Kezar Life Sciences - KZR 616
eröffnet am 10.02.20 22:36:11 von
neuester Beitrag 04.03.24 23:32:49 von
neuester Beitrag 04.03.24 23:32:49 von
Beiträge: 23
ID: 1.320.109
ID: 1.320.109
Aufrufe heute: 0
Gesamt: 2.661
Gesamt: 2.661
Aktive User: 0
ISIN: US49372L1008 · WKN: A2JPE7 · Symbol: 2KZ
0,7600
EUR
-3,80 %
-0,0300 EUR
Letzter Kurs 06.05.24 Tradegate
Neuigkeiten
06.05.24 · Business Wire (engl.) |
08.04.24 · Business Wire (engl.) |
14.03.24 · Business Wire (engl.) |
08.03.24 · Business Wire (engl.) |
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 6.300,00 | +4.940,00 | |
| 1,4600 | +34,94 | |
| 1,2300 | +24,24 | |
| 2,7600 | +20,00 | |
| 0,8000 | +19,40 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 21,600 | -20,00 | |
| 1,3800 | -20,80 | |
| 0,6700 | -26,58 | |
| 2,1300 | -34,41 | |
| 3,1600 | -38,64 |
Beitrag zu dieser Diskussion schreiben
Kezar Life Sciences Inc. : Mehr Chance als Risiko?
Bin hier seit einigen Tagen recht optimistisch investiert.3 Top Penny Stocks for 5,000% Upside in 2024 and Beyond
Josh Enomoto
February 25, 2024
https://www.nasdaq.com/market-activity/stocks/kzr/institutio…
3 Penny Stocks Poised for an Unbelievable 1,000% Jump
Yiannis Zourmpanos
February 2, 2024
https://finance.yahoo.com/news/3-top-penny-stocks-5-19121226…
https://finance.yahoo.com/news/3-penny-stocks-poised-unbelie…
https://finance.yahoo.com/news/bull-run-bonanza-top-3-135102…
Institutional Holdings 68,80 %
https://www.nasdaq.com/market-activity/stocks/kzr/institutio…
Kursziel 13,00 USD
https://www.nasdaq.com/market-activity/stocks/kzr
Novo Holdings beteiligt sich an Kezar:
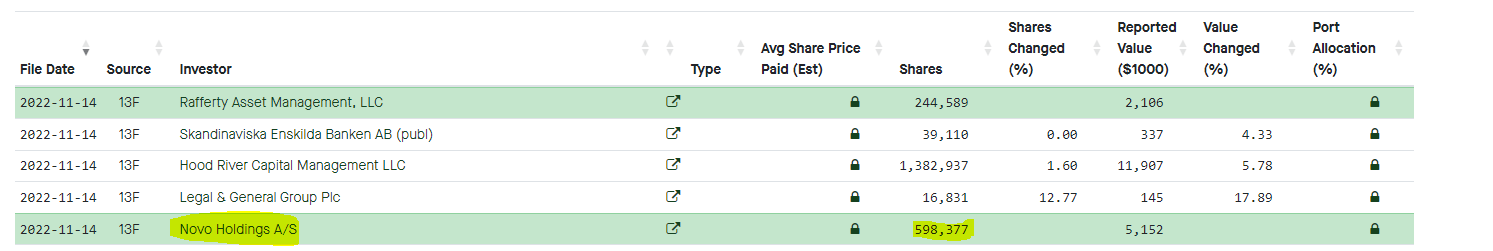
Novo Holdings A/S ist ein dänischer Pharmahersteller; zu der Novo-Gruppe gehören die eigenständigen agierenden Tochterunternehmen Novo Nordisk, Novozymes und NNIT. Die Novo Holdings gehört der Novo Nordisk Foundation und ist Hauptanteilseigner der drei Pharmaunternehmen.
https://fintel.io/so/us/kzr
https://de.wikipedia.org/wiki/Novo_Holdings

Novo Holdings A/S ist ein dänischer Pharmahersteller; zu der Novo-Gruppe gehören die eigenständigen agierenden Tochterunternehmen Novo Nordisk, Novozymes und NNIT. Die Novo Holdings gehört der Novo Nordisk Foundation und ist Hauptanteilseigner der drei Pharmaunternehmen.
https://fintel.io/so/us/kzr
https://de.wikipedia.org/wiki/Novo_Holdings
Kezar Life Sciences Presents Positive Complete Results from the MISSION Phase 2 Trial Evaluating Zetomipzomib in Lupus Nephritis
Summary of Results from the Completed MISSION Trial
In the MISSION Phase 2 clinical trial, 17 of 21 enrolled patients reached end-of-treatment at Week 25 and end-of-study at Week 37. Zetomipzomib treatment demonstrated steady and clinically meaningful renal responses with additional ORRs and CRRs observed during the safety follow-up period.
Overall Renal Responses:
10 out 17 patients (58.8%) achieved an ORR as early as Week 13.
At EOT, 11 of 17 patients (64.7%) achieved an ORR, measured as a 50% or greater reduction in UPCR compared to baseline, the primary endpoint of the clinical trial.
During the safety follow-up period, clinical responses deepened, and ORRs increased to 16 of 17 patients (94.1%) at Week 29 and 15 of 17 patients (88.2%) at EOS. In addition, UPCR was reduced to clinically meaningful levels:
11 of 17 patients (64.7%) achieved a UPCR of 0.5 or less at EOS.
15 of 17 patients (88.2%) achieved a UPCR of 0.7 or less at EOS.
Median UPCR was 0.32 at EOS.
Complete Renal Responses:
5 out 17 patients (29.4%) achieved a CRR as early as Week 13.
At EOT, 6 of 17 patients (35.3%) achieved a CRR, a key secondary efficacy endpoint, measured as a UPCR of 0.5 or less, stable eGFR, daily prednisone/prednisone equivalent dose of 10 mg or less, and no use of prohibited medication.
During the safety follow-up period, an additional patient achieved a CRR, with the total CRRs increasing to 7 of 17 patients (41.2%) at Week 29 and EOS, demonstrating a deepening renal response throughout the 37-week trial.
https://finance.yahoo.com/news/kezar-life-sciences-presents-…
Summary of Results from the Completed MISSION Trial
In the MISSION Phase 2 clinical trial, 17 of 21 enrolled patients reached end-of-treatment at Week 25 and end-of-study at Week 37. Zetomipzomib treatment demonstrated steady and clinically meaningful renal responses with additional ORRs and CRRs observed during the safety follow-up period.
Overall Renal Responses:
10 out 17 patients (58.8%) achieved an ORR as early as Week 13.
At EOT, 11 of 17 patients (64.7%) achieved an ORR, measured as a 50% or greater reduction in UPCR compared to baseline, the primary endpoint of the clinical trial.
During the safety follow-up period, clinical responses deepened, and ORRs increased to 16 of 17 patients (94.1%) at Week 29 and 15 of 17 patients (88.2%) at EOS. In addition, UPCR was reduced to clinically meaningful levels:
11 of 17 patients (64.7%) achieved a UPCR of 0.5 or less at EOS.
15 of 17 patients (88.2%) achieved a UPCR of 0.7 or less at EOS.
Median UPCR was 0.32 at EOS.
Complete Renal Responses:
5 out 17 patients (29.4%) achieved a CRR as early as Week 13.
At EOT, 6 of 17 patients (35.3%) achieved a CRR, a key secondary efficacy endpoint, measured as a UPCR of 0.5 or less, stable eGFR, daily prednisone/prednisone equivalent dose of 10 mg or less, and no use of prohibited medication.
During the safety follow-up period, an additional patient achieved a CRR, with the total CRRs increasing to 7 of 17 patients (41.2%) at Week 29 and EOS, demonstrating a deepening renal response throughout the 37-week trial.
https://finance.yahoo.com/news/kezar-life-sciences-presents-…
Kezar Life Sciences Receives FDA Clearance of IND for Zetomipzomib for the Treatment of Autoimmune Hepatitis
The PORTOLA trial (KZR-616-208) is a randomized, double-blind, placebo-controlled Phase 2a clinical trial evaluating the safety and efficacy of zetomipzomib in patients with AIH that are insufficiently responding to standard of care or have relapsed.
We plan to run PORTOLA, an early proof-of-concept study in parallel to our other development efforts, including a late-phase efficacy trial for patients with lupus nephritis and a potential program in patients with systemic lupus erythematosus," said Noreen R. Henig, M.D., Kezar’s Chief Medical Officer. "Autoimmune hepatitis is a disease where there is a good fit between the pathophysiology of the disease and mechanism of action of zetomipzomib, as well as significant unmet medical need and few alternative therapies.
https://finance.yahoo.com/news/kezar-life-sciences-receives-…
The PORTOLA trial (KZR-616-208) is a randomized, double-blind, placebo-controlled Phase 2a clinical trial evaluating the safety and efficacy of zetomipzomib in patients with AIH that are insufficiently responding to standard of care or have relapsed.
We plan to run PORTOLA, an early proof-of-concept study in parallel to our other development efforts, including a late-phase efficacy trial for patients with lupus nephritis and a potential program in patients with systemic lupus erythematosus," said Noreen R. Henig, M.D., Kezar’s Chief Medical Officer. "Autoimmune hepatitis is a disease where there is a good fit between the pathophysiology of the disease and mechanism of action of zetomipzomib, as well as significant unmet medical need and few alternative therapies.
https://finance.yahoo.com/news/kezar-life-sciences-receives-…
Wells Fargo reiterated its Overweight rating and raised its price target to $17 from $13 for Kezar Life Sciences Inc (NASDAQ: KZR) on zetomipzomib's (zeto) positive Ph2 MISSION data in lupus nephritis (LN).
The analysts say that the data is in-line with our bull case and well ahead of Street expectations since PRESIDIO's failure.
Wells Fargo says the high complete renal response/overall renal response, the durability of effect, and meaningful reductions in steroids in the study validate zeto as an active agent in LN.
They see a peak sales opportunity of approximately $1 billion. KZR plans to discuss zeto's next steps in LN with the FDA later this year.
"We think the "dream the dream" scenario is still alive for zeto as the co plans to explore additional indications, which may include systemic lupus erythematosus (SLE)." Wells Fargo notes.
The analysts think zeto's data is even more impressive given its ~35% CRR is with lower starting doses of MMF and lower/declining doses of steroids.
11 of 17 patients (64.7%) achieved an ORR measured, the primary efficacy endpoint of the clinical trial.
https://finance.yahoo.com/news/wells-fargo-beefs-kezar-life-…
The analysts say that the data is in-line with our bull case and well ahead of Street expectations since PRESIDIO's failure.
Wells Fargo says the high complete renal response/overall renal response, the durability of effect, and meaningful reductions in steroids in the study validate zeto as an active agent in LN.
They see a peak sales opportunity of approximately $1 billion. KZR plans to discuss zeto's next steps in LN with the FDA later this year.
"We think the "dream the dream" scenario is still alive for zeto as the co plans to explore additional indications, which may include systemic lupus erythematosus (SLE)." Wells Fargo notes.
The analysts think zeto's data is even more impressive given its ~35% CRR is with lower starting doses of MMF and lower/declining doses of steroids.
11 of 17 patients (64.7%) achieved an ORR measured, the primary efficacy endpoint of the clinical trial.
https://finance.yahoo.com/news/wells-fargo-beefs-kezar-life-…
KEZAR LIFE SCIENCES ANNOUNCES POSITIVE TOPLINE RESULTS FROM THE MISSION PHASE 2 TRIAL EVALUATING ZETOMIPZOMIB FOR THE TREATMENT OF PATIENTS WITH LUPUS NEPHRITIS
- 11 of 17 patients (64.7%) achieved an overall renal response of 50% or greater reduction in
urine protein to creatinine ratio (UPCR) at 6 months
- 6 of 17 patients (35.2%) achieved a complete renal response, including a UPCR of 0.5 or less at 6
months
- Zetomipzomib continues to demonstrate a favorable safety and tolerability profile for
administration over the 6-month treatment period
- Improvement seen in exploratory measures of extra-renal disease activity associated with systemic lupus erythematosus (SLE) in patients who completed treatment
https://ir.kezarlifesciences.com/news-releases/news-release-…
Morgen könnte hier eine große Party gefeiert werden, vielleicht kommt es sogar zu einer Schließung der großen Lücke nach oben:

💡





















