Imagin Medical -- wie sieht die Mehrheit die Entwicklung ? - 500 Beiträge pro Seite
eröffnet am 26.05.16 16:32:46 von
neuester Beitrag 14.11.20 09:53:23 von
neuester Beitrag 14.11.20 09:53:23 von
Beiträge: 186
ID: 1.232.392
ID: 1.232.392
Aufrufe heute: 0
Gesamt: 17.106
Gesamt: 17.106
Aktive User: 0
ISIN: CA45250L1058 · WKN: A2AE35
0,0145
EUR
0,00 %
0,0000 EUR
Letzter Kurs 26.10.20 Tradegate
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 4,0000 | +25,00 | |
| 0,6750 | +20,54 | |
| 2,3800 | +20,51 | |
| 0,8200 | +18,84 | |
| 5,7700 | +16,80 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,4300 | -14,37 | |
| 5,0639 | -16,13 | |
| 0,5350 | -20,15 | |
| 3,0800 | -23,76 | |
| 4,2000 | -26,96 |
gibt es hier jemanden mit Hintergrund Infos ?
Antwort auf Beitrag Nr.: 52.483.105 von OliKo am 26.05.16 16:32:46
Aktiencheck erscheint mir als bezahlter Pusher zu funktionieren... ständig sehe ich bei Facebook diese Nachrichten zu den nächsten Millionenaktien !
hat dort jemand Erfahrung ?
News
http://www.aktiencheck.de/exklusiv/Artikel-Dramatische_Forts…Aktiencheck erscheint mir als bezahlter Pusher zu funktionieren... ständig sehe ich bei Facebook diese Nachrichten zu den nächsten Millionenaktien !
hat dort jemand Erfahrung ?
Also ich denke diese Aktie ist vorerst stark News abhängig.
Aber ich denke sobald der erste kommerzielle Prototyp fertiggestellt ist, wird sich hier bestimmt was tun.
Vor allem wenn dieses neue Verfahren zur Krebserkennung so wirksam ist wie beschrieben.
Aber ich denke sobald der erste kommerzielle Prototyp fertiggestellt ist, wird sich hier bestimmt was tun.
Vor allem wenn dieses neue Verfahren zur Krebserkennung so wirksam ist wie beschrieben.
Aktuell ein pp am Laufen:
http://imaginmedical.com/wp-content/uploads/2016/09/Imagin-N…
"CSE Symbol: IME
OTC PINK Symbol: IMEXF
Frankfurt/Stuttgart Stock Exchanges: DPD2
NR-2016-IME-16
Imagin Medical
Announces Private Placement
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish a new standard of care for urologists in the early detection of bladder cancer through endoscopes.
Vancouver, B.C. and Boston, MA, September 13, 2016 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) announced today its intention to undertake a non-brokered private placement to raise $800,000 through the distribution of units (“Units”) at $0.08 per Unit.
Each Unit will consist of one common share and one common share purchase warrant; each warrant entitling the holder to acquire one additional common share at $0.12 in the first 12 months, and at $0.16 in the 12 months thereafter.
Finder’s fees may be paid, in accordance with Canadian Securities Exchange policies. Closing of the placement will be subject to making applicable filings with the CSE.
Proceeds from the placement will be used for general working capital purposes, including advancing the development of imaging solutions for the early detection of cancer through the use of endoscopes.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer through the use of endoscopes. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
..."
http://imaginmedical.com/wp-content/uploads/2016/09/Imagin-N…
"CSE Symbol: IME
OTC PINK Symbol: IMEXF
Frankfurt/Stuttgart Stock Exchanges: DPD2
NR-2016-IME-16
Imagin Medical
Announces Private Placement
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish a new standard of care for urologists in the early detection of bladder cancer through endoscopes.
Vancouver, B.C. and Boston, MA, September 13, 2016 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) announced today its intention to undertake a non-brokered private placement to raise $800,000 through the distribution of units (“Units”) at $0.08 per Unit.
Each Unit will consist of one common share and one common share purchase warrant; each warrant entitling the holder to acquire one additional common share at $0.12 in the first 12 months, and at $0.16 in the 12 months thereafter.
Finder’s fees may be paid, in accordance with Canadian Securities Exchange policies. Closing of the placement will be subject to making applicable filings with the CSE.
Proceeds from the placement will be used for general working capital purposes, including advancing the development of imaging solutions for the early detection of cancer through the use of endoscopes.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer through the use of endoscopes. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
..."
http://imaginmedical.com/wp-content/uploads/2016/09/News-Rel…
"890 West Pender Street, Suite 600
Vancouver, British Columbia
Canada V6C 1J9
778-998-5000
133 Hampshire Road
Wellesley, MA, 02481, USA
617-571-6006
www.imaginmedical.com
CSE Symbol: IME
OTC PINK Symbol: IMEXF
Frankfurt/Stuttgart Stock Exchanges: DPD2
NR-2016-IME-17
Disruptive Cancer Detection Technology Update
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish a new standard of care for urologists in the early detection of bladder cancer through endoscopes.
Vancouver, B.C. and Boston, MA, September 15, 2016 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) As a follow up to Imagin’s announcement of an $800,000 private placement Tuesday, September 13, 2016, the Company announces it is making significant progress not only in the capital raise, but also in developing its disruptive technology, the i/Blue Imaging System. Imagin’s goal is to establish a new standard of care in detecting cancer and reducing its recurrence through the use of endoscopes.
Imagin’s initial target is bladder cancer which has a greater than 50% recurrence rate and, although the 6th most prevalent cancer in the U.S., is the most expensive cancer to treat.
Imagin’s Winning Technology
Based on combination of improved optical designs, components and advanced light sensors expected to be 100,000 times more sensitive
Employs Simultaneous Acquisition of Differing Images, patented technology that automatically blends white light and fluorescence images into one, putting the cancer into context
Improves visualization to help reduce chances of recurrence by enabling physicians to more accurately visualize, assess and remove cancerous cells
Time savings will increase efficiency in the operating room and potentially enable procedures to be performed in less-expensive physician’s office for follow-up examinations
Dr. Stavros Demos invented the i/Blue Imaging System while at Lawrence Livermore National Lab and recently accepted the position of “Senior Scientist and Group Leader of the Optical Materials Group” at the University of Rochester Laboratory for Laser Energetics (LLE) in New York. Imagin has signed a Collaborative Research Agreement with The University of Rochester
LLE, as well as a loan agreement with the U.S. Department of Energy (D.O.E.), and expects
that both agreements will significantly facilitate the development of the i/Blue Imagining
System.
As Imagin’s Technical Scientific Advisor, Dr. Demos, will continue to provide support to
Imagin’s engineering team as it continues to build on the initial prototype. The steps include
refining the product’s design, requalifying the system’s optical model, components and
advanced light sensors, and helping create the controls needed for optimized operation and
patient safety. “I’ve worked on developing many medical device technologies over the last
twenty years. What makes i/Blue especially exciting is working with Dr. Demos and the rest of
the team on such innovative technology that holds so much potential for the future of
healthcare,” said Mike Vergano, Imagin’s Director of Engineering and Operations.
The Company’s immediate goal is to complete the retrofitting of the Alpha prototype by year
end. Bench testing and clinical studies will begin in the 1st quarter of 2017. These next
generation prototypes will be used as the basis for regulatory submission and eventual
commercial distribution.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer through the
use of endoscopes. The Company believes it will radically improve the way physicians detect
cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth
most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50%
recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced,
ultrasensitive imaging technology is based upon improved optical designs and advanced light
sensors. Learn more at www.imaginmedical.com.
ON BEHALF OF THE BOARD:
Jim Hutchens,
President & CEO
For further information, contact:
Bill Galine, Investor Relations
Telephone: (775) 737-3292
Email: billgaline@gmail.com
..."
"890 West Pender Street, Suite 600
Vancouver, British Columbia
Canada V6C 1J9
778-998-5000
133 Hampshire Road
Wellesley, MA, 02481, USA
617-571-6006
www.imaginmedical.com
CSE Symbol: IME
OTC PINK Symbol: IMEXF
Frankfurt/Stuttgart Stock Exchanges: DPD2
NR-2016-IME-17
Disruptive Cancer Detection Technology Update
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish a new standard of care for urologists in the early detection of bladder cancer through endoscopes.
Vancouver, B.C. and Boston, MA, September 15, 2016 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) As a follow up to Imagin’s announcement of an $800,000 private placement Tuesday, September 13, 2016, the Company announces it is making significant progress not only in the capital raise, but also in developing its disruptive technology, the i/Blue Imaging System. Imagin’s goal is to establish a new standard of care in detecting cancer and reducing its recurrence through the use of endoscopes.
Imagin’s initial target is bladder cancer which has a greater than 50% recurrence rate and, although the 6th most prevalent cancer in the U.S., is the most expensive cancer to treat.
Imagin’s Winning Technology
Based on combination of improved optical designs, components and advanced light sensors expected to be 100,000 times more sensitive
Employs Simultaneous Acquisition of Differing Images, patented technology that automatically blends white light and fluorescence images into one, putting the cancer into context
Improves visualization to help reduce chances of recurrence by enabling physicians to more accurately visualize, assess and remove cancerous cells
Time savings will increase efficiency in the operating room and potentially enable procedures to be performed in less-expensive physician’s office for follow-up examinations
Dr. Stavros Demos invented the i/Blue Imaging System while at Lawrence Livermore National Lab and recently accepted the position of “Senior Scientist and Group Leader of the Optical Materials Group” at the University of Rochester Laboratory for Laser Energetics (LLE) in New York. Imagin has signed a Collaborative Research Agreement with The University of Rochester
LLE, as well as a loan agreement with the U.S. Department of Energy (D.O.E.), and expects
that both agreements will significantly facilitate the development of the i/Blue Imagining
System.
As Imagin’s Technical Scientific Advisor, Dr. Demos, will continue to provide support to
Imagin’s engineering team as it continues to build on the initial prototype. The steps include
refining the product’s design, requalifying the system’s optical model, components and
advanced light sensors, and helping create the controls needed for optimized operation and
patient safety. “I’ve worked on developing many medical device technologies over the last
twenty years. What makes i/Blue especially exciting is working with Dr. Demos and the rest of
the team on such innovative technology that holds so much potential for the future of
healthcare,” said Mike Vergano, Imagin’s Director of Engineering and Operations.
The Company’s immediate goal is to complete the retrofitting of the Alpha prototype by year
end. Bench testing and clinical studies will begin in the 1st quarter of 2017. These next
generation prototypes will be used as the basis for regulatory submission and eventual
commercial distribution.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer through the
use of endoscopes. The Company believes it will radically improve the way physicians detect
cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth
most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50%
recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced,
ultrasensitive imaging technology is based upon improved optical designs and advanced light
sensors. Learn more at www.imaginmedical.com.
ON BEHALF OF THE BOARD:
Jim Hutchens,
President & CEO
For further information, contact:
Bill Galine, Investor Relations
Telephone: (775) 737-3292
Email: billgaline@gmail.com
..."
Antwort auf Beitrag Nr.: 53.299.410 von praesens am 18.09.16 17:41:19Ein Prototyp in 2016 (3 Monate) zu haben und Tests und klinische Studien im ersten Quartal 2017, hört sich so an, das es sich in den nächsten 6 Monaten entscheiden dürfte ...
Eine Geldmaschine die ausspuckt oder auffrisst ?
Eine Geldmaschine die ausspuckt oder auffrisst ?
Der Vollständigkeit halber:
http://imaginmedical.com/wp-content/uploads/2016/09/Imagin-N…
"... Dr. Edward Messing, Chief of Urology, U Rochester Medical Center
Joins Imagin Medical’s Scientific Board of Advisors
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish
a new standard of care for urologists in detecting bladder cancer through endoscopes.
...
Imagin Medical ... announced today that Dr. Edward Messing, Chair of Urology, University of Rochester Medical Center has joined Imagin’s Scientific Board of Advisors.
Dr. Messing is a renowned expert in the diagnosis and treatment of cancers
of the bladder, prostate, kidney, and other genitourinary organs.
He has conducted extensive research in the basic biology of bladder and prostate cancers
and has been the principal investigator on numerous clinical studies
for the detection, prevention and treatment of genitourinary cancers.
Dr. Messing received his M.D. degree from New York University,
performed his surgery training at
NYU-Bellevue Hospital and his Urology Residency at Stanford University. He was
Director of the Urologic Oncology Program
at the University of Wisconsin
and assumed his current position at
the University of Rochester School of Medicine and Dentistry in
August, 1995.
..."
http://imaginmedical.com/wp-content/uploads/2016/09/Imagin-N…
"... Dr. Edward Messing, Chief of Urology, U Rochester Medical Center
Joins Imagin Medical’s Scientific Board of Advisors
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will establish
a new standard of care for urologists in detecting bladder cancer through endoscopes.
...
Imagin Medical ... announced today that Dr. Edward Messing, Chair of Urology, University of Rochester Medical Center has joined Imagin’s Scientific Board of Advisors.
Dr. Messing is a renowned expert in the diagnosis and treatment of cancers
of the bladder, prostate, kidney, and other genitourinary organs.
He has conducted extensive research in the basic biology of bladder and prostate cancers
and has been the principal investigator on numerous clinical studies
for the detection, prevention and treatment of genitourinary cancers.
Dr. Messing received his M.D. degree from New York University,
performed his surgery training at
NYU-Bellevue Hospital and his Urology Residency at Stanford University. He was
Director of the Urologic Oncology Program
at the University of Wisconsin
and assumed his current position at
the University of Rochester School of Medicine and Dentistry in
August, 1995.
..."
http://imaginmedical.com/wp-content/uploads/2016/10/Imagin-N…
...
Imagin Medical ... announced today that Ken Daignault has joined Imagin’s Board of Directors.
With almost 30 years, experience in the medical device field, with an emphasis in urology,
Mr. Daignault will be welcomed as a valued member of the Board of Directors.
He has held senior level management positions in major medical device companies, most recently with Boston Scientific as Director of R& D, Urology.
He has also held positions at Kendall Healthcare (now Covidien)
and CR Bard, one of the world’s largest urology companies.
Ken has been involved in all aspects of the medical device business,
from product development and the design of protocols and procedures for bench and animal
pre-clinical testing, to building longterm
strategies for multiple - product portfolios at various stages of development.
Ken has strong relationships within the urology field and with key medical institutions,
as well as a track record of bringing products from concept through successful product launch.
“I’m excited to have Ken join our Board
at this stage of the Company’s development,”
said Jim Hutchens, Imagin President and CEO. “
His extensive practical experience in the medical device
field is a perfect match for Imagin’s market focus”.
...
Imagin Medical ... announced today that Ken Daignault has joined Imagin’s Board of Directors.
With almost 30 years, experience in the medical device field, with an emphasis in urology,
Mr. Daignault will be welcomed as a valued member of the Board of Directors.
He has held senior level management positions in major medical device companies, most recently with Boston Scientific as Director of R& D, Urology.
He has also held positions at Kendall Healthcare (now Covidien)
and CR Bard, one of the world’s largest urology companies.
Ken has been involved in all aspects of the medical device business,
from product development and the design of protocols and procedures for bench and animal
pre-clinical testing, to building longterm
strategies for multiple - product portfolios at various stages of development.
Ken has strong relationships within the urology field and with key medical institutions,
as well as a track record of bringing products from concept through successful product launch.
“I’m excited to have Ken join our Board
at this stage of the Company’s development,”
said Jim Hutchens, Imagin President and CEO. “
His extensive practical experience in the medical device
field is a perfect match for Imagin’s market focus”.
http://imaginmedical.com/wp-content/uploads/2016/10/Imagin-N…
"Imagin’s Quality Management System Initial Phase Complete
...
Imagin Medical ... announced today that the Company’s Quality Plan for compliance with U.S.
FDA regulations and International Standard ISO 13485 has been approved by the Company’s management team.
The Quality Management System (QMS) will detail the generation and ma nagement of all the
Company’s standard operating procedures, Design History File documents, and all quality
records to support the development of the i/Blue Imaging system for the detection of bladder cancer.
Design control procedures have been developed and implemented to support and document the prototype development underway at the University of Rochester’s Laboratory
for Laser Energetics.
During the coming months, the Company’s QMS will be expanded to build and document
pilot production units in support of initial bench testing and potential pilot clinical studies.
Advanced manufacturing documentation for eventual commercialization will be developed as
well.
According to Steve Ruggles, Director of QA / RA, “Imagin has assembled a team of brilliant
scientists, doctors, engineers and seasoned medical device management professionals.
Our devices will be designed, tested and manufactured in strict
compliance with all design, risk management and documentation requirements
of the U.S. FDA and other international regulatory bodies for worldwide marketing.”
"Imagin’s Quality Management System Initial Phase Complete
...
Imagin Medical ... announced today that the Company’s Quality Plan for compliance with U.S.
FDA regulations and International Standard ISO 13485 has been approved by the Company’s management team.
The Quality Management System (QMS) will detail the generation and ma nagement of all the
Company’s standard operating procedures, Design History File documents, and all quality
records to support the development of the i/Blue Imaging system for the detection of bladder cancer.
Design control procedures have been developed and implemented to support and document the prototype development underway at the University of Rochester’s Laboratory
for Laser Energetics.
During the coming months, the Company’s QMS will be expanded to build and document
pilot production units in support of initial bench testing and potential pilot clinical studies.
Advanced manufacturing documentation for eventual commercialization will be developed as
well.
According to Steve Ruggles, Director of QA / RA, “Imagin has assembled a team of brilliant
scientists, doctors, engineers and seasoned medical device management professionals.
Our devices will be designed, tested and manufactured in strict
compliance with all design, risk management and documentation requirements
of the U.S. FDA and other international regulatory bodies for worldwide marketing.”
http://imaginmedical.com/wp-content/uploads/2016/10/Imagin-N…
"... IMAGIN MEDICAL CLOSES TRANCHE I OF PRIVATE PLACEMENT FOR $385,800
Imagin Medical ... announced today that, further to its September 13, 2016 press release, it has closed Tranche I of a non-brokered private placement through the issuance of 4,822,500 units (the “Units”) at a price of Cdn $0.08 per Units for gross proceeds of Cdn $385,800 (the “Offering”).
The Company intends to close the remainder of this financing within the next two weeks.
Each Unit consists of one common share of the Company and one warrant (“Warrant”), each Warrant entitling the holder thereof to acquire one additional common share of the Company at a price of $0.12 in the first 12 months and $0.16 in the 12 months thereafter.
“This fundraising demonstrates the confidence our investors have in the value of the
Company’s technology and management’s ability to deliver it to the marketplace.”
commented Jim Hutchens, Imagin’s President and CEO. “It allows us to focus our resources on
development and preparation for the clinical trials.”
The Company intends to use the proceeds from the Offering
for advancing the development of the i/Blue Imaging System for the early detection of cancer through the use of endoscopes and for general working capital.
..."
"... IMAGIN MEDICAL CLOSES TRANCHE I OF PRIVATE PLACEMENT FOR $385,800
Imagin Medical ... announced today that, further to its September 13, 2016 press release, it has closed Tranche I of a non-brokered private placement through the issuance of 4,822,500 units (the “Units”) at a price of Cdn $0.08 per Units for gross proceeds of Cdn $385,800 (the “Offering”).
The Company intends to close the remainder of this financing within the next two weeks.
Each Unit consists of one common share of the Company and one warrant (“Warrant”), each Warrant entitling the holder thereof to acquire one additional common share of the Company at a price of $0.12 in the first 12 months and $0.16 in the 12 months thereafter.
“This fundraising demonstrates the confidence our investors have in the value of the
Company’s technology and management’s ability to deliver it to the marketplace.”
commented Jim Hutchens, Imagin’s President and CEO. “It allows us to focus our resources on
development and preparation for the clinical trials.”
The Company intends to use the proceeds from the Offering
for advancing the development of the i/Blue Imaging System for the early detection of cancer through the use of endoscopes and for general working capital.
..."
http://imaginmedical.com/wp-content/uploads/2016/10/Imagin-N…
"... Imagin Medical announced today that Dr. Liam J. Hurley, a member of the Northeast Urologic Surgery, PC, has joined Imagin’s Scientific Board of Advisors.
Dr. Hurley obtained his Bachelor of Arts degree from Harvard University and his M.D.
from Boston University School of Medicine. After spending three years in the U.S. Navy, he completed a General Surgery Residency at St. Elizabeth's
Medical Center of Boston and his Urology Residency at the Lahey Clinic in Burlington, MA.
He is trained as an adult and pediatric urologist with expertise in genitourinary oncology. Dr. Hurley is a private practice community urologist who has clinical affiliations with Tufts New England Medical Center and Lahey Clinic.
Other accomplishments include launching The Boston Urologic Society, serving on the Executive Committee for the Massachusetts Association of Practicing Urologists, and presiding as the
Eastern Massachusetts representative to the New England section of the AUA.
Dr. Hurley is Board certified in Urology and is a Fellow of the American College of Surgeons. In
2006, he was chosen as one of Boston 's “TOP DOCTORS” by Boston Magazine.“
Dr. Hurley’s knowledge and expertise in the urology field will be a great asset to our Advisory Board,” said Jim Hutchens, Imagin President and CEO.
“We look forward to his joining our team as we continue to add relevant expertise to help Imagin succeed.”
"... Imagin Medical announced today that Dr. Liam J. Hurley, a member of the Northeast Urologic Surgery, PC, has joined Imagin’s Scientific Board of Advisors.
Dr. Hurley obtained his Bachelor of Arts degree from Harvard University and his M.D.
from Boston University School of Medicine. After spending three years in the U.S. Navy, he completed a General Surgery Residency at St. Elizabeth's
Medical Center of Boston and his Urology Residency at the Lahey Clinic in Burlington, MA.
He is trained as an adult and pediatric urologist with expertise in genitourinary oncology. Dr. Hurley is a private practice community urologist who has clinical affiliations with Tufts New England Medical Center and Lahey Clinic.
Other accomplishments include launching The Boston Urologic Society, serving on the Executive Committee for the Massachusetts Association of Practicing Urologists, and presiding as the
Eastern Massachusetts representative to the New England section of the AUA.
Dr. Hurley is Board certified in Urology and is a Fellow of the American College of Surgeons. In
2006, he was chosen as one of Boston 's “TOP DOCTORS” by Boston Magazine.“
Dr. Hurley’s knowledge and expertise in the urology field will be a great asset to our Advisory Board,” said Jim Hutchens, Imagin President and CEO.
“We look forward to his joining our team as we continue to add relevant expertise to help Imagin succeed.”
Antwort auf Beitrag Nr.: 53.573.034 von praesens am 28.10.16 11:30:11das Unternehmen hält das ein, was es verspricht...weiter so...
es wird nicht lange dauern, dann kommt wieder so ein Pusher wie Aktiencheck und nimmt die Story auf... dann sieht man hier wieder mehr Bewegung ...
es wird nicht lange dauern, dann kommt wieder so ein Pusher wie Aktiencheck und nimmt die Story auf... dann sieht man hier wieder mehr Bewegung ...
Antwort auf Beitrag Nr.: 53.573.976 von OliKo am 28.10.16 13:34:16Ja, die "Unvermeidlichen" ... 
Gruß, praesens

Gruß, praesens
Hat sich nach der Präsentation des Prototyos nix mehr getan? Forum ist tot und keine Nachrichten.
Nach langem dahindämmern kommt wieder Bewegung rein.
Und richtig Gute !
Endlich Zulassung zu human trials !
Aber der Reihe nach ...
Und richtig Gute !
Endlich Zulassung zu human trials !
Aber der Reihe nach ...

https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2509…
"Imagin arranges $600,000 financing
2017-09-26 17:00 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES PRIVATE PLACEMENT
Imagin Medical, Inc. has arranged a non-brokered private placement to raise $600,000 through the distribution of units at five cents per unit.
Each unit will consist of one common share and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share at 10 cents for 12 months.
Finder's fees may be paid in accordance with Canadian Securities Exchange policies. Closing of the placement will be subject to making applicable filings with the CSE.
Proceeds from the placement will be used for general working capital purposes, including advancing the development of imaging solutions for the early detection of cancer.
Pursuant to the company's stock option plan, an aggregate of 1.6 million shares have been granted to consultants as incentive stock options at an exercise price of six cents per share. The options are exercisable for a period of five years, ending on Sept. 25, 2022.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
"Imagin arranges $600,000 financing
2017-09-26 17:00 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES PRIVATE PLACEMENT
Imagin Medical, Inc. has arranged a non-brokered private placement to raise $600,000 through the distribution of units at five cents per unit.
Each unit will consist of one common share and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share at 10 cents for 12 months.
Finder's fees may be paid in accordance with Canadian Securities Exchange policies. Closing of the placement will be subject to making applicable filings with the CSE.
Proceeds from the placement will be used for general working capital purposes, including advancing the development of imaging solutions for the early detection of cancer.
Pursuant to the company's stock option plan, an aggregate of 1.6 million shares have been granted to consultants as incentive stock options at an exercise price of six cents per share. The options are exercisable for a period of five years, ending on Sept. 25, 2022.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2510…
"Imagin receives OK for first i/Blue in-human study
2017-09-27 16:37 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL RECEIVES APPROVAL FOR 1ST IN-HUMAN RESEARCH STUDY
The University of Rochester (UR) Research Study Review Board (RSRB) has approved the first in-human study using Imagin Medical Inc.'s i/Blue imaging system. Recruitment of subjects will begin immediately and continue until the study is completed, which is expected to be a three-month process. More information can be found on the Clinical Trials website.
The pilot study, entitled "Near Infrared Fluorescence Imaging for Bladder Cancer Detection," is sponsored by Dr. Edward Messing, chair of urology at the University of Rochester Medical Center and the study's principal investigator. The primary purpose of the study is to determine the minimal dwell time needed for adequate detection of hexaminolevinulate HCL avid tumours using protoporphyrin IX (PpIX) near infrared fluorescence (NIRF).
The company has expressed confidence that the 10-patient study will validate previous bench-testing results that have shown: (1) physicians will be able to see the cancer in 10 minutes versus the full hour required by today's technology; and (2) the white and fluorescence images will blend into one to put the cancer into context within the bladder.
"We're very excited to receive the approval. It's a major milestone for Imagin and we're eager to see the study begin," said Jim Hutchens, Imagin's president and chief executive officer. "Based on previous testing, we anticipate positive results that will bring us closer to delivering this life-saving technology to the marketplace."
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer around the world, the sixth-most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
"Imagin receives OK for first i/Blue in-human study
2017-09-27 16:37 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL RECEIVES APPROVAL FOR 1ST IN-HUMAN RESEARCH STUDY
The University of Rochester (UR) Research Study Review Board (RSRB) has approved the first in-human study using Imagin Medical Inc.'s i/Blue imaging system. Recruitment of subjects will begin immediately and continue until the study is completed, which is expected to be a three-month process. More information can be found on the Clinical Trials website.
The pilot study, entitled "Near Infrared Fluorescence Imaging for Bladder Cancer Detection," is sponsored by Dr. Edward Messing, chair of urology at the University of Rochester Medical Center and the study's principal investigator. The primary purpose of the study is to determine the minimal dwell time needed for adequate detection of hexaminolevinulate HCL avid tumours using protoporphyrin IX (PpIX) near infrared fluorescence (NIRF).
The company has expressed confidence that the 10-patient study will validate previous bench-testing results that have shown: (1) physicians will be able to see the cancer in 10 minutes versus the full hour required by today's technology; and (2) the white and fluorescence images will blend into one to put the cancer into context within the bladder.
"We're very excited to receive the approval. It's a major milestone for Imagin and we're eager to see the study begin," said Jim Hutchens, Imagin's president and chief executive officer. "Based on previous testing, we anticipate positive results that will bring us closer to delivering this life-saving technology to the marketplace."
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer around the world, the sixth-most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
Nach einem Kursanstieg gestern von 6 cent auf 10 cent kam ein trading halt.
Geht aber heute weiter:
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2510…
"Imagin Medical to resume trading Sept. 28
2017-09-27 17:05 ET - Resume Trading
Imagin Medical Inc. wil resume trading, effective at 5 a.m. PT on Sept. 28, 2017.
© 2017 Canjex Publishing Ltd. All rights reserved."
Geht aber heute weiter:
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2510…
"Imagin Medical to resume trading Sept. 28
2017-09-27 17:05 ET - Resume Trading
Imagin Medical Inc. wil resume trading, effective at 5 a.m. PT on Sept. 28, 2017.
© 2017 Canjex Publishing Ltd. All rights reserved."
Antwort auf Beitrag Nr.: 55.843.047 von praesens am 28.09.17 15:18:00Weiß man schon wann das Trading weiter geht? Die Foren waren ja über das Jahr so gut wie tot
Antwort auf Beitrag Nr.: 55.844.202 von Poetfan am 28.09.17 17:03:50... guggst Du post über Deinem ... 
Gruß, praesens

Gruß, praesens
Antwort auf Beitrag Nr.: 55.844.382 von praesens am 28.09.17 17:28:59Ok, dachte nach unserer Zeit, sorry
Antwort auf Beitrag Nr.: 55.844.862 von praesens am 28.09.17 18:18:55Zum aktuellen sharecount:
Laut letztem sedar Eintrag 78 mio fully diluted.
Zzgl. akuellem pp wären wir dann bei ca. 102 mio.
Gruß, praesens
Laut letztem sedar Eintrag 78 mio fully diluted.
Zzgl. akuellem pp wären wir dann bei ca. 102 mio.
Gruß, praesens
Antwort auf Beitrag Nr.: 55.867.561 von praesens am 02.10.17 20:54:31http://far-resources.conversionir.com/landing-1/?gclid=CO2D4…
"Imagin closes $732,940 first tranche of financing
2017-10-04 13:42 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING TRANCHE 1 AND INCREASING PRIVATE PLACEMENT OFFERING
Further to its press release of Sept. 26, 2017, Imagin Medical Inc. is closing tranche 1 of its non-brokered private placement with the issuance of 14,658,800 units priced at five cents per unit for gross proceeds of $732,940. Due to oversubscription, the company has filed notice with the Canadian Securities Exchange to increase the private placement above $600,000. The second and final tranche will close imminently.
Each unit of this private placement will consist of one common share of the company and one common share purchase warrant, with each warrant entitling the holder to acquire one additional common share of the company at a price of 10 cents within the 12 months.
Finders' fees, in the form of warrants and cash, will be paid upon final closing. Proceeds from the offering will be used for general working capital purposes, including clinical trials and advancing the development of the i/Blue imaging system.
"We're very excited about the response to the private placement. It expresses confidence the company," said Jim Hutchens, Imagin's president and chief executive officer.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. Imagin's initial target market is bladder cancer, a major cancer around the world, the sixth-most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
"Imagin closes $732,940 first tranche of financing
2017-10-04 13:42 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING TRANCHE 1 AND INCREASING PRIVATE PLACEMENT OFFERING
Further to its press release of Sept. 26, 2017, Imagin Medical Inc. is closing tranche 1 of its non-brokered private placement with the issuance of 14,658,800 units priced at five cents per unit for gross proceeds of $732,940. Due to oversubscription, the company has filed notice with the Canadian Securities Exchange to increase the private placement above $600,000. The second and final tranche will close imminently.
Each unit of this private placement will consist of one common share of the company and one common share purchase warrant, with each warrant entitling the holder to acquire one additional common share of the company at a price of 10 cents within the 12 months.
Finders' fees, in the form of warrants and cash, will be paid upon final closing. Proceeds from the offering will be used for general working capital purposes, including clinical trials and advancing the development of the i/Blue imaging system.
"We're very excited about the response to the private placement. It expresses confidence the company," said Jim Hutchens, Imagin's president and chief executive officer.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. Imagin's initial target market is bladder cancer, a major cancer around the world, the sixth-most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
Antwort auf Beitrag Nr.: 55.880.707 von praesens am 04.10.17 20:02:04ENTSCHULDIGUNG !
Der link im letzten post ist mir aus Versehen reingerutscht. Hier der Richtige:
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2512…
Gruß, praesens
Der link im letzten post ist mir aus Versehen reingerutscht. Hier der Richtige:
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2512…
Gruß, praesens
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2513…
"2017-10-05 04:41 ET - News Release
Mr. Jim Hutchens reports
RESEARCH STUDY LISTED ON GOVERNMENT WEBSITE
The information about the coming clinical research study at the University of Rochester announced in a press release on Sept. 27, 2017, can be found on a government website maintained by the National Library of Medicine (NLM) at the National Institutes of Health (NIH).
The pilot study entitled, Near Infrared Fluorescence Imaging for Bladder Cancer Detection, is sponsored by Dr. Edward Messing, Chair of Urology at the University of Rochester Medical Center, and the study's principal investigator. The primary purpose of the study is to determine the minimal dwell time needed for adequate detection of cancerous cells using protoporphyrin IX (PpIX) near infrared fluorescence (NIRF). Imagin anticipates the study will prove that surgeons will be able to "see" the cancerous cells in less than 15 minutes, one of the key features of the i/Blue technology. The Study will also evaluate another feature of the i/Blue System, the patented Simultaneous Acquisition of Differing Images, which will blend the white light and fluorescing images into one, enabling the surgeon, in one step, to identify the precise location of the of the cancerous cells within the bladder.
"We're eager for this study to begin," commented Jim Hutchens, Imagin Medical's President and CEO. "We expect the outcome to confirm our bench test results and build on the company's momentum."
The NIH database contains information about medical studies in human volunteers for both federally and privately funded trials, describing the purpose of the studies conducted in all 50 States and in 197 countries. Each study record presents summary information about the study protocol, including the medical product, behavior, or procedure being studied, as well as requirements for participation and study outcomes.
..."
"2017-10-05 04:41 ET - News Release
Mr. Jim Hutchens reports
RESEARCH STUDY LISTED ON GOVERNMENT WEBSITE
The information about the coming clinical research study at the University of Rochester announced in a press release on Sept. 27, 2017, can be found on a government website maintained by the National Library of Medicine (NLM) at the National Institutes of Health (NIH).
The pilot study entitled, Near Infrared Fluorescence Imaging for Bladder Cancer Detection, is sponsored by Dr. Edward Messing, Chair of Urology at the University of Rochester Medical Center, and the study's principal investigator. The primary purpose of the study is to determine the minimal dwell time needed for adequate detection of cancerous cells using protoporphyrin IX (PpIX) near infrared fluorescence (NIRF). Imagin anticipates the study will prove that surgeons will be able to "see" the cancerous cells in less than 15 minutes, one of the key features of the i/Blue technology. The Study will also evaluate another feature of the i/Blue System, the patented Simultaneous Acquisition of Differing Images, which will blend the white light and fluorescing images into one, enabling the surgeon, in one step, to identify the precise location of the of the cancerous cells within the bladder.
"We're eager for this study to begin," commented Jim Hutchens, Imagin Medical's President and CEO. "We expect the outcome to confirm our bench test results and build on the company's momentum."
The NIH database contains information about medical studies in human volunteers for both federally and privately funded trials, describing the purpose of the studies conducted in all 50 States and in 197 countries. Each study record presents summary information about the study protocol, including the medical product, behavior, or procedure being studied, as well as requirements for participation and study outcomes.
..."
Hier nochmal eine Präsentation des i/Blue Imaging Systems.
Es geht um eine bessere Erkennbarkeit von Krebszellen:
http://imaginmedical.com/technology/
Es geht um eine bessere Erkennbarkeit von Krebszellen:
http://imaginmedical.com/technology/
!
Dieser Beitrag wurde von MODelfin moderiert. Grund: themenfremd, Werbung
http://imaginmedical.com/wp-content/uploads/2017/10/Press-Re…
Ich stelle nur den link ein und nicht die "news" in Textform, da die Formatierung so gar nicht geht.
Thema: Praktizierende Urologen partizipieren am aktuellen pp.
Gruß, praesens
Ich stelle nur den link ein und nicht die "news" in Textform, da die Formatierung so gar nicht geht.
Thema: Praktizierende Urologen partizipieren am aktuellen pp.
Gruß, praesens
Antwort auf Beitrag Nr.: 55.932.260 von praesens am 11.10.17 20:56:10https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2515…
"Imagin closes $357,060 second tranche of financing
2017-10-13 11:54 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING SECOND AND FINAL TRANCHE OF $1,000,000 PRIVATE PLACEMENT
Imagin Medical, Inc. has closed the second and final tranche of its non-brokered private placement with the issuance of 7,141,200 units priced at five cents per unit for gross proceeds of $357,060. Due to oversubscription, the company will be closing on a total of 20 million units for aggregate gross proceeds of $1-million.
Each unit of this private placement consists of one common share of the company and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share of the company at a price of 10 cents within 12 months from the date of issue.
Total finders' fees for tranches one and two have been paid in the form of cash in the amount $20,240 and 404,800 finders' warrants (exercisable at 10 cents per share for 12 months). All securities issued will be subject to a four-month hold period. Proceeds from the offering will be used for general working capital purposes, including clinical trials and advancing the development of the i/Blue imaging system.
"The success of this private placement speaks volumes about the confidence our investors have in our technology and the future of the company in the marketplace," said Jim Hutchens, Imagin's president and chief executive officer.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
"Imagin closes $357,060 second tranche of financing
2017-10-13 11:54 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING SECOND AND FINAL TRANCHE OF $1,000,000 PRIVATE PLACEMENT
Imagin Medical, Inc. has closed the second and final tranche of its non-brokered private placement with the issuance of 7,141,200 units priced at five cents per unit for gross proceeds of $357,060. Due to oversubscription, the company will be closing on a total of 20 million units for aggregate gross proceeds of $1-million.
Each unit of this private placement consists of one common share of the company and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share of the company at a price of 10 cents within 12 months from the date of issue.
Total finders' fees for tranches one and two have been paid in the form of cash in the amount $20,240 and 404,800 finders' warrants (exercisable at 10 cents per share for 12 months). All securities issued will be subject to a four-month hold period. Proceeds from the offering will be used for general working capital purposes, including clinical trials and advancing the development of the i/Blue imaging system.
"The success of this private placement speaks volumes about the confidence our investors have in our technology and the future of the company in the marketplace," said Jim Hutchens, Imagin's president and chief executive officer.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
http://usanewsgroup.com/2017/10/16/checkpoint-inhibitors-sho…
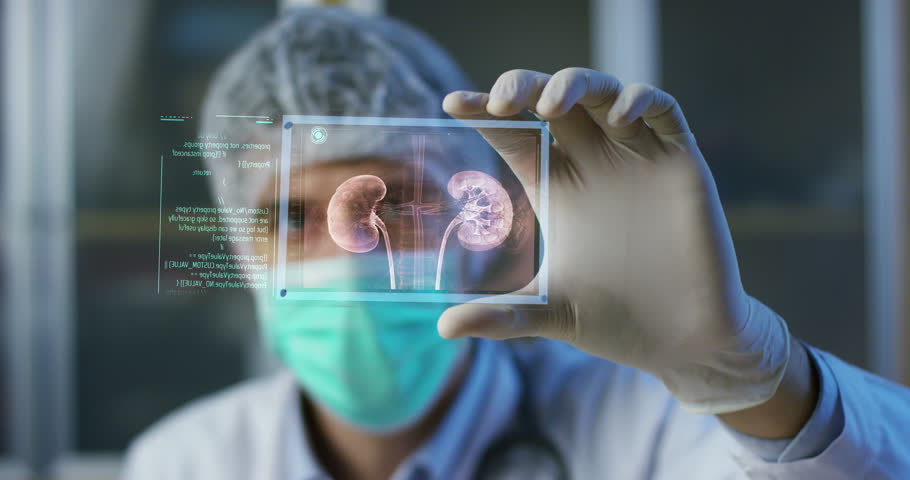
"Small Medical Wonders Hold Promise for Big Profits
Before we discuss this major technology shift, we need to add an important footnote: the company we discuss, Imagin Medical Inc. (CSE: IME OTC: IMEXF), just announced approval of human testing for their product.
...
We add this note because the news is not widely known and puts the company at a completely different stage of development.
Imagin Medical’s stock has not noticeably changed, likely because the news is so new and hasn’t been publicized except to peers in the medical industry.
Companies in the unique position of being in clinical testing with a breakthrough product who are virtually unknown don’t come along every day. We urge our readers to carefully consider that there are few public medical companies this operationally advanced – including in human testing with a fully patented medical system – but trading at pennies per share.
Imagin Medical is a Revolution in The Making
Can you remember the last time you watched a low-res, cathode ray TV? You might or you may be too young to have even experienced that ancient technology. But you certainly can remember experiencing the clarity and detail that came to life when you saw your first HD movie on a 1080i flat screen… and perhaps more recently when you saw an Ultra-High-Definition 4K TV at full resolution.
That is the kind of difference that Imagin Medical Inc. is bringing to the medical imaging and diagnostics market for the treatment of one of the most widespread and costly forms of disease: bladder cancer.
And that’s why its diagnostic system can revolutionize treatment.
Imagin Medical Inc. is an emerging medical imaging company that we think could make a fortune for investors who take advantage of its early stage development. We view this as a biotech style breakthrough without the many hurdles associated with biotech development and with a direct path to product sales as early as 2019.
Medical Imaging’s New Reality
Just as the other digital technologies have been advancing in leaps and bounds, so too has medical imaging. You’ve probably noticed this everywhere from your dentist’s chair, where x-rays now arrive on the screen, to orthoscopic surgery as an in-patient or even in a small clinic. Medical imaging and the diagnostics that it supports are being totally transformed.
There are powerful, new technologies and techniques that are being adopted almost daily. A select few companies have taken the opportunity to draw on the new capabilities and are developing patented approaches that can forever change the way that diseases are diagnosed and treated.
This is the calling of Imagin Medical Inc. and their i/Blue™ Imaging System for the imaging and treatment of tumors targeting bladder cancer.
You won’t likely know about Imagin Medical yet. Their patented i/Blue™ Imaging System just passed through the prototype stage and is about to go into human testing. For our readers, that’s a good thing. Since it’s not widely known, you can accumulate stock at pennies a share– well before the wider market takes notice and creates demand.
We’ve spelled out why we think the situation is a great opportunity that holds the potential for big profits.
One Hundred Times More Accurate at Visualizing Tumors
Imagin Medical is the developer of the i/Blue™ Imaging System to help detect bladder cancer and reduce its recurrence by dramatically improving the urologist’s ability to visualize, identify and then remove cancerous cells.
Here’s how the technology works:
With the development of compact color video cameras over 30 years, urologists have adopted endoscopes (or cystoscopes) combined with white light and video imaging to visualize the interior of the human bladder in order to detect cancer.
Using this technology, providers established that malignant lesions may not be visible and not detected during endoscopic examination alone. So in 2010, the FDA approved a combination of fluorescing drugs and specialized blue light imaging to improve the visualization of tumors.
Bladder cancer tumor, visualized through an endoscope using white light (left image) and blue light using Photocure’s chemical agent called Cysview®* (right image)

Cancerous tumor not visible with traditional white light

Imaging agents improve cancer detection, but not without limitations
Without question, this method produces better visualization and detection of malignant lesions. However, the available technology has several issues that are slowing the wide adoption of this technology.
The main drawbacks besides an inability to more accurately visualize the tumors, are the time needed to absorb fluorescing drugs and the inability to view multiple perspectives of the bladder at the same time.
Imagin Medical’s i/Blue™ patented computer technology, white light and near-infrared fluorescence overcomes all of these shortfalls.
The company’s approach allows the system to accurately image in less than 15 minutes vs. the one-hour period required to metabolize the drug using conventional fluorescing.
Current imaging technology offers only blue filtered images showing a highlighted fluorescence image of the cancer, but without a precise indication of where it is located in the bladder. To see that, the surgeon needs a white light only view to show the full landscape of the bladder, causing them to have to switch back and forth between the white and blue light images on separate systems.
Imagin Medical’s computer application cleans, reprocesses, blends and displays the image of the bladder and cancer onto a single monitor at the same time.
Most importantly, Imagin’s i/Blue™ system increases sensitivity for detecting the cancer specific contrast agent by 5 orders of magnitude or in terms of visibility, 100,000X.
This advancement in sensitivity is vitally important because it improves the surgeon’s ability to detect even flat cancers and to visualize their margins for more complete removal. Ultimately this can dramatically reduce the risk of recurrence, slowing the progression of the disease.(2)
Simply a Better Solution: Less Invasive, Less Costly, More Effective
Advancements in medical technology are crucial, but if they are impractical or too expensive, they simply won’t find their way into the medical system. It’s a fact.
What’s really exciting about the i/Blue™ System is its ability to improve results, simplify procedures, save time, and save money. And in the healthcare system, that is always going to be a major factor.
With a shorter period to review initial diagnostics (roughly 1/3 previous requirements), the time savings is expected to increase the efficiency of the Operating Room markedly. Since it is minimally invasive, the i/Blue™ System will also enable diagnostic and surgical procedures to be performed in the less expensive physician’s office for follow-up exams.
In the end, the recurring nature of the bladder cancer due to missed or undetected tumor removal has made it the most expensive form out of treatable cancers.
Through more effective diagnostics and vastly improved imaging, the i/Blue™ system can have a profound impact on the outcomes from surgery to remove tumors and thereby seriously reduce the cost of treatment or re-treatment

The i/Blue™ System is such a giant leap forward in the medical imaging space that it qualifies as a disruptive technology
Heavyweights in The Field Doing What They Do Best
Imagin Medical’s products are based on the technology invented by Dr. Stavros Demos at the Lawrence Livermore National Laboratory (LLNL).
Dr. Demos worked in collaboration with the UC Davis Comprehensive Cancer Center and Dr. Ralph deVere White, one of the world’s leading authorities on bladder cancer, for more than five years to determine the system’s feasibility.
Imagin Medical has now licensed the technology from LLNL and moved the final stages of development to the University of Rochester Laboratory for Laser Energetics (LLE).
We don’t often detail the backgrounds of management, but in this case we think it’s important to understand the professional level of the team responsible for the current and future path of Imagin Medical and its’ i/Blue™ technology.
Imagin’s management have successfully forwarded numerous products and valuable research to the medical field over their lengthy careers including:
Dr. Stavros G. Demos is the technical advisor to the company. He is a prolific inventor and scholar, as well as a laser materials expert. Companies using his technologies include Muse Microscopy, Near Infrared Imaging, and Biosense/Webster. Dr. Demos holds 20 patents and has published more than 115 scholarly journal articles.
Dr. Stavros is the original inventor of the i/Blue™ system and is continuing to work with Imagin in developing the technology. Imagin has an agreement with UC Davis and the University of Rochester Laboratory for Laser Energetics where Dr. Demos continues to support Imagin’s development team through clinical evaluations and into commercialization.
Dr. Ralph deVere White who serves as Imagin’s Medical Advisor, is one of the world’s foremost authorities on bladder cancer. He has authored more than 300 peer-reviewed scientific articles. He is also the director of the UC Davis Comprehensive Cancer Center and a professor of urology at the university. He will actively consult with the Imagin team about bladder cancer.
Rounding out this impressive group is Dr. Edward Messing, Chief of Urology at the University of Rochester Medical Center.
Dr. Messing is a renowned expert in the diagnosis and treatment of cancers of the bladder, prostate, kidney, and other genitourinary organs. He has conducted extensive research in the basic biology of bladder and prostate cancers and has been the principal investigator on numerous clinical studies for the detection, prevention and treatment of genitourinary cancers.
Drs. Demos and deVere White continue to the lead in the guidance of testing and development of the i/Blue™ System. Dr. Messing will sponsor the Clinical Research Study at the University of Rochester using i/Blue Technology.(3)
Bladder Cancer is the 6th Largest Cancer Market
Imagin Medical participates in the enormous medical imaging, diagnostics and treatment market. The US spends $4.5 billion per year on imaging alone. The diagnostic imaging segment has seen continual growth based on the increasing number of patients, as well as an increasing demand for minimally invasive surgery, which requires precise imaging.
Increasing investments from public-private organizations; growth in the number of diagnostic imaging centers; rising prevalence of cancer; increasing geriatric population and the subsequent growth in the incidence of various diseases; technological advancements in diagnostic imaging modalities; and increasing preference for minimally invasive treatments all drive the growth of this market.
More specifically, the i/Blue™ System is targeted initially at imaging and treatment of bladder cancer.
This year, an estimated 79,030 adults (60,490 men and 18,540 women) will be diagnosed with bladder cancer in the United States. It is estimated that 16,870 deaths from this disease will occur this year.
The treatment of bladder cancer is the most expensive form of cancer to treat, accounting for cumulative costs of $4 billion or about 3.2% of all cancer care each year. It is the sixth largest market for cancer treatment and yet treatment approaches have not changed significantly over the past 25 years.
Healthcare providers and the medical system are desperately seeking ways to reduce the cost of treatment for bladder cancer and provide better, more effective diagnostics and monitoring, since it often requires multiple treatments and continuous follow up over its course. (4)
Potential for Additional Applications Adds Major Value
While Imagin is initially focusing on the bladder cancer imaging market, the company expects that its dual-light, single-screen imaging system will set new standards of care for doctors and surgeons in detecting many other kinds of cancers and conditions.
These may include laparoscopic (general and gynecology), colorectal and thoracic procedures related to cancer and non-cancerous conditions.
According to Stratistics MRC, the Global Medical Imaging market is accounted for $29.8 billion in 2016 and is expected to reach $45.1 billion by 2022. (5)
The US represents the largest market where each day 10,000 people turn 65 years old. The impact of The Affordable Care Act (“Obamacare”) is also providing access to additional services like imaging for more of the population.
The Asia-Pacific region is the fastest growing area. This region is driven by the massive population base and the many environmental health issues created by rapid industrialization.
China is evolving into a massive market for healthcare products in dire need of effective diagnostics and treatments. This one market alone is a potential gold mine for IME to expand the application of its patented technology.
..."
"Small Medical Wonders Hold Promise for Big Profits

Before we discuss this major technology shift, we need to add an important footnote: the company we discuss, Imagin Medical Inc. (CSE: IME OTC: IMEXF), just announced approval of human testing for their product.
...
We add this note because the news is not widely known and puts the company at a completely different stage of development.
Imagin Medical’s stock has not noticeably changed, likely because the news is so new and hasn’t been publicized except to peers in the medical industry.
Companies in the unique position of being in clinical testing with a breakthrough product who are virtually unknown don’t come along every day. We urge our readers to carefully consider that there are few public medical companies this operationally advanced – including in human testing with a fully patented medical system – but trading at pennies per share.
Imagin Medical is a Revolution in The Making
Can you remember the last time you watched a low-res, cathode ray TV? You might or you may be too young to have even experienced that ancient technology. But you certainly can remember experiencing the clarity and detail that came to life when you saw your first HD movie on a 1080i flat screen… and perhaps more recently when you saw an Ultra-High-Definition 4K TV at full resolution.
That is the kind of difference that Imagin Medical Inc. is bringing to the medical imaging and diagnostics market for the treatment of one of the most widespread and costly forms of disease: bladder cancer.
And that’s why its diagnostic system can revolutionize treatment.
Imagin Medical Inc. is an emerging medical imaging company that we think could make a fortune for investors who take advantage of its early stage development. We view this as a biotech style breakthrough without the many hurdles associated with biotech development and with a direct path to product sales as early as 2019.
Medical Imaging’s New Reality
Just as the other digital technologies have been advancing in leaps and bounds, so too has medical imaging. You’ve probably noticed this everywhere from your dentist’s chair, where x-rays now arrive on the screen, to orthoscopic surgery as an in-patient or even in a small clinic. Medical imaging and the diagnostics that it supports are being totally transformed.
There are powerful, new technologies and techniques that are being adopted almost daily. A select few companies have taken the opportunity to draw on the new capabilities and are developing patented approaches that can forever change the way that diseases are diagnosed and treated.
This is the calling of Imagin Medical Inc. and their i/Blue™ Imaging System for the imaging and treatment of tumors targeting bladder cancer.
You won’t likely know about Imagin Medical yet. Their patented i/Blue™ Imaging System just passed through the prototype stage and is about to go into human testing. For our readers, that’s a good thing. Since it’s not widely known, you can accumulate stock at pennies a share– well before the wider market takes notice and creates demand.
We’ve spelled out why we think the situation is a great opportunity that holds the potential for big profits.
One Hundred Times More Accurate at Visualizing Tumors
Imagin Medical is the developer of the i/Blue™ Imaging System to help detect bladder cancer and reduce its recurrence by dramatically improving the urologist’s ability to visualize, identify and then remove cancerous cells.
Here’s how the technology works:
With the development of compact color video cameras over 30 years, urologists have adopted endoscopes (or cystoscopes) combined with white light and video imaging to visualize the interior of the human bladder in order to detect cancer.
Using this technology, providers established that malignant lesions may not be visible and not detected during endoscopic examination alone. So in 2010, the FDA approved a combination of fluorescing drugs and specialized blue light imaging to improve the visualization of tumors.
Bladder cancer tumor, visualized through an endoscope using white light (left image) and blue light using Photocure’s chemical agent called Cysview®* (right image)

Cancerous tumor not visible with traditional white light

Imaging agents improve cancer detection, but not without limitations
Without question, this method produces better visualization and detection of malignant lesions. However, the available technology has several issues that are slowing the wide adoption of this technology.
The main drawbacks besides an inability to more accurately visualize the tumors, are the time needed to absorb fluorescing drugs and the inability to view multiple perspectives of the bladder at the same time.
Imagin Medical’s i/Blue™ patented computer technology, white light and near-infrared fluorescence overcomes all of these shortfalls.
The company’s approach allows the system to accurately image in less than 15 minutes vs. the one-hour period required to metabolize the drug using conventional fluorescing.
Current imaging technology offers only blue filtered images showing a highlighted fluorescence image of the cancer, but without a precise indication of where it is located in the bladder. To see that, the surgeon needs a white light only view to show the full landscape of the bladder, causing them to have to switch back and forth between the white and blue light images on separate systems.
Imagin Medical’s computer application cleans, reprocesses, blends and displays the image of the bladder and cancer onto a single monitor at the same time.
Most importantly, Imagin’s i/Blue™ system increases sensitivity for detecting the cancer specific contrast agent by 5 orders of magnitude or in terms of visibility, 100,000X.
This advancement in sensitivity is vitally important because it improves the surgeon’s ability to detect even flat cancers and to visualize their margins for more complete removal. Ultimately this can dramatically reduce the risk of recurrence, slowing the progression of the disease.(2)
Simply a Better Solution: Less Invasive, Less Costly, More Effective
Advancements in medical technology are crucial, but if they are impractical or too expensive, they simply won’t find their way into the medical system. It’s a fact.
What’s really exciting about the i/Blue™ System is its ability to improve results, simplify procedures, save time, and save money. And in the healthcare system, that is always going to be a major factor.
With a shorter period to review initial diagnostics (roughly 1/3 previous requirements), the time savings is expected to increase the efficiency of the Operating Room markedly. Since it is minimally invasive, the i/Blue™ System will also enable diagnostic and surgical procedures to be performed in the less expensive physician’s office for follow-up exams.
In the end, the recurring nature of the bladder cancer due to missed or undetected tumor removal has made it the most expensive form out of treatable cancers.
Through more effective diagnostics and vastly improved imaging, the i/Blue™ system can have a profound impact on the outcomes from surgery to remove tumors and thereby seriously reduce the cost of treatment or re-treatment

The i/Blue™ System is such a giant leap forward in the medical imaging space that it qualifies as a disruptive technology
Heavyweights in The Field Doing What They Do Best
Imagin Medical’s products are based on the technology invented by Dr. Stavros Demos at the Lawrence Livermore National Laboratory (LLNL).
Dr. Demos worked in collaboration with the UC Davis Comprehensive Cancer Center and Dr. Ralph deVere White, one of the world’s leading authorities on bladder cancer, for more than five years to determine the system’s feasibility.
Imagin Medical has now licensed the technology from LLNL and moved the final stages of development to the University of Rochester Laboratory for Laser Energetics (LLE).
We don’t often detail the backgrounds of management, but in this case we think it’s important to understand the professional level of the team responsible for the current and future path of Imagin Medical and its’ i/Blue™ technology.
Imagin’s management have successfully forwarded numerous products and valuable research to the medical field over their lengthy careers including:
Dr. Stavros G. Demos is the technical advisor to the company. He is a prolific inventor and scholar, as well as a laser materials expert. Companies using his technologies include Muse Microscopy, Near Infrared Imaging, and Biosense/Webster. Dr. Demos holds 20 patents and has published more than 115 scholarly journal articles.
Dr. Stavros is the original inventor of the i/Blue™ system and is continuing to work with Imagin in developing the technology. Imagin has an agreement with UC Davis and the University of Rochester Laboratory for Laser Energetics where Dr. Demos continues to support Imagin’s development team through clinical evaluations and into commercialization.
Dr. Ralph deVere White who serves as Imagin’s Medical Advisor, is one of the world’s foremost authorities on bladder cancer. He has authored more than 300 peer-reviewed scientific articles. He is also the director of the UC Davis Comprehensive Cancer Center and a professor of urology at the university. He will actively consult with the Imagin team about bladder cancer.
Rounding out this impressive group is Dr. Edward Messing, Chief of Urology at the University of Rochester Medical Center.
Dr. Messing is a renowned expert in the diagnosis and treatment of cancers of the bladder, prostate, kidney, and other genitourinary organs. He has conducted extensive research in the basic biology of bladder and prostate cancers and has been the principal investigator on numerous clinical studies for the detection, prevention and treatment of genitourinary cancers.
Drs. Demos and deVere White continue to the lead in the guidance of testing and development of the i/Blue™ System. Dr. Messing will sponsor the Clinical Research Study at the University of Rochester using i/Blue Technology.(3)
Bladder Cancer is the 6th Largest Cancer Market
Imagin Medical participates in the enormous medical imaging, diagnostics and treatment market. The US spends $4.5 billion per year on imaging alone. The diagnostic imaging segment has seen continual growth based on the increasing number of patients, as well as an increasing demand for minimally invasive surgery, which requires precise imaging.
Increasing investments from public-private organizations; growth in the number of diagnostic imaging centers; rising prevalence of cancer; increasing geriatric population and the subsequent growth in the incidence of various diseases; technological advancements in diagnostic imaging modalities; and increasing preference for minimally invasive treatments all drive the growth of this market.
More specifically, the i/Blue™ System is targeted initially at imaging and treatment of bladder cancer.
This year, an estimated 79,030 adults (60,490 men and 18,540 women) will be diagnosed with bladder cancer in the United States. It is estimated that 16,870 deaths from this disease will occur this year.
The treatment of bladder cancer is the most expensive form of cancer to treat, accounting for cumulative costs of $4 billion or about 3.2% of all cancer care each year. It is the sixth largest market for cancer treatment and yet treatment approaches have not changed significantly over the past 25 years.
Healthcare providers and the medical system are desperately seeking ways to reduce the cost of treatment for bladder cancer and provide better, more effective diagnostics and monitoring, since it often requires multiple treatments and continuous follow up over its course. (4)
Potential for Additional Applications Adds Major Value
While Imagin is initially focusing on the bladder cancer imaging market, the company expects that its dual-light, single-screen imaging system will set new standards of care for doctors and surgeons in detecting many other kinds of cancers and conditions.
These may include laparoscopic (general and gynecology), colorectal and thoracic procedures related to cancer and non-cancerous conditions.
According to Stratistics MRC, the Global Medical Imaging market is accounted for $29.8 billion in 2016 and is expected to reach $45.1 billion by 2022. (5)
The US represents the largest market where each day 10,000 people turn 65 years old. The impact of The Affordable Care Act (“Obamacare”) is also providing access to additional services like imaging for more of the population.
The Asia-Pacific region is the fastest growing area. This region is driven by the massive population base and the many environmental health issues created by rapid industrialization.
China is evolving into a massive market for healthcare products in dire need of effective diagnostics and treatments. This one market alone is a potential gold mine for IME to expand the application of its patented technology.
..."
Antwort auf Beitrag Nr.: 55.961.791 von praesens am 16.10.17 23:17:44
was steht hier nun, was uns nicht bereits bekannt ist?
Ich sehe den Artikel in einer Linie mit den Veröffentlichungen, die bei WOL ständig auf den Bändern laufen... oder Goldseiten, Mining scout, Rockstone etc...
oder Goldseiten, Mining scout, Rockstone etc...
Wenn IME beginnt Geld zu verdienen ( und dies möglichst reichlich) gewinnen unsere Posis doch mit, egal ob mit oder ohne diese völlig einschränkungsfreien Jubelarien über "völlig unbekannte" (???) Knaller.
Gruß Greenfoxi
Bezahlte Werbung?
Hi praesens,was steht hier nun, was uns nicht bereits bekannt ist?

Ich sehe den Artikel in einer Linie mit den Veröffentlichungen, die bei WOL ständig auf den Bändern laufen...
 oder Goldseiten, Mining scout, Rockstone etc...
oder Goldseiten, Mining scout, Rockstone etc...Wenn IME beginnt Geld zu verdienen ( und dies möglichst reichlich) gewinnen unsere Posis doch mit, egal ob mit oder ohne diese völlig einschränkungsfreien Jubelarien über "völlig unbekannte" (???) Knaller.
Gruß Greenfoxi
Antwort auf Beitrag Nr.: 55.962.846 von Greenfoxi am 17.10.17 08:47:19Du hast Recht, dass ist bezahlte Werbung und wer sich mit IME beschäftigt hat für den steht da nichts Neues drin.
Aber da das mit Sicherheit nicht jeder hier Mitlesende getan hat habe ich den Artikel nachdem ich ueber ihn gestolpert bin eingestellt.
Er ist eine gute "Abschrift" der Präsentation auf der IME website.
Ich halte ihn für "relativ" sachlich in Bezug auf IME's Technik.
Aber natürlich - solche Artikel sind immer unter Vorbehalt zu sehen.
Die weiteren erwähnten Werte habe ich extra nicht mit eingestellt. Liest man den Artikel im Original nach anklicken des links findet man sie natürlich am Ende.
Kann man dann ja ignorieren ...
Gruß, praesens
Aber da das mit Sicherheit nicht jeder hier Mitlesende getan hat habe ich den Artikel nachdem ich ueber ihn gestolpert bin eingestellt.
Er ist eine gute "Abschrift" der Präsentation auf der IME website.
Ich halte ihn für "relativ" sachlich in Bezug auf IME's Technik.
Aber natürlich - solche Artikel sind immer unter Vorbehalt zu sehen.
Die weiteren erwähnten Werte habe ich extra nicht mit eingestellt. Liest man den Artikel im Original nach anklicken des links findet man sie natürlich am Ende.
Kann man dann ja ignorieren ...

Gruß, praesens
Antwort auf Beitrag Nr.: 55.963.206 von praesens am 17.10.17 09:21:52... und sei nicht immer so streng mit mir ! 

Gruß, praesens


Gruß, praesens
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2518…
"Imagin releases i/Blue imaging trial patient criteria
2017-10-19 11:33 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES CLINICAL RESEARCH CRITERIA 10.19.17
Imagin Medical Inc. has released the recruitment criteria for the upcoming research study at the University of Rochester Medical Center.
The pilot study, entitled Near Infrared Fluorescence Imaging for Bladder Cancer Detection, is sponsored by Dr. Edward Messing, chair of urology at the University of Rochester Medical Center, and the study's principal investigator. The study is registered on the National Institutes of Health (NIH) website.
Criteria to participate in the study include men and women 18 years or older, of either sex and any racial or ethnic origin, who are scheduled for a transurethral resection of bladder tumor (TURBT) in the operating room resulting from either a new diagnosis of a potentially malignant bladder mass or a recurrent tumor.
The procedure to remove the suspicious mass will remain the same, that is, TURBT; it is the method to detect the cancerous cells to be removed that will differ significantly. The i/Blue imaging system is expected to highlight the cancerous cells in less than 10 minutes versus the full hour required by current systems, leading to a substantial reduction in the time and cost of staff and facilities.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures, where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States and the most costly cancer to treat due to a greater than 50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
"Imagin releases i/Blue imaging trial patient criteria
2017-10-19 11:33 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES CLINICAL RESEARCH CRITERIA 10.19.17
Imagin Medical Inc. has released the recruitment criteria for the upcoming research study at the University of Rochester Medical Center.
The pilot study, entitled Near Infrared Fluorescence Imaging for Bladder Cancer Detection, is sponsored by Dr. Edward Messing, chair of urology at the University of Rochester Medical Center, and the study's principal investigator. The study is registered on the National Institutes of Health (NIH) website.
Criteria to participate in the study include men and women 18 years or older, of either sex and any racial or ethnic origin, who are scheduled for a transurethral resection of bladder tumor (TURBT) in the operating room resulting from either a new diagnosis of a potentially malignant bladder mass or a recurrent tumor.
The procedure to remove the suspicious mass will remain the same, that is, TURBT; it is the method to detect the cancerous cells to be removed that will differ significantly. The i/Blue imaging system is expected to highlight the cancerous cells in less than 10 minutes versus the full hour required by current systems, leading to a substantial reduction in the time and cost of staff and facilities.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures, where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States and the most costly cancer to treat due to a greater than 50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
Es tut sich was ...  :
:
 :
:
Antwort auf Beitrag Nr.: 56.025.576 von praesens am 25.10.17 18:06:43
das sieht echt gut aus:
FAT und IME im Gleichschritt auf dem Vormarsch...


Wo bleiben die NEWS ?

Gruß Greenfoxi
Doppelglück?
Hi praesens,das sieht echt gut aus:
FAT und IME im Gleichschritt auf dem Vormarsch...



Wo bleiben die NEWS ?


Gruß Greenfoxi
Antwort auf Beitrag Nr.: 56.025.912 von Greenfoxi am 25.10.17 18:40:35News (noch) nix, dafür "power hour".
Gruß, praesens
Gruß, praesens
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2520…
"Imagin Medical closes $120,000 financing
2017-10-26 09:10 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING FINANCING TO BROADEN COMMUNICATIONS PROGRAM
Imagin Medical Inc. is closing a $120,000 financing to raise funds to be used for the company's communications program. This financing will not be oversubscribed. The company is committed to continuing the development of the i/Blue Imaging System for the detection of bladder cancer and keeping its shareholders informed of its progress.
The funds raised pursuant to the financing will be specifically targeted for a communications and marketing program, allowing the company to continue to maintain its existing cash for product development and commercialization. The financing will consist of 800,000 units at a price of 15 cents per unit, each unit to be comprised of one common share and one half of one common share purchase warrant. Each whole warrant will be exercisable into one common share in the equity of the company at an exercise price of 25 cents per warrant share. The warrants expire one year from date of issuance.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth-most prevalent in the United States, and the most costly cancer to treat due to a greater than 50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved"
"Imagin Medical closes $120,000 financing
2017-10-26 09:10 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL CLOSING FINANCING TO BROADEN COMMUNICATIONS PROGRAM
Imagin Medical Inc. is closing a $120,000 financing to raise funds to be used for the company's communications program. This financing will not be oversubscribed. The company is committed to continuing the development of the i/Blue Imaging System for the detection of bladder cancer and keeping its shareholders informed of its progress.
The funds raised pursuant to the financing will be specifically targeted for a communications and marketing program, allowing the company to continue to maintain its existing cash for product development and commercialization. The financing will consist of 800,000 units at a price of 15 cents per unit, each unit to be comprised of one common share and one half of one common share purchase warrant. Each whole warrant will be exercisable into one common share in the equity of the company at an exercise price of 25 cents per warrant share. The warrants expire one year from date of issuance.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth-most prevalent in the United States, and the most costly cancer to treat due to a greater than 50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved"
Antwort auf Beitrag Nr.: 56.027.568 von praesens am 25.10.17 21:45:06Eine identische news gab es letzte Woche bei FAT ... siehe deren Kursentwicklung ...
Gruß, praesens
Gruß, praesens
Und noch eine news heute mit Fingerzeig !:
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2521…
"Imagin grants options to buy 2.25 million shares
2017-10-26 11:54 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES STOCK OPTIONS GRANTED
Pursuant to Imagin Medical Inc.'s stock option plan, an aggregate of 2.25 million shares have been granted to certain directors, officers and consultants as incentive stock options at an exercise price of 18 cents per share. The options are exercisable for a period of five years, ending on Oct. 26, 2022.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2521…
"Imagin grants options to buy 2.25 million shares
2017-10-26 11:54 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL ANNOUNCES STOCK OPTIONS GRANTED
Pursuant to Imagin Medical Inc.'s stock option plan, an aggregate of 2.25 million shares have been granted to certain directors, officers and consultants as incentive stock options at an exercise price of 18 cents per share. The options are exercisable for a period of five years, ending on Oct. 26, 2022.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for minimally invasive surgical procedures where endoscopes are used. The company believes it will radically improve the way physicians detect cancer and view the surgical field. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States, and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. The ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
We seek Safe Harbor.
© 2017 Canjex Publishing Ltd. All rights reserved."
Doppel news heute:
Geld für promo
&
options zu .18 ... heißt zum aktuellen Kurs !
Gruß, praesens
Geld für promo
&
options zu .18 ... heißt zum aktuellen Kurs !
Gruß, praesens
Antwort auf Beitrag Nr.: 56.033.322 von praesens am 26.10.17 15:27:03Inflationär die news  :
:
http://thecse.com/sites/default/files/News_Release_SIU_Roger…
"IMAGIN MEDICAL SCIENTIFIC ADVISOR ATTENDS SOCIETY OF INTERNATIONAL
UROLOGY MEETING
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will
establish a new standard of care for urologists in detecting cancers and visualizing the
surgical field in minimally invasive surgery. The Company’s initial focus is bladder cancer.
______________________________________________________________________
Vancouver, B.C. and Boston, MA, October 27, 2017 –
Imagin Medical, Inc. (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) announced today that Roger J. Buckley, M.D., Chief of Urology at North York General Hospital in Toronto, Vice President of the International Bladder Cancer Group (IBCG), and a member of Imagin’s Scientific Advisory Board, attended the 37th Congress of the Society of International Urology held October 19 through 22 in Lisbon, Portugal. More
than 3,000 urologists were in attendance.
The SIU facilitates the improvement of urological care worldwide through its collaborative
research, education, and philanthropic activities; and this year’s innovative and impactful
scientific program featured lectures and hands-on training courses from some of the most
respected experts in the field of urology.
During the Congress there were interesting debates and presentations on the white-
light vs. blue-light cystoscopy, which reflected differing opinions on time, cost and patient
outcomes.
Blue light cystoscopy improves the detection rate of urothelial bladder cancer and allows physicians to more accurately provide appropriate management of these patients.
At this Congress, the Joint Consultation on Bladder Cancer of the SIU-ICUD
(Society of International Urology- International Consultation on Urologic Diseases)
recommended inclusion of Enhanced Cystoscopy as a beneficial adjunct to the detection
and management of non-muscle invasive bladder cancer.
Imagin considers all recommendations for Enhanced Cystoscopy as beneficial for bladder
-cancer patients and Imagin medical.
More information on this Congress is available at
https://www.siu-urology.org/congress-2017/general-info.
Dr. Buckley stated, “Enhanced cystoscopy appears to be a significant improvement in
bladder-cancer detection, and all indications are that Imagin’s i/Blue Imaging System will
be a significant product in this market.”
Dr. Buckley joined Imagin’s Scientific Advisory Board in 2016, and his involvement and
relationships in the international urology community are of significant value to Imagin
as the development of the i/Blue Imaging System continues.
..."
 :
:http://thecse.com/sites/default/files/News_Release_SIU_Roger…
"IMAGIN MEDICAL SCIENTIFIC ADVISOR ATTENDS SOCIETY OF INTERNATIONAL
UROLOGY MEETING
Imagin Medical is the developer of the ultrasensitive i/Blue Imaging System that will
establish a new standard of care for urologists in detecting cancers and visualizing the
surgical field in minimally invasive surgery. The Company’s initial focus is bladder cancer.
______________________________________________________________________
Vancouver, B.C. and Boston, MA, October 27, 2017 –
Imagin Medical, Inc. (CSE: IME) (OTC PINK: IMEXF) (Frankfurt/Stuttgart Stock Exchanges: DPD2) (the “Company”) announced today that Roger J. Buckley, M.D., Chief of Urology at North York General Hospital in Toronto, Vice President of the International Bladder Cancer Group (IBCG), and a member of Imagin’s Scientific Advisory Board, attended the 37th Congress of the Society of International Urology held October 19 through 22 in Lisbon, Portugal. More
than 3,000 urologists were in attendance.
The SIU facilitates the improvement of urological care worldwide through its collaborative
research, education, and philanthropic activities; and this year’s innovative and impactful
scientific program featured lectures and hands-on training courses from some of the most
respected experts in the field of urology.
During the Congress there were interesting debates and presentations on the white-
light vs. blue-light cystoscopy, which reflected differing opinions on time, cost and patient
outcomes.
Blue light cystoscopy improves the detection rate of urothelial bladder cancer and allows physicians to more accurately provide appropriate management of these patients.
At this Congress, the Joint Consultation on Bladder Cancer of the SIU-ICUD
(Society of International Urology- International Consultation on Urologic Diseases)
recommended inclusion of Enhanced Cystoscopy as a beneficial adjunct to the detection
and management of non-muscle invasive bladder cancer.
Imagin considers all recommendations for Enhanced Cystoscopy as beneficial for bladder
-cancer patients and Imagin medical.
More information on this Congress is available at
https://www.siu-urology.org/congress-2017/general-info.
Dr. Buckley stated, “Enhanced cystoscopy appears to be a significant improvement in
bladder-cancer detection, and all indications are that Imagin’s i/Blue Imaging System will
be a significant product in this market.”
Dr. Buckley joined Imagin’s Scientific Advisory Board in 2016, and his involvement and
relationships in the international urology community are of significant value to Imagin
as the development of the i/Blue Imaging System continues.
..."
... geruechteweise ... gibt es einen interessanten Neuzugang in Management oder BOD.
Gruß, praesens
Gruß, praesens
Antwort auf Beitrag Nr.: 56.045.892 von praesens am 27.10.17 21:01:42Zum 2ten/3ten mal für heute Test der .20 ...
Schön langsam klettert IME jeden Tag ein bisschen höher ...
Gruß, praesens
Gruß, praesens
Antwort auf Beitrag Nr.: 56.085.899 von praesens am 02.11.17 19:53:01Wobei das heute mit 12% plus schon fast ein Sprint war ... 
Gruß, praesens

Gruß, praesens
Antwort auf Beitrag Nr.: 56.086.589 von praesens am 02.11.17 21:03:17Na das nenne ich mal tree shaking. innerhalb von 25 Minuten Kurs runter auf .215 und wieder rauf auf .23 mit über 1,8 mio Stücken.
Gruß, praesens
Gruß, praesens
Antwort auf Beitrag Nr.: 56.092.889 von praesens am 03.11.17 15:11:43Neueste Präsentation:
http://imaginmedical.com/wp-content/uploads/2017/10/CORPORAT…
Und der Kurs heute von .17 auf .195
Gruß, praesens
http://imaginmedical.com/wp-content/uploads/2017/10/CORPORAT…
Und der Kurs heute von .17 auf .195
Gruß, praesens
Antwort auf Beitrag Nr.: 56.241.044 von praesens am 21.11.17 23:54:24Kurse um 1,90 euro wären doch mal feines 

... bewegt sich mit gutem Volumen ...
Gruß, praesens
Gruß, praesens
Antwort auf Beitrag Nr.: 56.289.872 von praesens am 27.11.17 20:01:58
Deine Kurseinschätzung ist etwas zu bescheiden...
Die Fakten: Neues ATH (0,255 CAD), neuer Schlussrekord (0,245 CAD), 19,51 % Tagesgewinn, zweitlängste weiße Kerze ever, dritthöchster Tagesumsatz nach Stückzahl und absoluter Rekordumsatz auf CAD-Basis seit bei IME Leben in der Bude ist (nach meiner Beobachtung seit letzter Septemberdekade).
Das ist einfach nur super (und alles ohne kurstreibende News, kommt da bald was??).
Gruß Greenfoxi
Good understatement
Hi praesens,Deine Kurseinschätzung ist etwas zu bescheiden...

Die Fakten: Neues ATH (0,255 CAD), neuer Schlussrekord (0,245 CAD), 19,51 % Tagesgewinn, zweitlängste weiße Kerze ever, dritthöchster Tagesumsatz nach Stückzahl und absoluter Rekordumsatz auf CAD-Basis seit bei IME Leben in der Bude ist (nach meiner Beobachtung seit letzter Septemberdekade).

Das ist einfach nur super (und alles ohne kurstreibende News, kommt da bald was??).

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 56.297.876 von Greenfoxi am 28.11.17 14:06:10... wie kommst Du nur auf die Idee ?! 
Gruß, praesens

Gruß, praesens
Antwort auf Beitrag Nr.: 56.297.876 von Greenfoxi am 28.11.17 14:06:10
Imagin Medical beginnt mit der Patientenrekrutierung für seine Forschungsstudie
Imagin Medical ist der Entwickler des ultrasensitiven Bildgebungssystems i/Blue, das Urologen einen neuen Behandlungsstandard bei der endoskopischen Erkennung von Karzinomen und der Visualisierung des OP-Felds bei minimal-invasiven Eingriffen bietet. Das Unternehmen konzentriert sich zunächst auf das Blasenkarzinom.
Vancouver, B.C. und Boston, MA, 29. November 2017 - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Börsen Frankfurt/Stuttgart: DPD2) (das Unternehmen) hat heute bekannt gegeben, dass das Aufnahmeverfahren und die Rekrutierungsphase für die erste Forschungsstudie zur Anwendung des Bildgebungssystems i/Blue am Menschen am University of Rochester Medical Center mittlerweile eingeleitet wurde.
Hauptzweck der Studie mit dem Titel Nah-Infrarot-Fluoreszenz zur Erkennung des Blasenkarzinoms ist es, die erforderliche Mindestverweildauer zu ermitteln, die erforderlich ist, um Tumorzellen unter Einsatz der Protoporphyrin IX-(PpIX)-Nah-Infrarot-Fluoreszenz (NIRF) ausreichend zu erkennen. Basierend auf den Ergebnissen vorhergehender Studien im Labormaßstab geht Imagin davon aus, dass anhand dieser Studie der Nachweis erbracht werden kann, dass Chirurgen mit dieser Methode weniger als 15 Minuten zur Lokalisierung von Tumorzellen benötigen, während bei herkömmlichen Systemen dafür eine volle Stunde erforderlich ist. In der Studie wird außerdem das patentierte Verfahren der gleichzeitigen Darstellung unterschiedlicher Bilder (Simultaneous Acquisition of Differing Images) evaluiert, bei dem weißes Licht und fluoreszierende Bilder ineinander geblendet werden und dem Chirurgen so in nur einem Schritt die exakte Lokalisierung von Tumorzellen in der Harnblase ermöglichen.
Die Teilnehmer an dieser aus zehn Probanden bestehenden Studie werden voraussichtlich in erster Linie aus der Gruppe der Patienten des Medical Center, die aktuell an einem Blasentumor leiden, rekrutiert und müssen spezifische Kriterien erfüllen. Aufgenommen werden Männer und Frauen ab 18 Jahren, unabhängig von ihrer Rasse oder ethnischen Herkunft, bei denen - entweder aufgrund der Neudiagnose einer potenziell bösartigen Tumormasse in der Blase oder aufgrund eines Tumorrezidivs - ein operativer Eingriff in Form einer transurethralen Resektion des Blasentumors (TURBT) geplant ist.
Dr. Edward Messing, der Vorsitzende der urologischen Abteilung am University of Rochester Medical Center, ist der Hauptprüfer der Studie, welche auf der Webseite der National Institutes of Health (NIH - www.clinicaltrials.gov) registriert wurde. Das erste Teilverfahren wird voraussichtlich innerhalb eines Monats abgeschlossen, alle zehn Teilverfahren innerhalb von vier Monaten. Die Studie bleibt für weitere neun Monate offen, um eine Folgeuntersuchung an den Probanden durchführen zu können.
Wir freuen uns wirklich sehr, meint Jim Hutchens, President und CEO von Imagin. Dies ist ein großartiger Schritt in Richtung einer besseren Erkennung des Blasenkarzinoms für all jene, die von dieser Erkrankung betroffen sind.
Kommt da bald was?
IRW-PRESS: Imagin Medical: Imagin Medical beginnt mit der Patientenrekrutierung für seine ForschungsstudieImagin Medical beginnt mit der Patientenrekrutierung für seine Forschungsstudie
Imagin Medical ist der Entwickler des ultrasensitiven Bildgebungssystems i/Blue, das Urologen einen neuen Behandlungsstandard bei der endoskopischen Erkennung von Karzinomen und der Visualisierung des OP-Felds bei minimal-invasiven Eingriffen bietet. Das Unternehmen konzentriert sich zunächst auf das Blasenkarzinom.
Vancouver, B.C. und Boston, MA, 29. November 2017 - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Börsen Frankfurt/Stuttgart: DPD2) (das Unternehmen) hat heute bekannt gegeben, dass das Aufnahmeverfahren und die Rekrutierungsphase für die erste Forschungsstudie zur Anwendung des Bildgebungssystems i/Blue am Menschen am University of Rochester Medical Center mittlerweile eingeleitet wurde.
Hauptzweck der Studie mit dem Titel Nah-Infrarot-Fluoreszenz zur Erkennung des Blasenkarzinoms ist es, die erforderliche Mindestverweildauer zu ermitteln, die erforderlich ist, um Tumorzellen unter Einsatz der Protoporphyrin IX-(PpIX)-Nah-Infrarot-Fluoreszenz (NIRF) ausreichend zu erkennen. Basierend auf den Ergebnissen vorhergehender Studien im Labormaßstab geht Imagin davon aus, dass anhand dieser Studie der Nachweis erbracht werden kann, dass Chirurgen mit dieser Methode weniger als 15 Minuten zur Lokalisierung von Tumorzellen benötigen, während bei herkömmlichen Systemen dafür eine volle Stunde erforderlich ist. In der Studie wird außerdem das patentierte Verfahren der gleichzeitigen Darstellung unterschiedlicher Bilder (Simultaneous Acquisition of Differing Images) evaluiert, bei dem weißes Licht und fluoreszierende Bilder ineinander geblendet werden und dem Chirurgen so in nur einem Schritt die exakte Lokalisierung von Tumorzellen in der Harnblase ermöglichen.
Die Teilnehmer an dieser aus zehn Probanden bestehenden Studie werden voraussichtlich in erster Linie aus der Gruppe der Patienten des Medical Center, die aktuell an einem Blasentumor leiden, rekrutiert und müssen spezifische Kriterien erfüllen. Aufgenommen werden Männer und Frauen ab 18 Jahren, unabhängig von ihrer Rasse oder ethnischen Herkunft, bei denen - entweder aufgrund der Neudiagnose einer potenziell bösartigen Tumormasse in der Blase oder aufgrund eines Tumorrezidivs - ein operativer Eingriff in Form einer transurethralen Resektion des Blasentumors (TURBT) geplant ist.
Dr. Edward Messing, der Vorsitzende der urologischen Abteilung am University of Rochester Medical Center, ist der Hauptprüfer der Studie, welche auf der Webseite der National Institutes of Health (NIH - www.clinicaltrials.gov) registriert wurde. Das erste Teilverfahren wird voraussichtlich innerhalb eines Monats abgeschlossen, alle zehn Teilverfahren innerhalb von vier Monaten. Die Studie bleibt für weitere neun Monate offen, um eine Folgeuntersuchung an den Probanden durchführen zu können.
Wir freuen uns wirklich sehr, meint Jim Hutchens, President und CEO von Imagin. Dies ist ein großartiger Schritt in Richtung einer besseren Erkennung des Blasenkarzinoms für all jene, die von dieser Erkrankung betroffen sind.
Gute Nachrichten
Das update vom Tage bringt Bewegung in den IME-Kurs.Neues ATH und Rekordschlusskurs von 0,295 CAD, 37,2 % Kursgewinn in 2 Tagen...

Das kann ein schlafender Riese sein, zumal wenn die medizinische Lösung auch auf andere Krebstherapien übertragbar sein sollte. Aber auch der Erstmarkt, für den bereits Prototypen erstellt sind, ist sehr lukrativ.

Imagin's initial target market is bladder cancer, the sixth most common cancer in the U.S. and the third most common cancer in men. Worldwide, there are over 380,000 new cases diagnosed each year. With treatment costs of $4B per year in the U.S. alone, bladder cancer is the most expensive cancer to treat during the lifetime of a patient, with approximately 60% of these costs attributable to recurrence. New cases, as well as over 500,000 patients in the U.S. living in fear of their bladder cancer returning, represent a large market that can benefit from the Company's technology.

(aus dem update von heute)
Gruß Greenfoxi
Geht ...: 
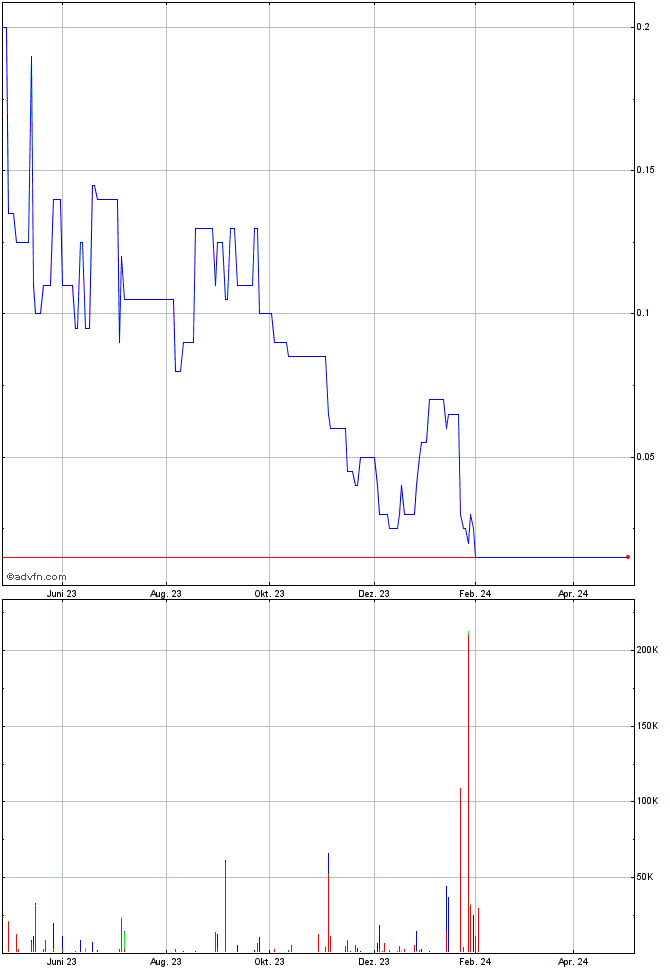

Antwort auf Beitrag Nr.: 56.689.304 von praesens am 11.01.18 18:36:06
stolze 33% Plus in einer Sitzung, alle Wetter.
Natürlich neues ATH und das bei enormen Umsätzen (Stückzahl + Volumen).
Man fasst offensichtlich immer mehr Vertrauen in die neue Analysemethodik von IME.
Gibt es hier auch einen Lagerwettbewerb ähnlich FAT?
Gruß Greenfoxi
Netter Tagesgewinn
Hi praesens,stolze 33% Plus in einer Sitzung, alle Wetter.

Natürlich neues ATH und das bei enormen Umsätzen (Stückzahl + Volumen).

Man fasst offensichtlich immer mehr Vertrauen in die neue Analysemethodik von IME.
Gibt es hier auch einen Lagerwettbewerb ähnlich FAT?

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 56.692.175 von Greenfoxi am 11.01.18 23:00:39Hi Greenfoxi,
ist mir zumindest nicht bekannt.
Gruß, praesens
ist mir zumindest nicht bekannt.
Gruß, praesens
Net schlecht / Wasserstandsmeldung
IME marschiert weiter Richtung Norden. 
Neues ATH und Rekordschlusskurs bei stattlichen Umsätzen gestern Abend in Kanada...


Gruß Greenfoxi
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2559…
"Imagin Medical provides more i/Blue study data
2018-01-22 09:05 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL RESEARCH STUDY BEGINS
Imagin Medical Inc., as a follow-up to the announcement dated Nov. 29, has released additional information concerning the research study using the i/Blue imaging system being conducted at the University of Rochester Medical Center.
As of today, initial subjects have been recruited and procedures are being coordinated with the lead investigator's surgical schedule. During the 10-subject research study, Imagin's development team will work with the surgical staff to evaluate outcomes and adjust system components as needed to optimize product performance. "We are excited to see the study underway and are fortunate to be working with the University of Rochester, one of the nation's leading academic medical centers, on this important step forward for Imagin," said Jim Hutchens, Imagin's President and CEO.
Demonstrating feasibility of the technology and defining the parameters for success will be followed by thorough assessment and refinement of all components for technical and cost efficiency. Progress will be reported as information becomes available. Imagin anticipates all ten procedures will be completed within four months, with the study remaining open for an additional nine months for subject follow-up. All updates will be published by the University of Rochester on www.clinicaltrials.gov according to the Research Study Guidelines.
The purpose of the study is to validate previous bench-testing results that showed the i/Blue Imaging System technology will enable physicians to "see" the cancer in less than 15 minutes vs. the full hour required by today's technology, as well as blend the white and florescence images into one, placing the cancer in context within the bladder. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
..."
"Imagin Medical provides more i/Blue study data
2018-01-22 09:05 ET - News Release
Mr. Jim Hutchens reports
IMAGIN MEDICAL RESEARCH STUDY BEGINS
Imagin Medical Inc., as a follow-up to the announcement dated Nov. 29, has released additional information concerning the research study using the i/Blue imaging system being conducted at the University of Rochester Medical Center.
As of today, initial subjects have been recruited and procedures are being coordinated with the lead investigator's surgical schedule. During the 10-subject research study, Imagin's development team will work with the surgical staff to evaluate outcomes and adjust system components as needed to optimize product performance. "We are excited to see the study underway and are fortunate to be working with the University of Rochester, one of the nation's leading academic medical centers, on this important step forward for Imagin," said Jim Hutchens, Imagin's President and CEO.
Demonstrating feasibility of the technology and defining the parameters for success will be followed by thorough assessment and refinement of all components for technical and cost efficiency. Progress will be reported as information becomes available. Imagin anticipates all ten procedures will be completed within four months, with the study remaining open for an additional nine months for subject follow-up. All updates will be published by the University of Rochester on www.clinicaltrials.gov according to the Research Study Guidelines.
The purpose of the study is to validate previous bench-testing results that showed the i/Blue Imaging System technology will enable physicians to "see" the cancer in less than 15 minutes vs. the full hour required by today's technology, as well as blend the white and florescence images into one, placing the cancer in context within the bladder. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
..."
Antwort auf Beitrag Nr.: 56.750.859 von Greenfoxi am 18.01.18 08:13:36Hallo Greenfoxi,
bitte schauen Sie doch mal in Ihren Briefkasten bei Wallstreet rein...
bitte schauen Sie doch mal in Ihren Briefkasten bei Wallstreet rein...
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2563…
Imagin Medical appoints Vacha as director
2018-01-30 13:23 ET - News Release
Mr. John Vacha reports
JOHN VACHA JOINS IMAGIN MEDICAL'S BOARD OF DIRECTORS
Imagin Medical Inc.'s John Vacha, the recently appointed chief financial officer, has joined Imagin's board of directors, replacing William Galine, who recently resigned for health reasons.
With over 20 years experience in the healthcare industry, Mr. Vacha is well versed in corporate governance and the medical device field. Prior to his appointment as Imagin's chief financial officer, Mr. Vacha served as president, chief executive officer and board member of Intact Medical Corp. for seven years before it was acquired by Medtronic in 2017, and previously served as the company's vice-president, CFO and treasurer.
"John brings significant business management and health care perspective to the board," said Jim Hutchens, Imagin's president and chief executive officer. "His heath care expertise is an ideal complement to Imagin s board at this stage of the company s development."
Mr. Vacha also served as senior consultant at Deloitte & Touche, LLP, where his clients included closely held high-tech manufacturing start-up companies, as well as large retail and manufacturing corporations. Mr. Vacha is a licensed chartered professional accountant in Massachusetts, holds an MBA and an MS in accounting from Northeastern University in Boston, Mass., and holds two patents in electrosurgical instrumentation.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for the early detection of cancer during minimally invasive surgeries. The company believes it will radically improve the way physicians detect cancer. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
© 2018 Canjex Publishing Ltd. All rights reserved."
Imagin Medical appoints Vacha as director
2018-01-30 13:23 ET - News Release
Mr. John Vacha reports
JOHN VACHA JOINS IMAGIN MEDICAL'S BOARD OF DIRECTORS
Imagin Medical Inc.'s John Vacha, the recently appointed chief financial officer, has joined Imagin's board of directors, replacing William Galine, who recently resigned for health reasons.
With over 20 years experience in the healthcare industry, Mr. Vacha is well versed in corporate governance and the medical device field. Prior to his appointment as Imagin's chief financial officer, Mr. Vacha served as president, chief executive officer and board member of Intact Medical Corp. for seven years before it was acquired by Medtronic in 2017, and previously served as the company's vice-president, CFO and treasurer.
"John brings significant business management and health care perspective to the board," said Jim Hutchens, Imagin's president and chief executive officer. "His heath care expertise is an ideal complement to Imagin s board at this stage of the company s development."
Mr. Vacha also served as senior consultant at Deloitte & Touche, LLP, where his clients included closely held high-tech manufacturing start-up companies, as well as large retail and manufacturing corporations. Mr. Vacha is a licensed chartered professional accountant in Massachusetts, holds an MBA and an MS in accounting from Northeastern University in Boston, Mass., and holds two patents in electrosurgical instrumentation.
About Imagin Medical Inc.
Imagin Medical is developing imaging solutions for the early detection of cancer during minimally invasive surgeries. The company believes it will radically improve the way physicians detect cancer. Imagin's initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the United States and the most costly cancer to treat due to a greater-than-50-per-cent recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors.
© 2018 Canjex Publishing Ltd. All rights reserved."
Antwort auf Beitrag Nr.: 56.791.768 von Ferdiswatchlist am 22.01.18 18:20:10
Gruß GF
BM
ist passiert...
Gruß GF
https://www.stockwatch.com/News/Item.aspx?bid=Z-C:IME-256640…
Imagin Medical continues recruitment for i/Blue
2018-02-07 09:27 ET - News Release
Mr. Jim Hutchens reports
UNIVERSITY OF ROCHESTER INITIAL RESEARCH STUDY PATIENTS COMPLETED
As a follow-up to the announcement dated Jan. 22, Imagin Medical Inc. has released additional information concerning the research study being conducted at the University of Rochester Medical Center using the i/Blue imaging system.
A number of clinical procedures have been performed and technical progress continues as this investigative research program focuses on the rapid interoperative identification of bladder cancer. Recruitment of additional subjects is ongoing, and procedures continue to be coordinated with the lead investigator's surgical schedule. Transurethral resection of bladder lesions is standard of care and is routinely performed when bladder cancer is suspected. The present clinical trial is being conducted in this context. Multispectral cystoscopic images are recorded and tissue samples sent to the pathology laboratory for evaluation immediately following each surgery. As experience is gained, the Company's development team, in concert with the UR investigative team, continues to refine components and methods in order to optimize performance in subsequent procedures.
"Imagin is pleased with the study's progress to date and we're confident we will achieve the goal of visualizing bladder cancer in less than 15 minutes," said Jim Hutchens, Imagin's President and CEO. "We will report on study progress as information becomes available."
After adequate data is collected, the study's investigative team will focus on achieving publication in peer-reviewed urologic literature and, until that time, results released will be those that will not compromise publication. Results will also be published by the University of Rochester on www.clinicaltrials.gov according to the Research Study Guidelines. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
..."
Imagin Medical continues recruitment for i/Blue
2018-02-07 09:27 ET - News Release
Mr. Jim Hutchens reports
UNIVERSITY OF ROCHESTER INITIAL RESEARCH STUDY PATIENTS COMPLETED
As a follow-up to the announcement dated Jan. 22, Imagin Medical Inc. has released additional information concerning the research study being conducted at the University of Rochester Medical Center using the i/Blue imaging system.
A number of clinical procedures have been performed and technical progress continues as this investigative research program focuses on the rapid interoperative identification of bladder cancer. Recruitment of additional subjects is ongoing, and procedures continue to be coordinated with the lead investigator's surgical schedule. Transurethral resection of bladder lesions is standard of care and is routinely performed when bladder cancer is suspected. The present clinical trial is being conducted in this context. Multispectral cystoscopic images are recorded and tissue samples sent to the pathology laboratory for evaluation immediately following each surgery. As experience is gained, the Company's development team, in concert with the UR investigative team, continues to refine components and methods in order to optimize performance in subsequent procedures.
"Imagin is pleased with the study's progress to date and we're confident we will achieve the goal of visualizing bladder cancer in less than 15 minutes," said Jim Hutchens, Imagin's President and CEO. "We will report on study progress as information becomes available."
After adequate data is collected, the study's investigative team will focus on achieving publication in peer-reviewed urologic literature and, until that time, results released will be those that will not compromise publication. Results will also be published by the University of Rochester on www.clinicaltrials.gov according to the Research Study Guidelines. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
..."
https://thecse.com/en/listings/technology/imagin-medical-inc
09/02/2018
American Urological Association (AUA) Honors Imagin Medical Scientific Advisor
Vancouver, B.C. and Boston, MA, February 9, 2018 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt & Stuttgart Symbol: DPD2) (the “Company”), announced today that the lead investigator on the research study currently underway at the University of Rochester Medical Center using the Company’s i/Blue Imaging System is being recognized for his career achievements.
Edward Messing, M.D., F.A.C.S., will receive the Ramon Guiteras Award at the AUA’s annual meeting in May in San Francisco for his 35 years of accomplishment in improving care for individuals with bladder, kidney, prostate, and testicular cancer. His contributions have enhanced knowledge of the basic molecular and genetic components that contribute to the development and progression of these diseases and translated this research into new ways of detection, prevention and treatment.
The award is presented annually to an individual deemed to have made outstanding contributions to the art and science of urology. Founded in 1902, AUA is the leading national and international educational, scientific, and clinical organization for the specialty of urology with more than 20,000 members worldwide. Guiteras was one of the founders of the AUA and the award named in his honor is considered the organization’s most prestigious recognition.
Since 1995, Dr. Messing has been a professor in the University of Rochester Medical Center Department of Urology and the Wilmot Cancer Institute where he designed and conducted landmark clinical trials.
“Dr. Messing is a renowned expert in the diagnosis and treatment of urological cancers, and we’re very fortunate that he is leading the research study on Imagin’s technology,” commented Jim Hutchens, Imagin’s President and CEO. “We appreciate his expertise and counsel.”
The purpose of the study, now in progress, is to validate Imagin’s previous bench-testing results that showed the i/Blue Imaging System technology will enable physicians to “see” the cancer in less than 15 minutes vs. the full hour required by today’s technology, as well as blend the white and florescence images into one, placing the cancer in context within the bladder. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer during minimally invasive surgeries. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
09/02/2018
American Urological Association (AUA) Honors Imagin Medical Scientific Advisor
Vancouver, B.C. and Boston, MA, February 9, 2018 – Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (Frankfurt & Stuttgart Symbol: DPD2) (the “Company”), announced today that the lead investigator on the research study currently underway at the University of Rochester Medical Center using the Company’s i/Blue Imaging System is being recognized for his career achievements.
Edward Messing, M.D., F.A.C.S., will receive the Ramon Guiteras Award at the AUA’s annual meeting in May in San Francisco for his 35 years of accomplishment in improving care for individuals with bladder, kidney, prostate, and testicular cancer. His contributions have enhanced knowledge of the basic molecular and genetic components that contribute to the development and progression of these diseases and translated this research into new ways of detection, prevention and treatment.
The award is presented annually to an individual deemed to have made outstanding contributions to the art and science of urology. Founded in 1902, AUA is the leading national and international educational, scientific, and clinical organization for the specialty of urology with more than 20,000 members worldwide. Guiteras was one of the founders of the AUA and the award named in his honor is considered the organization’s most prestigious recognition.
Since 1995, Dr. Messing has been a professor in the University of Rochester Medical Center Department of Urology and the Wilmot Cancer Institute where he designed and conducted landmark clinical trials.
“Dr. Messing is a renowned expert in the diagnosis and treatment of urological cancers, and we’re very fortunate that he is leading the research study on Imagin’s technology,” commented Jim Hutchens, Imagin’s President and CEO. “We appreciate his expertise and counsel.”
The purpose of the study, now in progress, is to validate Imagin’s previous bench-testing results that showed the i/Blue Imaging System technology will enable physicians to “see” the cancer in less than 15 minutes vs. the full hour required by today’s technology, as well as blend the white and florescence images into one, placing the cancer in context within the bladder. For further information on the study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer during minimally invasive surgeries. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
https://thecse.com/en/listings/technology/imagin-medical-inc
IMAGIN MEDICAL CLOSES PRIVATE PLACEMENT
Vancouver, B.C. and Boston, MA, February 15, 2018 – Imagin Medical Inc. (CSE: IME) (OTC PINK: IMEXF) (Frankfurt & Stuttgart Symbol: DPD2) (the “Company”) announced today the closing of a non-brokered private placement that raised $200,000 through the issuance of 625,000 shares at a price of $0.32 per share.
The funds raised pursuant to the financing will be specifically allocated for the Company’s ongoing communications and marketing program, allowing the Company to continue to maintain its existing cash for product development and commercialization.
The Company closed the private placement after submitting the required filings with the regulatory authorities.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer where endoscopes are used. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
For further information, contact:
Jim Hutchens, President & CEO
Telephone: 617-571-6006
Email: jhutchens@imaginmedical.com
Information set forth in this news release contains forward-looking statements. These statements reflect management’s current estimates, beliefs, intentions and expectations; they are not guarantees of future performance. The Company cautions that all forward-looking statements are inherently uncertain and that actual performance may be affected by a number of material factors, many of which are beyond the Company’s control. Accordingly, actual and future events, conditions and results may differ materially from the estimates, beliefs, intentions and expectations expressed or implied in the forward-looking information. Specifically, there is no assurance the Company’s imaging system will work in the manner expected, except as required under applicable securities legislation; the Company undertakes no obligation to publicly update or revise forward-looking information. The CSE has neither approved nor disapproved the information contained herein and does not accept responsibility for the adequacy or accuracy of this news release.
IMAGIN MEDICAL CLOSES PRIVATE PLACEMENT
Vancouver, B.C. and Boston, MA, February 15, 2018 – Imagin Medical Inc. (CSE: IME) (OTC PINK: IMEXF) (Frankfurt & Stuttgart Symbol: DPD2) (the “Company”) announced today the closing of a non-brokered private placement that raised $200,000 through the issuance of 625,000 shares at a price of $0.32 per share.
The funds raised pursuant to the financing will be specifically allocated for the Company’s ongoing communications and marketing program, allowing the Company to continue to maintain its existing cash for product development and commercialization.
The Company closed the private placement after submitting the required filings with the regulatory authorities.
About Imagin Medical
Imagin Medical is developing imaging solutions for the early detection of cancer where endoscopes are used. The Company believes it will radically improve the way physicians detect cancer. Imagin’s initial target market is bladder cancer, a major cancer worldwide, the sixth most prevalent in the U.S., and the most costly cancer to treat due to a greater than 50% recurrence rate. Developed at the Lawrence Livermore National Laboratory, this advanced, ultrasensitive imaging technology is based upon improved optical designs and advanced light sensors. Learn more at www.imaginmedical.com.
For further information, contact:
Jim Hutchens, President & CEO
Telephone: 617-571-6006
Email: jhutchens@imaginmedical.com
Information set forth in this news release contains forward-looking statements. These statements reflect management’s current estimates, beliefs, intentions and expectations; they are not guarantees of future performance. The Company cautions that all forward-looking statements are inherently uncertain and that actual performance may be affected by a number of material factors, many of which are beyond the Company’s control. Accordingly, actual and future events, conditions and results may differ materially from the estimates, beliefs, intentions and expectations expressed or implied in the forward-looking information. Specifically, there is no assurance the Company’s imaging system will work in the manner expected, except as required under applicable securities legislation; the Company undertakes no obligation to publicly update or revise forward-looking information. The CSE has neither approved nor disapproved the information contained herein and does not accept responsibility for the adequacy or accuracy of this news release.
http://www.stockhouse.com/news/press-releases/2018/02/26/ima…
"Imagin Medical Hires Director of Regulatory Affairs
C.IME | 1 hour ago
Imagin Medical Hires Director of Regulatory Affairs
VANCOUVER, BC and BOSTON, MA --(Marketwired - February 26, 2018) - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (FRANKFURT: DPD2) (STUT: DPD2) (the "Company"), announced today the Company has hired Pam Papineau as the Director of Regulatory Affairs.
Ms. Papineau has over 30 years' experience in quality and regulatory affairs with Baxter, Boston Scientific and Cogentix (Vision-Sciences), and has served as a consultant on a wide variety of devices that includes endoscopy, imaging, GI/GU, orthopedic and cardiovascular. Pam has successfully prepared dozens of FDA premarket submissions and European Union (EU) technical files to support CE marking of a broad spectrum of medical devices and has overseen numerous clinical studies entailing protocol development, reporting and management of Clinical Research Organization (CRO) services. Pam is an ASQ Certified Quality Engineer (CQE), Certified Quality Auditor (CQA) and Certified Biomedical Auditor (CBA), an ISO 13485:2016 Lead Auditor, and holds Regulatory Affairs Professional Society (RAPS) certifications for the US, EU, CAN. Pam will work with Hogan Lovells, the Company's legal Counsel, preparing for a pre-submission meeting with the FDA and follow-on activities through the regulatory approval process.
"As the project progresses, we continue to put the needed talent in place to bring the i/Blue Imaging System to market," said Jim Hutchens, Imagin's President and CEO. "We have worked with Pam in the past and we're excited to have her expertise on our team."
For further information on the current i/Blue Imaging System research study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection on www.clinicaltrials.gov.
..."
"Imagin Medical Hires Director of Regulatory Affairs
C.IME | 1 hour ago
Imagin Medical Hires Director of Regulatory Affairs
VANCOUVER, BC and BOSTON, MA --(Marketwired - February 26, 2018) - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (FRANKFURT: DPD2) (STUT: DPD2) (the "Company"), announced today the Company has hired Pam Papineau as the Director of Regulatory Affairs.
Ms. Papineau has over 30 years' experience in quality and regulatory affairs with Baxter, Boston Scientific and Cogentix (Vision-Sciences), and has served as a consultant on a wide variety of devices that includes endoscopy, imaging, GI/GU, orthopedic and cardiovascular. Pam has successfully prepared dozens of FDA premarket submissions and European Union (EU) technical files to support CE marking of a broad spectrum of medical devices and has overseen numerous clinical studies entailing protocol development, reporting and management of Clinical Research Organization (CRO) services. Pam is an ASQ Certified Quality Engineer (CQE), Certified Quality Auditor (CQA) and Certified Biomedical Auditor (CBA), an ISO 13485:2016 Lead Auditor, and holds Regulatory Affairs Professional Society (RAPS) certifications for the US, EU, CAN. Pam will work with Hogan Lovells, the Company's legal Counsel, preparing for a pre-submission meeting with the FDA and follow-on activities through the regulatory approval process.
"As the project progresses, we continue to put the needed talent in place to bring the i/Blue Imaging System to market," said Jim Hutchens, Imagin's President and CEO. "We have worked with Pam in the past and we're excited to have her expertise on our team."
For further information on the current i/Blue Imaging System research study, visit, Near Infrared Fluorescence Imaging for Bladder Cancer Detection on www.clinicaltrials.gov.
..."
Antwort auf Beitrag Nr.: 57.121.740 von praesens am 26.02.18 15:21:53http://www.stockhouse.com/news/press-releases/2018/03/07/ima…
"Imagin Medical Product Re-Design on Schedule
C.IME | 40 minutes ago
Imagin Medical Product Re-Design on Schedule
VANCOUVER, BC and BOSTON, MA --(Marketwired - March 07, 2018) - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (FRANKFURT: DPD2) (STUT: DPD2) (the "Company"), announced today that the Company expects the Proof of Concept phase of the prototype re-design for manufacturing to be completed on schedule for this coming May.
As reported, Optel, Inc., Imagin's opto-electronic design firm, is working against three phases of development: Proof of Concept, Functional Unit and Verified Unit. The Proof of Concept effort is intended to benchmark the performance metrics of the i/Blue System's two key components -- the high-power LED based illumination module and the multi-channel imaging module -- against the performance of the i/Blue prototype currently being used in a research study at the University of Rochester, and the performance of various FDA approved light-based products.
Jay Eastman, Optel's CEO stated, "The prototype high current LED controller for the i/Blue illumination module has been tested and performs well above expectations. We will build the pre-production version of the prototype board and have it ready for the Proof of Concept testing in May. The prototype optical mechanical is nearing design completion and should be out for fabrication later this month."
The next step in the proof of concept effort will address the design and prototyping of the i/Blue imaging module which provides separate imaging channels for white light viewing of the interior of the bladder through a traditional cystoscope and near infrared imaging of the Protoporphyrin IX fluorescence emitted by a tumor in the bladder. The imaging module will utilize state-of-the-art optics, filters and cameras to simultaneously image and display both the white light and near infrared imaging channels. Optel has substantial experience in the design of similar miniaturized optical systems and has high confidence the imaging module will also be ready to meet the May Proof of Concept milestone.
"We're excited to reach this first phase in the redesign which keeps us on track as the research study at the university of Rochester Medical Center progresses," said Jim Hutchens, Imagin's President and CEO.
The University of Rochester Medical Center's research study using the Company's i/Blue Imaging System continues to recruit patients, refine the prototype per procedure, work to validate bench-testing results and prove human feasibility.
..."
"Imagin Medical Product Re-Design on Schedule
C.IME | 40 minutes ago
Imagin Medical Product Re-Design on Schedule
VANCOUVER, BC and BOSTON, MA --(Marketwired - March 07, 2018) - Imagin Medical (CSE: IME) (OTC PINK: IMEXF) (FRANKFURT: DPD2) (STUT: DPD2) (the "Company"), announced today that the Company expects the Proof of Concept phase of the prototype re-design for manufacturing to be completed on schedule for this coming May.
As reported, Optel, Inc., Imagin's opto-electronic design firm, is working against three phases of development: Proof of Concept, Functional Unit and Verified Unit. The Proof of Concept effort is intended to benchmark the performance metrics of the i/Blue System's two key components -- the high-power LED based illumination module and the multi-channel imaging module -- against the performance of the i/Blue prototype currently being used in a research study at the University of Rochester, and the performance of various FDA approved light-based products.
Jay Eastman, Optel's CEO stated, "The prototype high current LED controller for the i/Blue illumination module has been tested and performs well above expectations. We will build the pre-production version of the prototype board and have it ready for the Proof of Concept testing in May. The prototype optical mechanical is nearing design completion and should be out for fabrication later this month."
The next step in the proof of concept effort will address the design and prototyping of the i/Blue imaging module which provides separate imaging channels for white light viewing of the interior of the bladder through a traditional cystoscope and near infrared imaging of the Protoporphyrin IX fluorescence emitted by a tumor in the bladder. The imaging module will utilize state-of-the-art optics, filters and cameras to simultaneously image and display both the white light and near infrared imaging channels. Optel has substantial experience in the design of similar miniaturized optical systems and has high confidence the imaging module will also be ready to meet the May Proof of Concept milestone.
"We're excited to reach this first phase in the redesign which keeps us on track as the research study at the university of Rochester Medical Center progresses," said Jim Hutchens, Imagin's President and CEO.
The University of Rochester Medical Center's research study using the Company's i/Blue Imaging System continues to recruit patients, refine the prototype per procedure, work to validate bench-testing results and prove human feasibility.
..."
Kapitalerhöhung
Die Art der zuletzt annoncierten Privatplatzierung kann meine Zustimmung nicht finden. 
Nicht nur, dass allen aktiven Shareholders nicht ein gleichlautendes Angebot unterbreitet wurde, um an der KE teilzuhaben, sondern den neuen Besitzern werden die Teile zu einem Kurs von 0,22 CAD verscherbelt (als der Börsenkurs bei 0,31 stand).


Entsprechend ist der Kurs auch abgeschmiert (von 0,315 auf 0,26 --> 18%), hat sich jetzt wieder leicht erholt.

Womit wird dieser enorme Bonus gerechtfertigt, kommt man zu besseren Konditionen nicht an Gelder?

Wieso werden die bestehenden Aktionäre nicht gleichbehandelt?

Habe da von anderen Firmen schon wesentlich faireren Umgang erleben dürfen...

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 57.564.801 von praesens am 17.04.18 16:04:07Damit wäre dieses unschöne pp dann auch aus dem Weg:
http://www.stockhouse.com/news/press-releases/2018/04/19/ima…
"Imagin Medical Closes Oversubscribed Private Placement
C.IME | 26 minutes ago
VANCOUVER, British Columbia and BOSTON, April 19, 2018 (GLOBE NEWSWIRE) -- Imagin Medical, Inc. (CSE:IME) (OTC PINK:IMEXF) (Frankfurt/Stuttgart Stock Exchanges PD2) (the “Company”) announces that, further to its press releases of April 3 and April 12, 2018, the Company is closing Tranche 2 of its non-brokered private placement, issuing 6,197,037 Units at $0.22 per Unit for proceeds of $1,363,648. This brings the total offering to 17,919,820 Units for aggregate gross proceeds of $3,942,360.
PD2) (the “Company”) announces that, further to its press releases of April 3 and April 12, 2018, the Company is closing Tranche 2 of its non-brokered private placement, issuing 6,197,037 Units at $0.22 per Unit for proceeds of $1,363,648. This brings the total offering to 17,919,820 Units for aggregate gross proceeds of $3,942,360.
The Private Placement was originally announced as a $3,500,000 offering. The placement has been oversubscribed, resulting in the issuance of an additional 2,010,729 Units for $442,360. Proceeds from the offering will be used for general working capital purposes, including advancing the development of the i/Blue Imaging System, the regulatory approval process and on-going clinical trials.
Each Unit of this Private Placement consists of one common share of the Company and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share of the Company at a price of $0.38 within 24 months from the date of issue.
Total finders’ fees will be paid in the form of cash in the amount of $210,256.27 and 1,117,110 finders’ warrants (exercisable at $0.38 per share for 24 months). All securities issued will be subject to a four-month hold period.
The Company also announced today that, pursuant to the Company’s Stock Option Plan, an aggregate of 2,750,000 options have been granted to certain directors, officers and consultants. The options are exercisable at $.31 per share for a period of five years.
“The success of this private placement strengthens Imagin’s financial position and our ability to drive the i/Blue technology to the marketplace,” said Jim Hutchens, Imagin President and CEO. “We appreciate our investors’ confidence.”
..."
http://www.stockhouse.com/news/press-releases/2018/04/19/ima…
"Imagin Medical Closes Oversubscribed Private Placement
C.IME | 26 minutes ago
VANCOUVER, British Columbia and BOSTON, April 19, 2018 (GLOBE NEWSWIRE) -- Imagin Medical, Inc. (CSE:IME) (OTC PINK:IMEXF) (Frankfurt/Stuttgart Stock Exchanges
 PD2) (the “Company”) announces that, further to its press releases of April 3 and April 12, 2018, the Company is closing Tranche 2 of its non-brokered private placement, issuing 6,197,037 Units at $0.22 per Unit for proceeds of $1,363,648. This brings the total offering to 17,919,820 Units for aggregate gross proceeds of $3,942,360.
PD2) (the “Company”) announces that, further to its press releases of April 3 and April 12, 2018, the Company is closing Tranche 2 of its non-brokered private placement, issuing 6,197,037 Units at $0.22 per Unit for proceeds of $1,363,648. This brings the total offering to 17,919,820 Units for aggregate gross proceeds of $3,942,360.The Private Placement was originally announced as a $3,500,000 offering. The placement has been oversubscribed, resulting in the issuance of an additional 2,010,729 Units for $442,360. Proceeds from the offering will be used for general working capital purposes, including advancing the development of the i/Blue Imaging System, the regulatory approval process and on-going clinical trials.
Each Unit of this Private Placement consists of one common share of the Company and one common share purchase warrant, each warrant entitling the holder to acquire one additional common share of the Company at a price of $0.38 within 24 months from the date of issue.
Total finders’ fees will be paid in the form of cash in the amount of $210,256.27 and 1,117,110 finders’ warrants (exercisable at $0.38 per share for 24 months). All securities issued will be subject to a four-month hold period.
The Company also announced today that, pursuant to the Company’s Stock Option Plan, an aggregate of 2,750,000 options have been granted to certain directors, officers and consultants. The options are exercisable at $.31 per share for a period of five years.
“The success of this private placement strengthens Imagin’s financial position and our ability to drive the i/Blue technology to the marketplace,” said Jim Hutchens, Imagin President and CEO. “We appreciate our investors’ confidence.”
..."
Imagin Medical verbessert die Diagnose und OP von Blasenkrebs
http://www.goldinvest.de/aus-der-redaktion/1627-imagin-medic…
http://www.goldinvest.de/aus-der-redaktion/1627-imagin-medic…
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2598…
"Imagin Medical evaluates five subjects with i/Blue
2018-04-26 09:59 ET - News Release
Mr. Jim Hutchens reports
IMAGIN GAINS MOMENTUM; POSITIVE PROGRESS IN RESEARCH STUDY
Imagin Medical Inc. has provided the following update on the company's current activities.
The recent closing of a Private Placement for approximately $4,000,000 reinforces Imagin's financial runway, enabling the Company to progress through the next critical steps toward product commercialization. Imagin's management team has been strengthened with the recent appointments of a new CFO and Director of Regulatory Affairs who add the essential experience and knowledge base in the medical device industry to drive the Company through several paths concurrently.
To date, five subjects have been evaluated using the i/Blue System. Given that this study is the i/Blue system's initial use in a human bladder, approximately two weeks have been allowed between procedures for the development team to assess performance and to make hardware improvements, such as lenses and filters, as well as software adjustments. More subjects will be scheduled for evaluation, and further updates will be issued. The data and knowledge Imagin is acquiring is essential in refining the images that the company believes will lead to optimal performance.
Simultaneously, the Company continues to work with Optel, Inc. on the product redesign for future clinical trials and commercialization. The miniaturization process will add significant imaging capability and lower the actual cost of the product. In addition, documentation for federal regulatory approval continues in anticipation of meetings expected to take place during the summer months.
"This is an exciting time. With proper funding and an experienced management team in place, we're confident 2018 will be a pivotal year," said Jim Hutchens, Imagin's President and CEO.
..."
"Imagin Medical evaluates five subjects with i/Blue
2018-04-26 09:59 ET - News Release
Mr. Jim Hutchens reports
IMAGIN GAINS MOMENTUM; POSITIVE PROGRESS IN RESEARCH STUDY
Imagin Medical Inc. has provided the following update on the company's current activities.
The recent closing of a Private Placement for approximately $4,000,000 reinforces Imagin's financial runway, enabling the Company to progress through the next critical steps toward product commercialization. Imagin's management team has been strengthened with the recent appointments of a new CFO and Director of Regulatory Affairs who add the essential experience and knowledge base in the medical device industry to drive the Company through several paths concurrently.
To date, five subjects have been evaluated using the i/Blue System. Given that this study is the i/Blue system's initial use in a human bladder, approximately two weeks have been allowed between procedures for the development team to assess performance and to make hardware improvements, such as lenses and filters, as well as software adjustments. More subjects will be scheduled for evaluation, and further updates will be issued. The data and knowledge Imagin is acquiring is essential in refining the images that the company believes will lead to optimal performance.
Simultaneously, the Company continues to work with Optel, Inc. on the product redesign for future clinical trials and commercialization. The miniaturization process will add significant imaging capability and lower the actual cost of the product. In addition, documentation for federal regulatory approval continues in anticipation of meetings expected to take place during the summer months.
"This is an exciting time. With proper funding and an experienced management team in place, we're confident 2018 will be a pivotal year," said Jim Hutchens, Imagin's President and CEO.
..."
Was sagt ihr zur Kursentwicklung von dieser Woche?
Antwort auf Beitrag Nr.: 57.731.809 von f1re am 10.05.18 09:19:33Ein write-up:
https://www.imaginmedicalstock.com/national-investor-analysi…
https://www.imaginmedicalstock.com/national-investor-analysi…
Good or bad???
https://www.beprofiteer.com/nlhtm/show.php?id=260Kennt jemand diese Research-Quelle bezüglich SERIOSITÄT ?

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 57.900.567 von Greenfoxi am 04.06.18 09:22:19Für mich vergleichbar mit Bullvestor.
Bereits bekannte Informationen zusammengetragen und optisch aufgewertet.
Es generiert Käufer und das ist zunächst mal gut für mein Invest von dem ich schon vorher überzeugt war.
Bereits bekannte Informationen zusammengetragen und optisch aufgewertet.
Es generiert Käufer und das ist zunächst mal gut für mein Invest von dem ich schon vorher überzeugt war.
Uii grade +21% bei Tradegate
Antwort auf Beitrag Nr.: 57.902.487 von f1re am 04.06.18 13:39:17

oder kennen nun schon alle seine spiele??
wer sein geld pollinger schenken möchte
der möge viele stücke kaufen,die nächste hole nuss muss ausgekocht werden

oder kennen nun schon alle seine spiele??
Bezüglich Pollinger gebe ich meinem Vorredner 110% Recht.
Aber ... der Clown ist hier auf einen fahrenden Zug aufgesprungen.
IME ist ein hochinteressantes Unternehmen.
Clinical studies am Laufen.
Interessante Partner.
Interessante Geldgeber.
Also ruhig etwas weiter graben als nur bis zum Pollinger (ausnahmsweise ...).
Gruß, praesens
Aber ... der Clown ist hier auf einen fahrenden Zug aufgesprungen.
IME ist ein hochinteressantes Unternehmen.
Clinical studies am Laufen.
Interessante Partner.
Interessante Geldgeber.
Also ruhig etwas weiter graben als nur bis zum Pollinger (ausnahmsweise ...).
Gruß, praesens
Antwort auf Beitrag Nr.: 57.928.512 von praesens am 07.06.18 09:03:46
https://www.richard-wolf.com/no_cache/de/produkte.html?tx_sn…
was hälst du davon:Neue Wege für die Blaulichtanwendung bei Harnblasenkrebs
Mit dem neuen System blue von Richard Wolf, bei dem erstmals eine einzigartige PDD-Lichtquelle auf LED-Basis eingesetzt wird, kann ein Harnblasenkarzinom nun noch deutlicher vom gesunden Gewebe abgegrenzt werden.
ich weiss zwar nicht was daran neu sein soll
aber ok,du musst es ja wissen!https://www.richard-wolf.com/no_cache/de/produkte.html?tx_sn…
was hälst du davon:Neue Wege für die Blaulichtanwendung bei Harnblasenkrebs
Mit dem neuen System blue von Richard Wolf, bei dem erstmals eine einzigartige PDD-Lichtquelle auf LED-Basis eingesetzt wird, kann ein Harnblasenkarzinom nun noch deutlicher vom gesunden Gewebe abgegrenzt werden.
Antwort auf Beitrag Nr.: 57.930.501 von partisan0815 am 07.06.18 12:03:21... wieso fühlt man sich auf w:o eigentlich immer so schnell auf's Fuesschen getreten bzw. fühlt sich genötigt sofort persönlich zu werden ...?:
"aber ok,du musst es ja wissen!"
Ganz einfach sachlich bleiben und das Gegenargument bringen wäre doch auch nicht schlecht ...
Zum Vergleich, zwar wohl schon x-mal hier im thread zu lesen gewesen, aber na gut ...:
https://imaginmedical.com/technology/
Gruß, praesens
"aber ok,du musst es ja wissen!"
Ganz einfach sachlich bleiben und das Gegenargument bringen wäre doch auch nicht schlecht ...
Zum Vergleich, zwar wohl schon x-mal hier im thread zu lesen gewesen, aber na gut ...:
https://imaginmedical.com/technology/
Gruß, praesens
Antwort auf Beitrag Nr.: 57.928.410 von partisan0815 am 07.06.18 08:52:36Stell mal nen Link rein. Hab nämlich nichts von Pollinger zu dieser Aktie gesehen.
Imagin Medical taking bladder cancer imaging to next level
http://www.biotuesdays.com/features/2018/6/12/imagin-medical…
http://www.biotuesdays.com/features/2018/6/12/imagin-medical…
Könnte vielleicht helfen...
IME soll mehr in den Fokus von Investoren gerückt werden. 
https://mobile.tmxmoney.com/quote/readnews/?id=4743631293348…
Wäre nötig dem zuletzt stetig bröckelnden Kurs etwas auf die Beine zu helfen.

Mein dickes Plus schmilzt allmählich...

Gruß Greenfoxi
Nachschlag zur letzten News
https://mobile.tmxmoney.com/quote/readnews/?id=5956831916438…Kilmer Lucas bekommt 7.500 $ mtl. für sein Dienste für zunächst 60 Tage...
Wenn es hilft, okay

Gruß Greenfoxi
IME ist heute ziemlich abgerutscht. Was meint ihr dazu? Erwartungen auf News nicht erfüllt? Ungeduldige Anleger?
Antwort auf Beitrag Nr.: 58.182.275 von f1re am 10.07.18 22:49:16Aufgrund sonstiger fehlender Anhaltspunkte ... m.E. Letzteres.
Ein Grund, warum ich z.B. Medtech Aktien eigentlich nicht so mag -
während wie hier der clinical trials gibt es keinen newsflow. Worüber auch ?
Das heißt der Kurs duempelt/bewegt sich antriebs- und planlos durch die Gegend ...
Gruß, praesens
Ein Grund, warum ich z.B. Medtech Aktien eigentlich nicht so mag -
während wie hier der clinical trials gibt es keinen newsflow. Worüber auch ?
Das heißt der Kurs duempelt/bewegt sich antriebs- und planlos durch die Gegend ...
Gruß, praesens
Antwort auf Beitrag Nr.: 58.186.505 von praesens am 11.07.18 13:50:09http://www.stockhouse.com/news/press-releases/2018/07/17/ima…
"Imagin Medical Reports Research Study Progress US
C.IME | 30 minutes ago
VANCOUVER, British Columbia and BOSTON, July 17, 2018 (GLOBE NEWSWIRE) -- Imagin Medical (CSE:IME) (OTCQB:IMEXF) (Frankfurt PD2) (Stuttgart
PD2) (Stuttgart PD2) (“Imagin” or the “Company”) announced today favorable results in the 10-subject investigator-sponsored research study being conducted at the University of Rochester Medical Center using the i/Blue Imaging System (the “URMC Research Study”), an important step in the development of a commercially-viable product.
PD2) (“Imagin” or the “Company”) announced today favorable results in the 10-subject investigator-sponsored research study being conducted at the University of Rochester Medical Center using the i/Blue Imaging System (the “URMC Research Study”), an important step in the development of a commercially-viable product.
In the URMC Research Study, fluorescence was successfully demonstrated in 15 minutes, confirming that the i/Blue Imaging System will potentially allow physicians to “see” bladder cancer significantly faster than the full hour or more required by currently available imaging technology.
“While a backorder on certain component parts needed to make system adjustments during the URMC Research Study caused some delay, we are obviously very pleased with the outcome,” said Jim Hutchens, Imagin’s President and CEO. “This is an exciting time. We believe that the i/Blue Imaging System holds great promise for improving physicians’ ability to detect cancer and visualize the surgical field where endoscopes are used. To that end, we look forward to updating our stakeholders as we progress.”
Information gathered from the URMC Research Study is being used to help optimize Imagin’s i/Blue technology to further the development of a commercially-viable device system in preparation for the Company’s planned clinical trials to support its application to the U.S. Food and Drug Administration (“FDA”) for approval to market the i/Blue Imaging System in the United States.
“Imagin is grateful to the University of Rochester Medical Center for initiating and conducting this investigator-sponsored human research study,” commented Pam Papineau, Imagin’s Director of Regulatory and Clinical Affairs. “Imagin was pleased to provide early prototype equipment to assist the investigators in testing the underlying hypothesis that the i/Blue Imaging System can potentially identify protoporphyrin IX (PpIX) fluorescence associated with bladder cancer lesions in the presence of the contrasting agent, hexaminolevulinate hydrochloride. This project was undertaken prior to the commencement of an FDA application, and provides important insights that will help guide the Company in an FDA-compliant process. Imagin anticipates initiating preliminary discussions with the FDA regarding the i/Blue Imaging System as soon as possible.”
Running parallel to the URMC Research Study, the i/Blue Imaging System design and development has moved forward, incorporating feedback from the study into the system’s opto-electronic functions and features. As previously reported, Optel, Inc., Imagin’s opto-electronic design firm, is working against three phases of development: Proof of Concept, Functional Unit and Verified Unit. Of the three phases, the Proof of Concept phase has now been completed, verifying the performance of the two critical optical modules of the i/Blue Imaging System – the light source module and the imaging module.
Jay Eastman, Ph.D., CEO of Optel, stated, “The lessons learned from the URMC Research Study has provided valuable technical data, clinical insight and physician feedback that is significantly influencing the design of the i/Blue System. All of this information continues to increase our confidence that the miniaturization, imaging quality and cost reduction goals for the i/Blue System important to surgeons performing bladder cancer procedures.”
..."
"Imagin Medical Reports Research Study Progress US
C.IME | 30 minutes ago
VANCOUVER, British Columbia and BOSTON, July 17, 2018 (GLOBE NEWSWIRE) -- Imagin Medical (CSE:IME) (OTCQB:IMEXF) (Frankfurt
 PD2) (Stuttgart
PD2) (Stuttgart PD2) (“Imagin” or the “Company”) announced today favorable results in the 10-subject investigator-sponsored research study being conducted at the University of Rochester Medical Center using the i/Blue Imaging System (the “URMC Research Study”), an important step in the development of a commercially-viable product.
PD2) (“Imagin” or the “Company”) announced today favorable results in the 10-subject investigator-sponsored research study being conducted at the University of Rochester Medical Center using the i/Blue Imaging System (the “URMC Research Study”), an important step in the development of a commercially-viable product. In the URMC Research Study, fluorescence was successfully demonstrated in 15 minutes, confirming that the i/Blue Imaging System will potentially allow physicians to “see” bladder cancer significantly faster than the full hour or more required by currently available imaging technology.
“While a backorder on certain component parts needed to make system adjustments during the URMC Research Study caused some delay, we are obviously very pleased with the outcome,” said Jim Hutchens, Imagin’s President and CEO. “This is an exciting time. We believe that the i/Blue Imaging System holds great promise for improving physicians’ ability to detect cancer and visualize the surgical field where endoscopes are used. To that end, we look forward to updating our stakeholders as we progress.”
Information gathered from the URMC Research Study is being used to help optimize Imagin’s i/Blue technology to further the development of a commercially-viable device system in preparation for the Company’s planned clinical trials to support its application to the U.S. Food and Drug Administration (“FDA”) for approval to market the i/Blue Imaging System in the United States.
“Imagin is grateful to the University of Rochester Medical Center for initiating and conducting this investigator-sponsored human research study,” commented Pam Papineau, Imagin’s Director of Regulatory and Clinical Affairs. “Imagin was pleased to provide early prototype equipment to assist the investigators in testing the underlying hypothesis that the i/Blue Imaging System can potentially identify protoporphyrin IX (PpIX) fluorescence associated with bladder cancer lesions in the presence of the contrasting agent, hexaminolevulinate hydrochloride. This project was undertaken prior to the commencement of an FDA application, and provides important insights that will help guide the Company in an FDA-compliant process. Imagin anticipates initiating preliminary discussions with the FDA regarding the i/Blue Imaging System as soon as possible.”
Running parallel to the URMC Research Study, the i/Blue Imaging System design and development has moved forward, incorporating feedback from the study into the system’s opto-electronic functions and features. As previously reported, Optel, Inc., Imagin’s opto-electronic design firm, is working against three phases of development: Proof of Concept, Functional Unit and Verified Unit. Of the three phases, the Proof of Concept phase has now been completed, verifying the performance of the two critical optical modules of the i/Blue Imaging System – the light source module and the imaging module.
Jay Eastman, Ph.D., CEO of Optel, stated, “The lessons learned from the URMC Research Study has provided valuable technical data, clinical insight and physician feedback that is significantly influencing the design of the i/Blue System. All of this information continues to increase our confidence that the miniaturization, imaging quality and cost reduction goals for the i/Blue System important to surgeons performing bladder cancer procedures.”
..."
News:
17/07/2018
Imagin Medical Reports research study Progress
https://webfiles.thecse.com/Press_Release_-_IME_-_Research_S…
17/07/2018
Imagin Medical Reports research study Progress
https://webfiles.thecse.com/Press_Release_-_IME_-_Research_S…
Antwort auf Beitrag Nr.: 58.238.139 von praesens am 17.07.18 23:05:33Du warst schneller ;-)
Was hälst du von der News?
Für mich ist es einerseits positiv aber andererseits scheint es auch gewisse Probleme gegeben zu haben. Auch fehlen mir konkrete Daten und Termine wie es weitergeht.
Gruß
Was hälst du von der News?
Für mich ist es einerseits positiv aber andererseits scheint es auch gewisse Probleme gegeben zu haben. Auch fehlen mir konkrete Daten und Termine wie es weitergeht.
Gruß
Antwort auf Beitrag Nr.: 58.238.184 von f1re am 17.07.18 23:14:04Hi f1re,

Ja, Verzögerungen. Denke mal bei technischen Entwicklungen gehört das zum täglichen Brot.
Ansonsten, s.o., zäh ...
Gruß, praesens

Ja, Verzögerungen. Denke mal bei technischen Entwicklungen gehört das zum täglichen Brot.
Ansonsten, s.o., zäh ...

Gruß, praesens
Antwort auf Beitrag Nr.: 58.238.199 von praesens am 17.07.18 23:15:55Hi praesens,
Danke für deine Meinung.
Dann schauen wir morgen mal was CSE und OTC dazu sagen.
Gute Nacht
Danke für deine Meinung.
Dann schauen wir morgen mal was CSE und OTC dazu sagen.
Gute Nacht
Interessanter Turnaround-Kandidat vom aktuellen Niveau.
Quartalsbericht
https://mobile.tmxmoney.com/quote/readnews/?id=4757942685490…6 Mio Cash, tolles Produkt, gute Unterstützer und weniger als 15 Mio Börsenwert...

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 58.444.396 von Greenfoxi am 14.08.18 15:34:46Heute sieht es übel aus...unter 0,10CAD...
Heimlich still und leiste steigt der Kurs die letzten Tage. Noch jemand an Board hier? Volumen war auch sehr stark die letzten Tage.
https://www.advfn.com/stock-market/CNSX/IME/chart
https://www.advfn.com/stock-market/CNSX/IME/chart
Bin auch investiert, gucke auch jeden Tag, als der Kurs vor ein paar Tagen tiefer stand, habe ich es verpasst ein wenig nachzulegen. Was mich gestern auch gewundert hat, wenig Umsatz in Deutschland aber ziemlich hoher in Kanada. Ob die was wissen, was wir nicht wissen?? Sollten September nicht Mitteilungen kommen?? Lassen wir uns mal überraschen. Daumen drücken.
Antwort auf Beitrag Nr.: 58.725.712 von florfliege am 18.09.18 11:00:16Umsatz in D ist erstmal zweitrangig. Wichtig ist der Umsatz an der Heimatbörse.
Wenn gerade bei CAN-Werten das Interesse in D größer ist als in CAN, dann geht das meist nach hinten los.
Ich denke hier kommt bald was. Habe noch etwas verbilligt neulich zu 0,08 EUR, bin jetzt minimal im Plus. Kann gern starten jetzt
Wenn gerade bei CAN-Werten das Interesse in D größer ist als in CAN, dann geht das meist nach hinten los.

Ich denke hier kommt bald was. Habe noch etwas verbilligt neulich zu 0,08 EUR, bin jetzt minimal im Plus. Kann gern starten jetzt
Der grössere Umsatz lässt mich auch hoffen.....Bin auch grün, hoffentlich für eine längere Zeit.
Die Geschichte und Fakten klingen plausibel und logisch. Abwarten und Tee trinken.
Schönen Tag noch wünscht florfliege
Die Geschichte und Fakten klingen plausibel und logisch. Abwarten und Tee trinken.
Schönen Tag noch wünscht florfliege
Antwort auf Beitrag Nr.: 58.724.290 von boersenkeks am 18.09.18 08:47:03
bin noch dabei, hatte aber zwischdurch mal verbilligt (siehe meine Forumhistorie bei IME).
Mein Average liegt jetzt bei 0,068 €, ich handele aber in Kanada.
Die Nachrichtenlage war zuletzt recht dünn, das Management ist allerdings beachtenswert (fachlich hochgradig und sehr gut vernetzt). Wenn der Prototyp endlich Richtung Serienreife geht und der Verkauf wirklich anläuft sehe ich weiter gute Chancen, zumal ja bei Blasenkrebs nicht Endstation sein muss...
Vllt. hilfreich: Die letzte Kapitalerhöhung lief zu 0,22 CAD, allerdings nur an ausgesuchte Investoren (als der Kurs bei 0,31 (!) CAD stand)....
Gruß Greenfoxi
Fundamental unverändert
Hi Keks,bin noch dabei, hatte aber zwischdurch mal verbilligt (siehe meine Forumhistorie bei IME).

Mein Average liegt jetzt bei 0,068 €, ich handele aber in Kanada.

Die Nachrichtenlage war zuletzt recht dünn, das Management ist allerdings beachtenswert (fachlich hochgradig und sehr gut vernetzt). Wenn der Prototyp endlich Richtung Serienreife geht und der Verkauf wirklich anläuft sehe ich weiter gute Chancen, zumal ja bei Blasenkrebs nicht Endstation sein muss...

Vllt. hilfreich: Die letzte Kapitalerhöhung lief zu 0,22 CAD, allerdings nur an ausgesuchte Investoren (als der Kurs bei 0,31 (!) CAD stand)....

Gruß Greenfoxi
Hallo Greenfoxi,
hast Du vielleicht mehr Info über den Zeitplan Tests, Zulassung? Man findet wenig Info über den zeitlichen Ablauf, Zulassung, Ergebnisse etc. Danke und grüne Grüsse
hast Du vielleicht mehr Info über den Zeitplan Tests, Zulassung? Man findet wenig Info über den zeitlichen Ablauf, Zulassung, Ergebnisse etc. Danke und grüne Grüsse
Neues Boardmitglied
Nach vollen 7 Wochen (!) mal wieder eine offizielle Verlautbarung. 
https://mobile.tmxmoney.com/quote/readnews/?id=5014113831175…
Scheint ein alter Hase zu sein.
Wenn er IME weiter nach vorn bringt..., soll es mir sehr recht sein.

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 58.736.458 von florfliege am 19.09.18 11:50:14
kann man mal lesen.
https://www.goldinvest.de/aus-der-redaktion/2127-imagin-medi…
Ist zwar von GOLGINVEST
 , fasst aber so ziemlich den aktuellen Stand zusammen.
, fasst aber so ziemlich den aktuellen Stand zusammen. 
Gruß Greenfoxi
Hilft das etwas?
Hi Flori,kann man mal lesen.
https://www.goldinvest.de/aus-der-redaktion/2127-imagin-medi…
Ist zwar von GOLGINVEST

 , fasst aber so ziemlich den aktuellen Stand zusammen.
, fasst aber so ziemlich den aktuellen Stand zusammen. 
Gruß Greenfoxi
Hallo Greenfoxi,
danke für die Infos. Wollte sowieso noch schreiben. Ich hatte recherchiert und in einem Schreiben von imagin August2018, steht dass diese ein beschleunigtes Verfahren anstreben. Dieses beschleunigte Verfahren würde bedeuten, dass die FDA, wegen der bedeutsamen Behandlung bei Menschen, dieses Verfahren schneller zulassen würde. Bei ClinicalTrials.gov fand ich, dass die Studien an Menschen schon abgeschlossen seien. Ich muss mich an die Üb ersetzungen halten, kann kein so gutes medizinisches Englisch. Was ich aber nicht finde, ist die Einreichung von imagin bei der FDA, wann das geschehen sein sollte oder noch eingereicht wird. Kannst Du mir schreiben, ob ich da richtig liege oder etwas falsch deute? Auf jeden Fall werde ich noch Aktien kaufen und dann mal abwarten, liest sich alles vielversprechend. Hast Du noch andere Favoriten ? Danke nochmals und Grüsse Florfliege
danke für die Infos. Wollte sowieso noch schreiben. Ich hatte recherchiert und in einem Schreiben von imagin August2018, steht dass diese ein beschleunigtes Verfahren anstreben. Dieses beschleunigte Verfahren würde bedeuten, dass die FDA, wegen der bedeutsamen Behandlung bei Menschen, dieses Verfahren schneller zulassen würde. Bei ClinicalTrials.gov fand ich, dass die Studien an Menschen schon abgeschlossen seien. Ich muss mich an die Üb ersetzungen halten, kann kein so gutes medizinisches Englisch. Was ich aber nicht finde, ist die Einreichung von imagin bei der FDA, wann das geschehen sein sollte oder noch eingereicht wird. Kannst Du mir schreiben, ob ich da richtig liege oder etwas falsch deute? Auf jeden Fall werde ich noch Aktien kaufen und dann mal abwarten, liest sich alles vielversprechend. Hast Du noch andere Favoriten ? Danke nochmals und Grüsse Florfliege
Hallo Greenfoxi, ist so ruhig hier. Habe nochmal ein wenig aufgestockt. Müssten doch langsam mal wieder Nachrichten kommen oder es gibt einen Paukenschlag....Was denkst Du?
Grüsse Florfliege
Grüsse Florfliege
Antwort auf Beitrag Nr.: 59.052.244 von florfliege am 25.10.18 11:42:39Denke ich werde auch nochmal nachlegen. Glaube weiterhin an die Story und lasse mich von dem niedrigen Umsatz der den Preis nach unten drückt nicht verunsichern.
Gruß f1re
Gruß f1re
Hallo an alle Mitleidenden,
ist so ruhig hier, von der Fa. kommt keine Info, nicht mal eine kleine Info.
Könnten doch mal mitteilen, wie der Stand der Dinge ist.
Produkt überzeugt mich, sonst hätte ich ja nix investiert, aber diese Funkstille. Na ja, abwarten und Tee trinken.
Florfliege grüsst die Investierten
ist so ruhig hier, von der Fa. kommt keine Info, nicht mal eine kleine Info.
Könnten doch mal mitteilen, wie der Stand der Dinge ist.
Produkt überzeugt mich, sonst hätte ich ja nix investiert, aber diese Funkstille. Na ja, abwarten und Tee trinken.
Florfliege grüsst die Investierten
Antwort auf Beitrag Nr.: 59.104.754 von florfliege am 31.10.18 15:06:02Aktuell wurden einige Warrants gewandelt:
"Imagin Medical receives $954,854 from warrant exercise. The exercise price of 9,508,540 of the Warrants was $0.10 and 25,000 of the Warrants was $0.16."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2677…
"Imagin Medical receives $954,854 from warrant exercise. The exercise price of 9,508,540 of the Warrants was $0.10 and 25,000 of the Warrants was $0.16."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2677…
damit sollte der Druck von der Aktie weg sein - die nächsten warrants sind 0.16+x und nicht viele
Vielen Dank für die Infos.
Ich kann die Homepage der Firma leider nicht finden. Könnte mir da jemand helfen?
Ich kann die Homepage der Firma leider nicht finden. Könnte mir da jemand helfen?
Antwort auf Beitrag Nr.: 59.121.608 von Badenfinke am 02.11.18 10:38:44https://imaginmedical.com
seit 2 Tagen ist in Kanada starkes Kaufinteresse zu verzeichnen...
irgendwas köchelt
FDA?
irgendwas köchelt
FDA?
CEO von Imagin Medical hat sich in der Runde hier präsentiert
auch interessant Desert Mountain Energy und Nexoptic - die beiden Herren die neben Jim Hutchins sitzen
auch interessant Desert Mountain Energy und Nexoptic - die beiden Herren die neben Jim Hutchins sitzen
Antwort auf Beitrag Nr.: 59.237.305 von techinvestor69 am 16.11.18 21:57:22Ich weiß bei Pennystocks sollte man nicht mit charttechnik anfangen aber so wie es aussieht baut sich grade eine schöne W-Formation auf. Wenn ich richtig liege werden wir bald ein neues ath sehen ;-)
Antwort auf Beitrag Nr.: 59.261.875 von f1re am 20.11.18 19:16:02
Kleine Position
Das wäre ja sehr schön, bin gespannt ob tatsächlich eine News geleakt wurde. Steht nicht noch eine FDA Entscheidung aus?
Antwort auf Beitrag Nr.: 59.262.796 von Plomo_o_plata am 20.11.18 20:59:05Wenn ich es richtig bei stockhouse rausgelesen habe gibt es so direkt keine news sondern das sind finanzstarke Investoren die aufgesprungen sind nachdem der CEO diese besucht und Werbung gemacht hat.
Die werden wohl nicht investieren wenn an der Story nichts dran ist....
Außer die zocken das Ding hoch und lassen es dann fallen. Kann ich mir aber nicht vorstellen
Die werden wohl nicht investieren wenn an der Story nichts dran ist....
Außer die zocken das Ding hoch und lassen es dann fallen. Kann ich mir aber nicht vorstellen
Lebenszeichen
Nach dem Erlös von 1 Mio CAD aus eingelösten Warrants Anfang November mal wieder ´ne News, allerdings ohne Infos zum aktuellen Arbeitsstand... 
Hat doch ein "unbekannter Dritter" Reklame für IME gemacht...

https://mobile.tmxmoney.com/quote/readnews/?id=5025921814400…
Gruß Greenfoxi
Hallo Greenfoxi,
schaue auch jeden Tag mehrmals kurz rein, ob eine Nachricht von der Firma da ist. Keine Mitteilungen über Zulassung oder andere Fortschritte. Tee trinken und abwarten.
Grüsse florfliege
schaue auch jeden Tag mehrmals kurz rein, ob eine Nachricht von der Firma da ist. Keine Mitteilungen über Zulassung oder andere Fortschritte. Tee trinken und abwarten.
Grüsse florfliege
Antwort auf Beitrag Nr.: 59.323.256 von Greenfoxi am 29.11.18 09:01:37Ja so ein Mist das verunsichert natürlich jetzt manche Anleger...grade kurz bevor die w Formation zünden konnte...
Neues Zahlenwerk:
Imagin Medical Reports Fiscal Year End 2018 Results
Insgesamt wurden 2018 $7,958,086 ausgegeben.
Noch vorhandenes Cash: $5,818,840
Ausblick:
Im 1. Halbjahr 2019 will man Produkt finalisieren und klinische Studien initiieren. Außerdem will man mit der FDA den Regulierungspfad definieren. 2020 will man das Produkt dann auf den Markt bringen können. Klingt mir recht ambitioniert.
Wie weit das Cash noch reicht, sagte man nicht. Aber wenn dieses Jahr rund 8 MIO ausgegeben wurden, nur noch 5,8 vorhanden sind, wird für klinische Studien, etc. sicher noch einiges an Cash benötigt, um auch bis zur Kommerzialisierung zu kommen. Bin mal gespannt, über welche Wege man das Geld beschafft.
https://seekingalpha.com/pr/17364989-imagin-medical-reports-…
Imagin Medical Reports Fiscal Year End 2018 Results
Insgesamt wurden 2018 $7,958,086 ausgegeben.
Noch vorhandenes Cash: $5,818,840
Ausblick:
Im 1. Halbjahr 2019 will man Produkt finalisieren und klinische Studien initiieren. Außerdem will man mit der FDA den Regulierungspfad definieren. 2020 will man das Produkt dann auf den Markt bringen können. Klingt mir recht ambitioniert.
Wie weit das Cash noch reicht, sagte man nicht. Aber wenn dieses Jahr rund 8 MIO ausgegeben wurden, nur noch 5,8 vorhanden sind, wird für klinische Studien, etc. sicher noch einiges an Cash benötigt, um auch bis zur Kommerzialisierung zu kommen. Bin mal gespannt, über welche Wege man das Geld beschafft.
https://seekingalpha.com/pr/17364989-imagin-medical-reports-…
Antwort auf Beitrag Nr.: 59.486.223 von boersenkeks am 21.12.18 06:38:02das Management muss die 0,38 CAD warrants ins Spiel bringen, dann fließt genug Cash in die Box
Antwort auf Beitrag Nr.: 59.486.223 von boersenkeks am 21.12.18 06:38:02Nach Bilanzstichtag kamen nochmal 500k in die Box.
SUBSEQUENT EVENTS
Subsequent to the year end, the following occurred:
• 5,133,340 finance warrants with an exercise price of $0.10 were exercised for gross proceeds of $513,334;
Quelle: www.stockwatch.com
SUBSEQUENT EVENTS
Subsequent to the year end, the following occurred:
• 5,133,340 finance warrants with an exercise price of $0.10 were exercised for gross proceeds of $513,334;
Quelle: www.stockwatch.com
Antwort auf Beitrag Nr.: 59.487.408 von techinvestor69 am 21.12.18 09:22:05
https://ceo.ca/ime?fcbea5566922
Habe im Sofa nach Kleingeld gesucht und nochmal ein paar Shares draufgelegt.
Moin
Bin ja seit November dabei und warte auf Neuigkeiten, im Can Forum sind sie der Meinung es könnte Donnerstag Neuigkeiten geben? Was könnte das sein, Nachrichten von der FDA? https://ceo.ca/ime?fcbea5566922
Habe im Sofa nach Kleingeld gesucht und nochmal ein paar Shares draufgelegt.
Antwort auf Beitrag Nr.: 59.326.988 von f1re am 29.11.18 16:55:46
Meinungen
Meinungen?
Antwort auf Beitrag Nr.: 59.590.253 von Plomo_o_plata am 09.01.19 09:43:06Könnte was dran sein. In der Vergangenheit haben die CANs schon ein paar mal bei IME mit der Ankündigung von news richtig gelegen.
Ob es FDA ist oder was anderes mal sehen.
Hatte auch überlegt nochmals nachzulegen aber ich warte erstmal ab.
Ob es FDA ist oder was anderes mal sehen.
Hatte auch überlegt nochmals nachzulegen aber ich warte erstmal ab.
Antwort auf Beitrag Nr.: 59.591.567 von f1re am 09.01.19 11:50:36
Donnerstag
Sehe ich genauso, dann lassen wir uns mal überraschen was am Donnerstag so rüberkommt. Solange schaue ich mal bei Futura medical rein 😉
Hallo, finde das Produkt auch vielversprechend. Wenn ich das mit Donnerstag früher gewusst hätte, hätte ich mit meinem Kauf von Futura Medical Aktien noch gewartet. Abwarten und Tee trinken und Daumen drücken. Grüsse Florfliege
Antwort auf Beitrag Nr.: 59.592.917 von florfliege am 09.01.19 14:26:16
da dürfte in Zukunft einiges an positiven Newsflow kommen. Laut CAN Forum soll noch eine bessere News in Kürze kommen. Mal schauen.
https://ceo.ca/ime?496f5f4a720f
news wie angekündigt
https://ceo.ca/@nasdaq/imagin-medical-posts-new-presentation…" target="_blank" rel="nofollow ugc noopener">https://ceo.ca/@nasdaq/imagin-medical-posts-new-presentation…da dürfte in Zukunft einiges an positiven Newsflow kommen. Laut CAN Forum soll noch eine bessere News in Kürze kommen. Mal schauen.
https://ceo.ca/ime?496f5f4a720f
News überfällig
Nachdem es mal eine Zeit lang einen steten Newsflow gab, sind die letzten Monate eher informationsschwach... 
Allerdings ist die neue Präsi durchaus lesenswert...

https://imaginmedical.com/wp-content/uploads/2019/01/Imagin-…
Gruß Greenfoxi
Antwort auf Beitrag Nr.: 59.640.773 von Greenfoxi am 16.01.19 08:15:50Präsentation online:
http://noble.mediasite.com/Mediasite/Play/14eeb5b989c64caea9…
http://noble.mediasite.com/Mediasite/Play/14eeb5b989c64caea9…
Antwort auf Beitrag Nr.: 59.740.620 von f1re am 29.01.19 23:44:50
Super video
Denke auch das bald eine FDA konforme Testreihe starten wird. Denke günstiger wird’s hier nicht.
Antwort auf Beitrag Nr.: 59.740.704 von Plomo_o_plata am 30.01.19 00:21:43Der CEO kann gut präsentieren, aber das können ja die meisten Amis.
Bleibt zu hoffen, dass die FDA innerhalb von drei Wochen (shutdown Zeitfenster) grünes Licht für den Start des Trials gibt.
Interessant, dass der CFO seine Firma im letzten Jahr für 100 Mio. USD an Medtronic verkauft hat.
Wäre das auch ein möglicher Käufer für Imagin?
Bleibt zu hoffen, dass die FDA innerhalb von drei Wochen (shutdown Zeitfenster) grünes Licht für den Start des Trials gibt.
Interessant, dass der CFO seine Firma im letzten Jahr für 100 Mio. USD an Medtronic verkauft hat.
Wäre das auch ein möglicher Käufer für Imagin?
Antwort auf Beitrag Nr.: 59.743.233 von techinvestor69 am 30.01.19 11:01:03
FDA
Moin, ich weiß nicht inwieweit die FDA nun Verzug in ihren Arbeitsabläufen hat, aber ich erwarte hier eine Meldung über einen offiziellen Trial mit einer neuen FDA aproved Human Test Trial. Erwarte die Meldung im Februar, mal schauen ob an den Gerüchten etwas dran ist. Mir ist aufgefallen in der Präsentation im Video, dass dort etwas stand wie: es gibt kein organisches Wachstum sondern Wachstum entsteht in der Branche durch „übernahmen“ . Naja und die Erfahrung des Board ist in der Tat sehr herausragend. Mal schauen was die Tage bringen. Fakt ist der Boden sollte hier bei 0,07xxx liegen.
Gov’t Shutdown Delays Device Applications and More
https://ryortho.com/breaking/govt-shutdown-delays-device-app…
https://ryortho.com/breaking/govt-shutdown-delays-device-app…
es kommt Bewegung auf in Ca
Antwort auf Beitrag Nr.: 59.827.884 von Plomo_o_plata am 08.02.19 18:49:59Ja, nur in die falsche Richtung
Letztes Quartal wurden $1,011,586 ausgegeben, aktuell noch $5,365,497 in der Kasse. Reicht also noch knapp 5 Quartale.
"IMAGIN MEDICAL REPORTS FISCAL 2019 FIRST QUARTER RESULTS
Imagin Medical Inc. has released financial results for the three months ended Dec. 31, 2018. All amounts, unless otherwise specified, are expressed in Canadian dollars and are presented in accordance with International Financial Reporting Standards (IFRS).
Recent Corporate Developments
Completed, via Imagin's opto-electronic design firm, Optel, Inc., the Proof of Concept Phase of the redesigned i/BlueTM Imaging System's control unit and validated the performance of the light source and imaging modules.
Met with the U.S. Food and Drug Administration ("FDA") to discuss the i/Blue Imaging System's regulatory path and the potential need for a clinical study. Imagin will use the FDA's feedback to help refine its regulatory strategy and expects that discussions with the FDA will be ongoing.
Welcomed Chris Bleck as a director of the Company.
Received proceeds of approximately $1 million as a result of the exercise of 9,533,540 previously issued common share purchase warrants.
"This quarter, Optel completed the redesign of the i/Blue Imaging System's control unit, resulting in an innovative modular system that has a 90% smaller footprint than our original prototype," said Jim Hutchens, Imagin's President and CEO. "We believe the redesign has resulted in a highly manufacturable and cost-effective product that conforms to all FDA specifications. As we continue the dialogue with the FDA to refine our regulatory strategy, we will work with Optel to prepare the system's Dual Control Camera Unit for functional unit testing in the coming weeks."
Summary Fiscal 2019 First Quarter Financial Results
Total operating expenses for the first quarter of fiscal 2019 were $1,011,586, compared with $1,270,498 for the same quarter in 2018, and consisted primarily of research and development ("R&D") and general and administrative ("G&A") expenses. R&D expenses for the first quarter of fiscal 2019 were $372,494 compared with $38,012 in the same quarter in 2018. The increase in R&D was primarily attributable to development, design, engineering and regulatory expenses. G&A expenses for the first quarter of fiscal 2019 were $639,092 compared with $1,232,486 for the same quarter in 2018. The decrease in G&A expenses was primarily attributable to the decrease in stock-based compensation.
Net loss for the three months ended December 31, 2018 was $1,011,586, or $0.01 loss per common share, compared to a net loss of $1,270,498, or $0.02 loss per common share for the three months ended December 31, 2017.
Liquidity and Outstanding Share Capital
As at December 31, 2018, the Company had cash of $5,365,497.
As at February 27, 2019, Imagin had an unlimited number of authorized common shares with 139,060,278 common shares issued and outstanding."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2723…
"IMAGIN MEDICAL REPORTS FISCAL 2019 FIRST QUARTER RESULTS
Imagin Medical Inc. has released financial results for the three months ended Dec. 31, 2018. All amounts, unless otherwise specified, are expressed in Canadian dollars and are presented in accordance with International Financial Reporting Standards (IFRS).
Recent Corporate Developments
Completed, via Imagin's opto-electronic design firm, Optel, Inc., the Proof of Concept Phase of the redesigned i/BlueTM Imaging System's control unit and validated the performance of the light source and imaging modules.
Met with the U.S. Food and Drug Administration ("FDA") to discuss the i/Blue Imaging System's regulatory path and the potential need for a clinical study. Imagin will use the FDA's feedback to help refine its regulatory strategy and expects that discussions with the FDA will be ongoing.
Welcomed Chris Bleck as a director of the Company.
Received proceeds of approximately $1 million as a result of the exercise of 9,533,540 previously issued common share purchase warrants.
"This quarter, Optel completed the redesign of the i/Blue Imaging System's control unit, resulting in an innovative modular system that has a 90% smaller footprint than our original prototype," said Jim Hutchens, Imagin's President and CEO. "We believe the redesign has resulted in a highly manufacturable and cost-effective product that conforms to all FDA specifications. As we continue the dialogue with the FDA to refine our regulatory strategy, we will work with Optel to prepare the system's Dual Control Camera Unit for functional unit testing in the coming weeks."
Summary Fiscal 2019 First Quarter Financial Results
Total operating expenses for the first quarter of fiscal 2019 were $1,011,586, compared with $1,270,498 for the same quarter in 2018, and consisted primarily of research and development ("R&D") and general and administrative ("G&A") expenses. R&D expenses for the first quarter of fiscal 2019 were $372,494 compared with $38,012 in the same quarter in 2018. The increase in R&D was primarily attributable to development, design, engineering and regulatory expenses. G&A expenses for the first quarter of fiscal 2019 were $639,092 compared with $1,232,486 for the same quarter in 2018. The decrease in G&A expenses was primarily attributable to the decrease in stock-based compensation.
Net loss for the three months ended December 31, 2018 was $1,011,586, or $0.01 loss per common share, compared to a net loss of $1,270,498, or $0.02 loss per common share for the three months ended December 31, 2017.
Liquidity and Outstanding Share Capital
As at December 31, 2018, the Company had cash of $5,365,497.
As at February 27, 2019, Imagin had an unlimited number of authorized common shares with 139,060,278 common shares issued and outstanding."
https://www.stockwatch.com/News/Item.aspx?bid=Z-C%3aIME-2723…
Noch jemand da?
Heute schöner Umsatz an der CSE ;-)
Gruß
Heute schöner Umsatz an der CSE ;-)
Gruß
netter Volume Spike
ich rieche moneh
totes Kapital bis 2020
Antwort auf Beitrag Nr.: 60.288.909 von techinvestor69 am 05.04.19 15:40:15Ganz sicher?
Antwort auf Beitrag Nr.: 60.734.766 von ThatsIt35 am 04.06.19 21:21:09schon mal auf den Kurs geschaut?
Sirona läuft für mich weitaus besser.
Sirona läuft für mich weitaus besser.

Zahlen soweit unspektakulär.
Etwas weniger Geld ausgegeben, als im Vor-Quartal: $926,070 gegenüber $1,011,586
CASH: 4,5 MIO
Pro Quartal werden also rund 1 MIO verbraucht.
Theoretisch also noch 4 Quartale finanziert, dürfte also bald frisches Geld nötig werden.
"“This quarter, Imagin completed the first step of the functional system stage, with both components of the Functional Unit operating as anticipated to provide simultaneous high-resolution images in both blue and white light modes,” said Jim Hutchens, Imagin’s President and CEO. “In addition, data from the previous Research Study conducted at the University of Rochester Medical Center was used to refine and optimize the technology for performance, size and cost, resulting in a current Functional Unit that is 70% smaller than the proof of principle prototype. As we continued to progress through the functional system stage, we conducted focus groups at the American Urological Association’s Annual Meeting last month. We are now using the valuable feedback we gathered to advance the functional system phase toward pilot production later this year.”"
https://seekingalpha.com/pr/17541494-imagin-medical-reports-…
Etwas weniger Geld ausgegeben, als im Vor-Quartal: $926,070 gegenüber $1,011,586
CASH: 4,5 MIO
Pro Quartal werden also rund 1 MIO verbraucht.
Theoretisch also noch 4 Quartale finanziert, dürfte also bald frisches Geld nötig werden.
"“This quarter, Imagin completed the first step of the functional system stage, with both components of the Functional Unit operating as anticipated to provide simultaneous high-resolution images in both blue and white light modes,” said Jim Hutchens, Imagin’s President and CEO. “In addition, data from the previous Research Study conducted at the University of Rochester Medical Center was used to refine and optimize the technology for performance, size and cost, resulting in a current Functional Unit that is 70% smaller than the proof of principle prototype. As we continued to progress through the functional system stage, we conducted focus groups at the American Urological Association’s Annual Meeting last month. We are now using the valuable feedback we gathered to advance the functional system phase toward pilot production later this year.”"
https://seekingalpha.com/pr/17541494-imagin-medical-reports-…
keine Riesen-Size, aber CEO und CFO mit dabei - ein Signal an den Markt
Antwort auf Beitrag Nr.: 60.796.531 von techinvestor69 am 13.06.19 10:39:25
https://ceo.ca/@nasdaq/imagin-medical-announces-stock-option…
Bonus
Jemand bekommt einen Bonus https://ceo.ca/@nasdaq/imagin-medical-announces-stock-option…
Imagin Medical Reports Fiscal 2019 Third Quarter Results https://www.globenewswire.com/news-release/2019/08/27/190741…
News:
September 26, 2019 – Imagin Medical Announces Meeting Regarding i/Blues’s Regulatory Pathway
Hallo, es gibt Neuigkeiten, scheint ja alles gut zu laufen. Vielleicht kommt ja jetzt ein wenig mehr Bewegung rein.
Grüsse florfliege
September 26, 2019 – Imagin Medical Announces Meeting Regarding i/Blues’s Regulatory Pathway
Hallo, es gibt Neuigkeiten, scheint ja alles gut zu laufen. Vielleicht kommt ja jetzt ein wenig mehr Bewegung rein.
Grüsse florfliege
Neuer Artikel über Imagin Medical und die möglichen Chancen:
https://www.investwithpal.com/post/imagin-medical-inc-junior…
https://www.investwithpal.com/post/imagin-medical-inc-junior…
Übersetzung: IMAGIN MEDICAL BESTÄTIGT FUNKTIONALE EINHEITEN IM ZEITPLAN FÜR DIE ÜBERPRÜFUNG DER PRODUKTDESIGN_________________________________________________Vancouver, B.C. und Boston, MA, 12. November 2019 - Imagin Medical (CSE: IME) (OTCQB: IMEXF) (Frankfurt und Stuttgarter Symbol: DPD2) („Imagin“ oder das „Unternehmen“) meldeten heute weitere Fortschritte bei der Verifizierung ihres i / BlueImagingSystemfunktionales Produkt. Das i / Blue-System befindet sich derzeit in der Entwurfsüberprüfung des Entwicklungsprozesses. Seit der letzten Fortschrittsaktualisierung von Imagin am 15. Juli 2019 hat das Unternehmen Komponenten für den ursprünglichen Aufbau der Funktionseinheiten gekauft und erhalten. Komponenten wurden von etablierten Lieferanten gekauft, um die kundenspezifischen Spezifikationen von Imagin zu erfüllen. Das i / Blue-System besteht aus fünf weiteren Subsystemen. Dazu gehören: 1) Steuergerät, das die Licht- und Energiequellen beherbergt2) Doppelkamera-Handstück3) optische Lichtkabel4) Gerätewagen für medizinische Zwecke und5) Drucker für medizinische Zwecke. Das System wird mit einem Zystoskop von Drittanbietern verwendet, das nicht von Imagin bereitgestellt wird. Derzeit werden Komponenten für die Steuereinheit zusammengebaut, die die Lichtquelle mit zwei Wellenlängen in das Handstück mit zwei Kameras integrieren und die gleichzeitige Übertragung von Weißlicht- und Blaulichtbildern zur Seite ermöglichen. neben dem Monitor. Die eingehende Evaluierung dieses Systems wird von einem unabhängigen Prüflabor durchgeführt, um die Einhaltung der Vorschriften für elektrische Sicherheit, Strahlenemission und Störanfälligkeit sowie anderer Anforderungen zu bestätigen. Daten aus diesen unabhängigen Labortests werden mit Daten aus internen Tests kombiniert, die von Imagins optoelektronischem Designunternehmen Optel, technischen Berechnungen, Komponentenlieferanten und der Analyse wettbewerbsfähiger Geräte durchgeführt wurden. Alle diese Daten werden als Grundlage für den Verifizierungsbericht des Unternehmens dienen. Wie bereits prognostiziert, geht Imagin davon aus, dass diese Arbeiten vor Ende 2019 abgeschlossen sein werden. Jim Hutchens, President und CEO von Imagin, kommentierte: „Wir freuen uns über die bisherigen Fortschritte. Die funktionsfähigen Einheiten sind für die Entwurfsüberprüfung im Zeitplan, was den Weg zu unseren Pilotproduktionsläufen darstellt. Während dieses Prozesses arbeiten wir weiterhin mit der US-amerikanischen Food and Drug Administration (FDA) zusammen, um die endgültigen Anforderungen für die Erlangung der Marktzulassung zu bestimmen. “
Hallo, läuft ja ziemlich gut....hier neue Artikel von der IM Website, steht unter news - articles. Für den ersten Artikel haben die von IM bezahlt, ist aber sehr aufschlussreich und interessant https://imaginmedical.com/wp-content/uploads/2020/01/Nationa… und der 2. Artikel wurde nicht bezahlt und der Schreiber hält auch keine IM Aktien, auch sehr interessant: https://imaginmedical.com/wp-content/uploads/2020/01/Streetw… Auf spannende Wochen und Monate
Weiteres Lebenszeichen...
https://web.tmxmoney.com/article.php?newsid=7630672305054349… 
DYOR

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 62.303.663 von florfliege am 07.01.20 15:08:18
Hilft das auch bei Corona Covid ?
Zitat von florfliege: Hallo, läuft ja ziemlich gut....hier neue Artikel von der IM Website, steht unter news - articles. Für den ersten Artikel haben die von IM bezahlt, ist aber sehr aufschlussreich und interessant https://imaginmedical.com/wp-content/uploads/2020/01/National-Investor-Imagin-Profile-Jan-2020.pdf und der 2. Artikel wurde nicht bezahlt und der Schreiber hält auch keine IM Aktien, auch sehr interessant: https://imaginmedical.com/wp-content/uploads/2020/01/Streetwise-Report-IME-Poised-For-Breakout-Dec-19.pdf. Auf spannende Wochen und Monate
Hilft das auch bei Corona Covid ?

Antwort auf Beitrag Nr.: 63.252.073 von Coriolanos am 06.04.20 15:19:48Hallo, in diesen Zeiten sollte man den Humor nicht verlieren, lachen ist ja gut fürs Immunsystem. Hier im Forum für sich ja nicht viel. Im Moment auch keine Neuigkeiten seitens IM. Wie ist Deine Einschätzung über IM? Gruss florfliege
https://finance.yahoo.com/news/imagin-medical-receives-appro…
vor 1 Stunde
Imagin Medical Receives Approval to Reprice and Extend Term of Warrants
[ACCESSWIRE]
ACCESSWIREApril 9, 2020
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / April 9, 2020 / Imagin Medical (IME.CN)(IMEXF)(DPD2.F)(DPD2.F) ("Imagin" or the "Company") announces that it has received approval from the Canadian Securities Exchange to extend the term of an aggregate of 17,919,820 outstanding common share purchase warrants (the "Warrants"), issued pursuant to a private placement that closed in two tranches on April 12, 2018 and April 19, 2018. The Warrants had a term of two years and one full Warrant was exercisable to acquire one common share at a price of $0.38 per share. The Company has received approval to extend the term of the Warrants to June 30, 2020 and to reduce the exercise price of the Warrants to $0.05 per share. All other terms of the Warrants shall remain the same.
The securities referred to in this news release have not been, nor will they be, registered under the United States Securities Act of 1933, as amended, and may not be offered or sold within the United States or to, or for the account or benefit of, U.S. persons absent U.S. registration or an applicable exemption from the U.S. registration requirements. This news release does not constitute an offer for sale of securities for sale, nor a solicitation for offers to buy any securities.
About Imagin Medical
Imagin Medical is a surgical imaging company focused on advancing new methods of visualizing cancer during minimally invasive procedures. The Company believes its first product, the i/Blue™ Imaging System, with its proprietary optics and light sensors, will greatly increase the efficiency and accuracy of detecting cancer for removal, helping to reduce recurrence rates. The Company's initial focus is bladder cancer. Learn more at www.imaginmedical.com.
Forward-Looking Statements
Information set forth in this news release contains forward-looking statements. These statements reflect management's current estimates, beliefs, intentions and expectations; they are not guarantees of future performance. The Company cautions that all forward- looking statements are inherently uncertain, and that actual performance may be affected by a number of material factors, many of which are beyond the Company's control. Accordingly, actual and future events, conditions and results may differ materially from the estimates, beliefs, intentions and expectations expressed or implied in the forward-looking information. Specifically, there is no assurance the Company's imaging system will work in the manner expected. Except as required under applicable securities legislation, the Company undertakes no obligation to publicly update or revise forward-looking information. The CSE has neither approved nor disapproved the information contained herein and
does not accept responsibility for the adequacy or accuracy of this news release.
Contacts:
Stephen Kilmer,
Investor Relations
Telephone: 647-872-4849
Email: stephen@kilmerlucas.com
Jim Hutchens,
President & CEO
Telephone: 833-246-2446
SOURCE: Imagin Medical
View source version on accesswire.com:
https://www.accesswire.com/584491/Imagin-Medical-Receives-Ap…
vor 1 Stunde
Imagin Medical Receives Approval to Reprice and Extend Term of Warrants
[ACCESSWIRE]
ACCESSWIREApril 9, 2020
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / April 9, 2020 / Imagin Medical (IME.CN)(IMEXF)(DPD2.F)(DPD2.F) ("Imagin" or the "Company") announces that it has received approval from the Canadian Securities Exchange to extend the term of an aggregate of 17,919,820 outstanding common share purchase warrants (the "Warrants"), issued pursuant to a private placement that closed in two tranches on April 12, 2018 and April 19, 2018. The Warrants had a term of two years and one full Warrant was exercisable to acquire one common share at a price of $0.38 per share. The Company has received approval to extend the term of the Warrants to June 30, 2020 and to reduce the exercise price of the Warrants to $0.05 per share. All other terms of the Warrants shall remain the same.
The securities referred to in this news release have not been, nor will they be, registered under the United States Securities Act of 1933, as amended, and may not be offered or sold within the United States or to, or for the account or benefit of, U.S. persons absent U.S. registration or an applicable exemption from the U.S. registration requirements. This news release does not constitute an offer for sale of securities for sale, nor a solicitation for offers to buy any securities.
About Imagin Medical
Imagin Medical is a surgical imaging company focused on advancing new methods of visualizing cancer during minimally invasive procedures. The Company believes its first product, the i/Blue™ Imaging System, with its proprietary optics and light sensors, will greatly increase the efficiency and accuracy of detecting cancer for removal, helping to reduce recurrence rates. The Company's initial focus is bladder cancer. Learn more at www.imaginmedical.com.
Forward-Looking Statements
Information set forth in this news release contains forward-looking statements. These statements reflect management's current estimates, beliefs, intentions and expectations; they are not guarantees of future performance. The Company cautions that all forward- looking statements are inherently uncertain, and that actual performance may be affected by a number of material factors, many of which are beyond the Company's control. Accordingly, actual and future events, conditions and results may differ materially from the estimates, beliefs, intentions and expectations expressed or implied in the forward-looking information. Specifically, there is no assurance the Company's imaging system will work in the manner expected. Except as required under applicable securities legislation, the Company undertakes no obligation to publicly update or revise forward-looking information. The CSE has neither approved nor disapproved the information contained herein and
does not accept responsibility for the adequacy or accuracy of this news release.
Contacts:
Stephen Kilmer,
Investor Relations
Telephone: 647-872-4849
Email: stephen@kilmerlucas.com
Jim Hutchens,
President & CEO
Telephone: 833-246-2446
SOURCE: Imagin Medical
View source version on accesswire.com:
https://www.accesswire.com/584491/Imagin-Medical-Receives-Ap…
Imagin Medical to begin testing of i/Blue Imaging System on Anatomical Bladder Models
New Video, Podcast Episode Available on Imagin Medical's Website
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / July 15, 2020 / Imagin Medical (CSE:IME)(OTCQB:IMEXF)(FRA PD2) ("Imagin" or the "Company") today announced that it will begin testing the i/BlueTM Imaging System using anatomical bladder models to confirm simultaneous white and blue light image display.
PD2) ("Imagin" or the "Company") today announced that it will begin testing the i/BlueTM Imaging System using anatomical bladder models to confirm simultaneous white and blue light image display.
The anatomical bladder models simulate pathologic defects representing bladder cancer tumors that have absorbed an imaging agent which fluoresces when exposed to blue light. Imagin will use these bladder models to 1) conduct extensive optical and image benchtop testing; 2) verify the i/Blue System's production of separate white and blue light images; and 3) conduct virtual demonstrations for evaluation by leading urologists.
"This testing brings us closer to conducting our first live subject studies later this year while we continue to introduce i/Blue's capabilities to the medical community," said Jim Hutchens, Imagin's President and CEO.
The Company also announced that it has posted on its website an animated short that illustrates the differences between the current visualization technology and the i/Blue System. Another addition is a "StockTalk" podcast that Stockhouse Media recently published on its website.
Mr. Hutchens added, "While today's blue light imaging has been shown to identify over 24% more tumors than white light, the high cost of equipment and the need to toggle or switch between white and blue light images helps explain why its use is limited. Imagin expects that i/Blue will greatly expand adoption of blue light technology due to its additional features and ease-of-use. We encourage investors to view the animated short and listen to the StockTalk podcast in order to gain a better understanding of i/Blue's value proposition, as well as Imagin's plans to bring this game-changing technology to market."
https://imaginmedical.com/wp-content/uploads/2020/07/Imagin-…
https://imaginmedical.com/wp-content/uploads/2020/07/Stockho…
New Video, Podcast Episode Available on Imagin Medical's Website
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / July 15, 2020 / Imagin Medical (CSE:IME)(OTCQB:IMEXF)(FRA
 PD2) ("Imagin" or the "Company") today announced that it will begin testing the i/BlueTM Imaging System using anatomical bladder models to confirm simultaneous white and blue light image display.
PD2) ("Imagin" or the "Company") today announced that it will begin testing the i/BlueTM Imaging System using anatomical bladder models to confirm simultaneous white and blue light image display.The anatomical bladder models simulate pathologic defects representing bladder cancer tumors that have absorbed an imaging agent which fluoresces when exposed to blue light. Imagin will use these bladder models to 1) conduct extensive optical and image benchtop testing; 2) verify the i/Blue System's production of separate white and blue light images; and 3) conduct virtual demonstrations for evaluation by leading urologists.
"This testing brings us closer to conducting our first live subject studies later this year while we continue to introduce i/Blue's capabilities to the medical community," said Jim Hutchens, Imagin's President and CEO.
The Company also announced that it has posted on its website an animated short that illustrates the differences between the current visualization technology and the i/Blue System. Another addition is a "StockTalk" podcast that Stockhouse Media recently published on its website.
Mr. Hutchens added, "While today's blue light imaging has been shown to identify over 24% more tumors than white light, the high cost of equipment and the need to toggle or switch between white and blue light images helps explain why its use is limited. Imagin expects that i/Blue will greatly expand adoption of blue light technology due to its additional features and ease-of-use. We encourage investors to view the animated short and listen to the StockTalk podcast in order to gain a better understanding of i/Blue's value proposition, as well as Imagin's plans to bring this game-changing technology to market."
https://imaginmedical.com/wp-content/uploads/2020/07/Imagin-…
https://imaginmedical.com/wp-content/uploads/2020/07/Stockho…
Antwort auf Beitrag Nr.: 64.418.165 von techinvestor69 am 15.07.20 14:32:03Danke für die guten Neuigkeiten. Wieder ein Stück weiter Richtung Ziel. Sind das jetzt schon Tests an Patienten ? Danke nochmals für das schnelle Einstellen.
Antwort auf Beitrag Nr.: 64.426.496 von florfliege am 15.07.20 22:10:15Imagin Medical beginnt mit der Erprobung des i/Blue-Bildgebungssystems an anatomischen Blasenmodellen
Neues Video, Podcast-Episode verfügbar auf der Website von Imagin Medical VANCOUVER, BC und BOSTON, MA / ACCESSWIRE / 15. Juli 2020 / Imagin Medical (CSE: IME)(OTCQB:IMEXF)(FRA PD2) ("Imagin" oder das "Unternehmen") gab heute bekannt, dass das Unternehmen mit der Erprobung des i/BlueTM Imaging Systems unter Verwendung anatomischer Blasenmodelle beginnen wird, um die gleichzeitige Anzeige von Weiß- und Blaulichtbildern zu bestätigen. Die anatomischen Blasenmodelle simulieren pathologische Defekte, die Blasenkrebstumore darstellen, die ein Bildgebungsmittel absorbiert haben, das bei der Exposition mit blauem Licht fluoresziert. Imagin wird diese Blasenmodelle verwenden, um 1) umfangreiche optische Tests und Bildtests am Prüfstand durchzuführen; 2) die Erzeugung getrennter Weiß- und Blaulichtbilder durch das i/Blue-System zu verifizieren; und 3) virtuelle Demonstrationen zur Auswertung durch führende Urologen durchzuführen. "Diese Tests bringen uns der Durchführung unserer ersten Studien mit lebenden Probanden im Laufe dieses Jahres näher, während wir die Fähigkeiten von i/Blue weiterhin der medizinischen Fachwelt vorstellen", sagte Jim Hutchens, Präsident und CEO von Imagin. Das Unternehmen gab außerdem bekannt, dass es auf seiner Website einen animierten Kurzfilm veröffentlicht hat, der die Unterschiede zwischen der aktuellen Visualisierungstechnologie und dem i/Blue-System veranschaulicht. Eine weitere Ergänzung ist ein "StockTalk"-Podcast, den Stockhouse Media kürzlich auf seiner Website veröffentlicht hat. Herr Hutchens fügte hinzu: "Zwar hat sich gezeigt, dass die heutige Blaulicht-Bildgebung über 24% mehr Tumore identifiziert als weißes Licht, aber die hohen Kosten der Ausrüstung und die Notwendigkeit, zwischen weißen und blauen Lichtbildern hin- und herzuschalten oder zwischen ihnen zu wechseln, erklären, warum ihr Einsatz begrenzt ist. Imagin geht davon aus, dass i/Blue die Akzeptanz der Blaulicht-Technologie aufgrund ihrer zusätzlichen Funktionen und ihrer Benutzerfreundlichkeit stark ausweiten wird. Wir ermutigen Investoren, sich die animierte Kurzfassung anzusehen und den StockTalk-Podcast anzuhören, um ein besseres Verständnis des Wertversprechens von i/Blue sowie der Pläne von Imagin zu erhalten, diese spielverändernde Technologie auf den Markt zu bringen".
PD2) ("Imagin" oder das "Unternehmen") gab heute bekannt, dass das Unternehmen mit der Erprobung des i/BlueTM Imaging Systems unter Verwendung anatomischer Blasenmodelle beginnen wird, um die gleichzeitige Anzeige von Weiß- und Blaulichtbildern zu bestätigen. Die anatomischen Blasenmodelle simulieren pathologische Defekte, die Blasenkrebstumore darstellen, die ein Bildgebungsmittel absorbiert haben, das bei der Exposition mit blauem Licht fluoresziert. Imagin wird diese Blasenmodelle verwenden, um 1) umfangreiche optische Tests und Bildtests am Prüfstand durchzuführen; 2) die Erzeugung getrennter Weiß- und Blaulichtbilder durch das i/Blue-System zu verifizieren; und 3) virtuelle Demonstrationen zur Auswertung durch führende Urologen durchzuführen. "Diese Tests bringen uns der Durchführung unserer ersten Studien mit lebenden Probanden im Laufe dieses Jahres näher, während wir die Fähigkeiten von i/Blue weiterhin der medizinischen Fachwelt vorstellen", sagte Jim Hutchens, Präsident und CEO von Imagin. Das Unternehmen gab außerdem bekannt, dass es auf seiner Website einen animierten Kurzfilm veröffentlicht hat, der die Unterschiede zwischen der aktuellen Visualisierungstechnologie und dem i/Blue-System veranschaulicht. Eine weitere Ergänzung ist ein "StockTalk"-Podcast, den Stockhouse Media kürzlich auf seiner Website veröffentlicht hat. Herr Hutchens fügte hinzu: "Zwar hat sich gezeigt, dass die heutige Blaulicht-Bildgebung über 24% mehr Tumore identifiziert als weißes Licht, aber die hohen Kosten der Ausrüstung und die Notwendigkeit, zwischen weißen und blauen Lichtbildern hin- und herzuschalten oder zwischen ihnen zu wechseln, erklären, warum ihr Einsatz begrenzt ist. Imagin geht davon aus, dass i/Blue die Akzeptanz der Blaulicht-Technologie aufgrund ihrer zusätzlichen Funktionen und ihrer Benutzerfreundlichkeit stark ausweiten wird. Wir ermutigen Investoren, sich die animierte Kurzfassung anzusehen und den StockTalk-Podcast anzuhören, um ein besseres Verständnis des Wertversprechens von i/Blue sowie der Pläne von Imagin zu erhalten, diese spielverändernde Technologie auf den Markt zu bringen".
Übersetzt mit www.DeepL.com/Translator (kostenlose Version)
Neues Video, Podcast-Episode verfügbar auf der Website von Imagin Medical VANCOUVER, BC und BOSTON, MA / ACCESSWIRE / 15. Juli 2020 / Imagin Medical (CSE: IME)(OTCQB:IMEXF)(FRA
 PD2) ("Imagin" oder das "Unternehmen") gab heute bekannt, dass das Unternehmen mit der Erprobung des i/BlueTM Imaging Systems unter Verwendung anatomischer Blasenmodelle beginnen wird, um die gleichzeitige Anzeige von Weiß- und Blaulichtbildern zu bestätigen. Die anatomischen Blasenmodelle simulieren pathologische Defekte, die Blasenkrebstumore darstellen, die ein Bildgebungsmittel absorbiert haben, das bei der Exposition mit blauem Licht fluoresziert. Imagin wird diese Blasenmodelle verwenden, um 1) umfangreiche optische Tests und Bildtests am Prüfstand durchzuführen; 2) die Erzeugung getrennter Weiß- und Blaulichtbilder durch das i/Blue-System zu verifizieren; und 3) virtuelle Demonstrationen zur Auswertung durch führende Urologen durchzuführen. "Diese Tests bringen uns der Durchführung unserer ersten Studien mit lebenden Probanden im Laufe dieses Jahres näher, während wir die Fähigkeiten von i/Blue weiterhin der medizinischen Fachwelt vorstellen", sagte Jim Hutchens, Präsident und CEO von Imagin. Das Unternehmen gab außerdem bekannt, dass es auf seiner Website einen animierten Kurzfilm veröffentlicht hat, der die Unterschiede zwischen der aktuellen Visualisierungstechnologie und dem i/Blue-System veranschaulicht. Eine weitere Ergänzung ist ein "StockTalk"-Podcast, den Stockhouse Media kürzlich auf seiner Website veröffentlicht hat. Herr Hutchens fügte hinzu: "Zwar hat sich gezeigt, dass die heutige Blaulicht-Bildgebung über 24% mehr Tumore identifiziert als weißes Licht, aber die hohen Kosten der Ausrüstung und die Notwendigkeit, zwischen weißen und blauen Lichtbildern hin- und herzuschalten oder zwischen ihnen zu wechseln, erklären, warum ihr Einsatz begrenzt ist. Imagin geht davon aus, dass i/Blue die Akzeptanz der Blaulicht-Technologie aufgrund ihrer zusätzlichen Funktionen und ihrer Benutzerfreundlichkeit stark ausweiten wird. Wir ermutigen Investoren, sich die animierte Kurzfassung anzusehen und den StockTalk-Podcast anzuhören, um ein besseres Verständnis des Wertversprechens von i/Blue sowie der Pläne von Imagin zu erhalten, diese spielverändernde Technologie auf den Markt zu bringen".
PD2) ("Imagin" oder das "Unternehmen") gab heute bekannt, dass das Unternehmen mit der Erprobung des i/BlueTM Imaging Systems unter Verwendung anatomischer Blasenmodelle beginnen wird, um die gleichzeitige Anzeige von Weiß- und Blaulichtbildern zu bestätigen. Die anatomischen Blasenmodelle simulieren pathologische Defekte, die Blasenkrebstumore darstellen, die ein Bildgebungsmittel absorbiert haben, das bei der Exposition mit blauem Licht fluoresziert. Imagin wird diese Blasenmodelle verwenden, um 1) umfangreiche optische Tests und Bildtests am Prüfstand durchzuführen; 2) die Erzeugung getrennter Weiß- und Blaulichtbilder durch das i/Blue-System zu verifizieren; und 3) virtuelle Demonstrationen zur Auswertung durch führende Urologen durchzuführen. "Diese Tests bringen uns der Durchführung unserer ersten Studien mit lebenden Probanden im Laufe dieses Jahres näher, während wir die Fähigkeiten von i/Blue weiterhin der medizinischen Fachwelt vorstellen", sagte Jim Hutchens, Präsident und CEO von Imagin. Das Unternehmen gab außerdem bekannt, dass es auf seiner Website einen animierten Kurzfilm veröffentlicht hat, der die Unterschiede zwischen der aktuellen Visualisierungstechnologie und dem i/Blue-System veranschaulicht. Eine weitere Ergänzung ist ein "StockTalk"-Podcast, den Stockhouse Media kürzlich auf seiner Website veröffentlicht hat. Herr Hutchens fügte hinzu: "Zwar hat sich gezeigt, dass die heutige Blaulicht-Bildgebung über 24% mehr Tumore identifiziert als weißes Licht, aber die hohen Kosten der Ausrüstung und die Notwendigkeit, zwischen weißen und blauen Lichtbildern hin- und herzuschalten oder zwischen ihnen zu wechseln, erklären, warum ihr Einsatz begrenzt ist. Imagin geht davon aus, dass i/Blue die Akzeptanz der Blaulicht-Technologie aufgrund ihrer zusätzlichen Funktionen und ihrer Benutzerfreundlichkeit stark ausweiten wird. Wir ermutigen Investoren, sich die animierte Kurzfassung anzusehen und den StockTalk-Podcast anzuhören, um ein besseres Verständnis des Wertversprechens von i/Blue sowie der Pläne von Imagin zu erhalten, diese spielverändernde Technologie auf den Markt zu bringen".Übersetzt mit www.DeepL.com/Translator (kostenlose Version)
Ausbruch aus der monatelangen Trading-Range nach oben!
Ziel Jahreshoch bei 0,095 CAD

Ziel Jahreshoch bei 0,095 CAD
Antwort auf Beitrag Nr.: 64.890.503 von techinvestor69 am 26.08.20 17:14:27Liegt was in der Luft ? Wird auch Zeit, gucke jeden Tag und warte auf positive Neuigkeiten. Danke für die Info. Gruss florfliege
Antwort auf Beitrag Nr.: 64.893.641 von florfliege am 26.08.20 20:47:59Hi bin auch noch dabei.
Soweit ich weiß arbeiten sie daran eine Demo zu veröffentlichen, die den Einsatz des Gerätes im Betrieb zeigt.
Bin gespannt ob es die Erwartungen erfüllt.
Gruß
Soweit ich weiß arbeiten sie daran eine Demo zu veröffentlichen, die den Einsatz des Gerätes im Betrieb zeigt.
Bin gespannt ob es die Erwartungen erfüllt.
Gruß
Antwort auf Beitrag Nr.: 64.948.743 von f1re am 01.09.20 11:53:59Hallo, die Erwartungen scheint das System anscheinend zu erfüllen.Eine anderes Unternehmen hatte ja den Auftrag das System in ein funktionelles Gerät zu integrieren und die Anforderungen zur Produktsicherheit der Zulassung zu gewährleisten. Es ist auch in klinischer Erprobung, wir sind auf der Zielgeraden. Bilder von dem Gerät, stehen 2 oder 3 Stück nebeneinander, findest du auf der imagin Seite. Ich schreibe im Moment auf dem Handy, nervt ein wenig. Das mit dem Praxisfilm wäre toll. Die fertigen Geräte müssen ja eine Sicherheit bieten für Benutzer und Patienten. IM könnte nur ein bisschen mehr kommunizieren. Ich wünsche uns allen viel Erfolg. Grüsse florfliege
Hallo, IM könnte doch mal einen Zwischenstand der Tests mitteilen. Die Informationsmitteilungen sind ziemlich dürftig. Einen kleinen Zwischenbericht zu geben, wäre ja wohl nicht zu viel verlangt.
Wer wartet auch auf Nachrichten? Grüsse, florfliege
Wer wartet auch auf Nachrichten? Grüsse, florfliege
Imagin Medical to Host Kol Webcast Event on October 29, 2020
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / September 29, 2020 / Imagin Medical (CSE:IME)(OTCQB:IMEXF)(Frankfurt & Stuttgart Symbol: DPD2) ("Imagin" or the "Company") today announced that it will host a virtual investor event with Key Opinion Leaders ("KOLs") to highlight the game-changing potential of the i/Blue™ Imaging System on Thursday, October 29, 2020 at 12:00 pm Eastern Daylight Time.
The webcast event will feature opinion leaders from major academic centers who will share insight into the current state of bladder cancer imaging and the acute need for improved methods to help reduce recurrence and mortality.
"We encourage people to join this informative webcast to learn more about how Imagin is working to address the shortcomings of current bladder cancer imaging technologies, while building a solid product portfolio to support the Company's success," said Jim Hutchens, Imagin's President and CEO.
A live and archived webcast of the KOL event will be available on the Company's website at www.imaginmedical.com under "Events & Presentations."
VANCOUVER, BC and BOSTON, MA / ACCESSWIRE / September 29, 2020 / Imagin Medical (CSE:IME)(OTCQB:IMEXF)(Frankfurt & Stuttgart Symbol: DPD2) ("Imagin" or the "Company") today announced that it will host a virtual investor event with Key Opinion Leaders ("KOLs") to highlight the game-changing potential of the i/Blue™ Imaging System on Thursday, October 29, 2020 at 12:00 pm Eastern Daylight Time.
The webcast event will feature opinion leaders from major academic centers who will share insight into the current state of bladder cancer imaging and the acute need for improved methods to help reduce recurrence and mortality.
"We encourage people to join this informative webcast to learn more about how Imagin is working to address the shortcomings of current bladder cancer imaging technologies, while building a solid product portfolio to support the Company's success," said Jim Hutchens, Imagin's President and CEO.
A live and archived webcast of the KOL event will be available on the Company's website at www.imaginmedical.com under "Events & Presentations."
Ich erhoffe mir das diese Konferenz IME wieder mehr in den Fokus rücken wird. Sorgen mache ich mir eher um die Finanzierung. Cash sollte mittlerweile aufgebraucht sein...
Kommerzialisierung rückt näher
https://money.tmx.com/en/quote/IME:CNX/news/5404120568453944… 


Wichtiger Schritt für den Markteintritt.
Leider gibt es keine zeitlichen Angaben zur Produktfertigstellung...

Die derzeitige Marktkapitalisierung von ca. 5 Mio kanadischen Dollars scheint sehr bescheiden, zumal der Hauptmarkt zunächst die USA sein sollen und das System auf andere Cancerbehandlungssektoren ausweitbar sein wird.
I.d.R. werden solche Innovationen von großen MedTec-Unternehmen weggekauft... mal schaun.

DYOR

Gruß Greenfoxi
Antwort auf Beitrag Nr.: 65.237.338 von f1re am 30.09.20 14:57:08ich denke, hinter den Kulissen läuft ein 0,02 CAD pp
Antwort auf Beitrag Nr.: 65.237.338 von f1re am 30.09.20 14:57:08
https://onlinexperiences.com/scripts/Server.nxp
DYOR
Gruß Greenfoxi
Online
Die Präsi von IME ist jetzt bei Virtual Investor Conferences.com verfügbar...https://onlinexperiences.com/scripts/Server.nxp

DYOR

Gruß Greenfoxi
Hallo, Seite geht nicht auf, danke
Antwort auf Beitrag Nr.: 65.366.598 von florfliege am 13.10.20 09:56:03
versuchs mal über die Homepage von IME.
Du musst Dich allerdings erst richtig anmelden...

Gruß GF
Procedere
Hi Fliege,versuchs mal über die Homepage von IME.
Du musst Dich allerdings erst richtig anmelden...


Gruß GF
Hallo Greenfoxi, meinst Du das Zeichentrickfilmchen?? Das steht aber schon länger auf der Website von IME. Die Verpflichtung von Lighthouse sehe ich als gut an, denn anscheinend haben die klinischen Tests (angefangen Mitte Juli) gezeigt, daß das System funktioniert. Was nicht so funktioniert sind die langen Mitteilungspausen von IME. Die könnten schon ein wenig mehr mitteilen und auch öfters. Ich denke wir sind im Endspurt, hoffentlich...Dank und Grüsse florfliege
Es bewegt sich was bei IME. Ich bin gespannt auf die conference Ende des Monats.
Gruß
Gruß
Antwort auf Beitrag Nr.: 65.428.458 von f1re am 19.10.20 12:19:50Nicht nur Du 😀
Aufgepasst:
es wird eine Finanzierung zu umgerechnet 0,026 CAD durchgeführt (Wandelschuldverschreibung 3 Mio. USD)
ABER es wird kurzfristig - mit dem Closing einer ersten 750.000 USD Tranche (heute nach Börsenschluss?) eine Aktienzusammenlegung im Verhältnis 20:1 durchgeführt
d.h. für 20 alte Aktien gibt es eine neue / also reduziert sich unser Aktienbestand / Im Gegenzug wird der Aktienkurs theoretisch von 0,025 CAD auf 0,50 CAD steigen
Link zur News: https://www.stockwatch.com/News/Item?bid=Z-C:IME-2978289&sym…
3 Mio. + das Geld aus den warrants sollte genug sein, um das Produkt auf den Markt zu bringen
Verwässerung ist 💩, aber war unvermeidbar. Lieber so als pleite.
es wird eine Finanzierung zu umgerechnet 0,026 CAD durchgeführt (Wandelschuldverschreibung 3 Mio. USD)
ABER es wird kurzfristig - mit dem Closing einer ersten 750.000 USD Tranche (heute nach Börsenschluss?) eine Aktienzusammenlegung im Verhältnis 20:1 durchgeführt
d.h. für 20 alte Aktien gibt es eine neue / also reduziert sich unser Aktienbestand / Im Gegenzug wird der Aktienkurs theoretisch von 0,025 CAD auf 0,50 CAD steigen
Link zur News: https://www.stockwatch.com/News/Item?bid=Z-C:IME-2978289&sym…
3 Mio. + das Geld aus den warrants sollte genug sein, um das Produkt auf den Markt zu bringen
Verwässerung ist 💩, aber war unvermeidbar. Lieber so als pleite.
Antwort auf Beitrag Nr.: 65.472.834 von techinvestor69 am 23.10.20 09:12:29Danke für das Update im deutschen Forum. Ich habe gestern nach erscheinen der News sogar nochmals nachgekauft um meinen EK etwas runter zu bekommen.
Ob das die richtige Entscheidung war wird sich in den nächsten Monaten zeigen. Was am Ende zählt ist die MarketCap und wenn die es tatsächlich schaffen die Richtung 100Mio oder mehr zu bringen dann könnte das eine Goldgrube werden. Aber bis dahin kann leider alles passieren...
Ob das die richtige Entscheidung war wird sich in den nächsten Monaten zeigen. Was am Ende zählt ist die MarketCap und wenn die es tatsächlich schaffen die Richtung 100Mio oder mehr zu bringen dann könnte das eine Goldgrube werden. Aber bis dahin kann leider alles passieren...
Re-split 1 zu 20 abgeschlossen / KOL VIRTUAL INVESTOR Event / Kohle für Produkteinführung
https://money.tmx.com/en/quote/IME:CNX/news/6514269646232962… 
https://money.tmx.com/en/quote/IME:CNX/news/7954608530944320…

https://money.tmx.com/en/quote/IME:CNX/news/7037284040593653…

DYOR

Gruß Greenfoxi
Wandelschuldverschreibungen werden im Insolvenzfall wohl anders behandelt als Aktien, ein
Zugriff auf die Kunkursmasse scheint hier möglich zu sein. Ein ausgesprochen schwieriges
Thema von dem ich keinerlei Ahnung habe.
Hatte aber mal eine Aktie für 20 US Cent bei der die Firma unter Chapter 11 gestellt wurde.
Irgendwann war der Schein dann unter gleicher WKN mit 55 Dollar eingebucht. Versilbern
konnte ich das aber nicht, da es sich nach Angaben der Bank um den übriggebliebenen Rest
der Aktien, nämlich Wandelschuldverschreibungen handelte.
Vielleicht weiß jemand mehr darüber als ich?
Zugriff auf die Kunkursmasse scheint hier möglich zu sein. Ein ausgesprochen schwieriges
Thema von dem ich keinerlei Ahnung habe.
Hatte aber mal eine Aktie für 20 US Cent bei der die Firma unter Chapter 11 gestellt wurde.
Irgendwann war der Schein dann unter gleicher WKN mit 55 Dollar eingebucht. Versilbern
konnte ich das aber nicht, da es sich nach Angaben der Bank um den übriggebliebenen Rest
der Aktien, nämlich Wandelschuldverschreibungen handelte.
Vielleicht weiß jemand mehr darüber als ich?
Beitrag zu dieser Diskussion schreiben
Zu dieser Diskussion können keine Beiträge mehr verfasst werden, da der letzte Beitrag vor mehr als zwei Jahren verfasst wurde und die Diskussion daraufhin archiviert wurde.
Bitte wenden Sie sich an feedback@wallstreet-online.de und erfragen Sie die Reaktivierung der Diskussion oder starten Sie eine neue Diskussion.
Investoren beobachten auch:
| Wertpapier | Perf. % |
|---|---|
| +0,08 | |
| -14,29 | |
| 0,00 | |
| +900,00 | |
| -1,85 | |
| -1,21 | |
| 0,00 | |
| +0,51 | |
| -2,96 | |
| 0,00 |
Meistdiskutiert
| Wertpapier | Beiträge | |
|---|---|---|
| 137 | ||
| 56 | ||
| 37 | ||
| 24 | ||
| 23 | ||
| 20 | ||
| 20 | ||
| 16 | ||
| 13 | ||
| 13 |



















