://www.breastcancerdeadline2020.org/about-the-deadline/

http://www.breastcancerdeadline2020.org/about-the-deadline/















