Amarin - The Science Of Lipid Therapy (Seite 107)
eröffnet am 03.01.14 20:10:32 von
neuester Beitrag 04.04.24 15:47:54 von
neuester Beitrag 04.04.24 15:47:54 von
Beiträge: 1.840
ID: 1.190.027
ID: 1.190.027
Aufrufe heute: 0
Gesamt: 156.391
Gesamt: 156.391
Aktive User: 0
ISIN: US0231112063 · WKN: A0NBNG · Symbol: EH3A
0,9100
USD
-0,87 %
-0,0080 USD
Letzter Kurs 04.05.24 Nasdaq
Neuigkeiten
01.05.24 · globenewswire |
24.04.24 · globenewswire |
22.04.24 · globenewswire |
15.04.24 · globenewswire |
Amarin Highlights Key Data Providing Mechanistic Insights into Eicosapentaenoic Acid (EPA) at ACC.24 08.04.24 · globenewswire |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,2000 | +471,16 | |
| 13,110 | +38,44 | |
| 6,5000 | +27,45 | |
| 1,2100 | +21,00 | |
| 48,25 | +19,94 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 6,2600 | -14,25 | |
| 3,8500 | -14,45 | |
| 1,0011 | -15,88 | |
| 36,70 | -22,87 | |
| 2,5600 | -70,32 |
Beitrag zu dieser Diskussion schreiben
Hier CaptBeer auf stockwits, wie ich finde mit beeindruckenden Statements bzgl. möglicher Patientenanzahl für die neue VASCEPA Indikation:
Good Morning Friends! I've been trying to figure out the potential patient population for the new VASCEPA Indication.
The language is very broad and flexible allowing doctors plenty of interpretative license. I'll be presenting statistics from various sources (referenced in the illustrations).
PART # 1: The Indication as "Adjunct to Statin Therapy" :
Here's the Indication: “as an adjunct to maximally tolerated statin therapy to reduce the risk of myocardial infarction, stroke, coronary revascularization, and unstable angina requiring hospitalization in adult patients with elevated triglyceride (TG) levels (≥150 mg/dL) and established cardiovascular disease or diabetes mellitus and two or more additional risk factors for cardiovascular disease.”
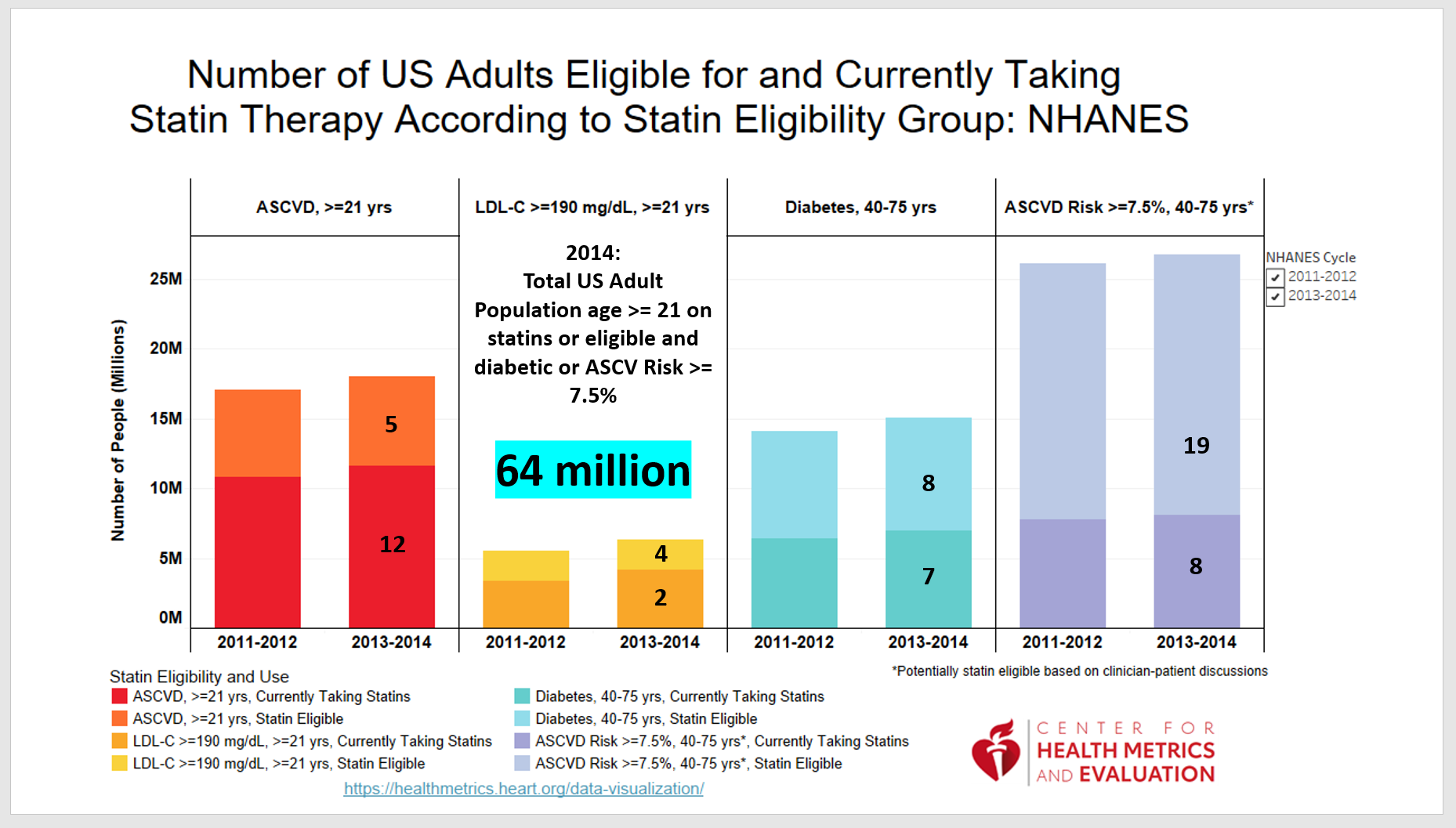
In 2014 there were approximately 64 million adult patients on statins or eligible for the use.
This chart groups those patients as illustrated:
Good Morning Friends! I've been trying to figure out the potential patient population for the new VASCEPA Indication.
The language is very broad and flexible allowing doctors plenty of interpretative license. I'll be presenting statistics from various sources (referenced in the illustrations).
PART # 1: The Indication as "Adjunct to Statin Therapy" :
Here's the Indication: “as an adjunct to maximally tolerated statin therapy to reduce the risk of myocardial infarction, stroke, coronary revascularization, and unstable angina requiring hospitalization in adult patients with elevated triglyceride (TG) levels (≥150 mg/dL) and established cardiovascular disease or diabetes mellitus and two or more additional risk factors for cardiovascular disease.”
In 2014 there were approximately 64 million adult patients on statins or eligible for the use.
This chart groups those patients as illustrated:

Antwort auf Beitrag Nr.: 62.159.984 von Magnetfeldfredy am 14.12.19 16:27:41..und auch ein Artikel im Wallstreet-Journal
FDA Approves Fish-Oil-Derived Drug for Use Preventing Heart Attacks, Strokes
Vascepa becomes new agent to help millions of heart-disease patients who have high cholesterol despite taking medicine for it
By Jared S. Hopkins
Dec. 13, 2019 5:48 pm ET
The U.S. Food and Drug Administration on Friday expanded the approved use of a fish-oil-derived drug to reduce the likelihood of heart attacks and strokes in high-risk patients.
The drug, Vascepa from Amarin Corp. PLC, now becomes a new tool for reducing the risk of heart attacks, strokes and deaths in millions of heart-disease or diabetes patients with elevated triglycerides while opening up a multibillion-dollar commercial opportunity for its maker. The expanded label could mean Vascepa sales surpass $3 billion, analysts say. Last year’s sales approached $230 million.
analysts say. Last year’s sales approached $230 million.
Vascepa was approved in the U.S. in 2012 to treat adults with severe hypertriglyceridemia, or very high levels of triglycerides, which are fats that circulate in the blood k of heart disease.
High triglyceride levels can signal the presence of metabolic abnormalities that can damage the heart and blood vessels. About four million Americans have severe hypertriglyceridemia, according to Amarin, and 70 million have elevated triglyceride levels.
The FDA said Vascepa could be taken on top of cholesterol drugs known as statins. Patients must have either heart disease, or diabetes and at least two additional risk factors for cardiovascular disease.
“For the first time, physicians, patients and payers have an FDA-approved treatment option beyond cholesterol lowering that has been demonstrated to significantly reduce major adverse cardiovascular events when used on top of a statin,” Amarin Chief Executive John Thero said.
A large late-stage study, commissioned by Amarin, showed that Vascepa can reduce the risk of cardiovascular-disease events in patients with elevated triglyceride levels who are at high risk and on cholesterol-lowering drugs.
Most patients in the study had already had a heart attack, stroke or other cardiovascular event. The rest were at high risk: They had Type 2 diabetes and at least one other risk factor, such as tobacco use.
The FDA acted earlier than the expected Dec. 28 date, the latest in a series of recent earlier-than-expected decisions from the FDA.
In November, a panel of outside advisers voted 16 to 0 in recommending the agency enlarge the approved use of the drug. The FDA wasn’t bound to follow the advice of its advisory committee, but it normally does.
The FDA rejected Amarin’s first request for a broader label in 2013, prompting the company to sponsor the latest study. In 2018, Amarin announced data from that study, which showed high-risk patients who took Vascepa in addition to a statin experienced a 25% reduction in risk of a heart attack, stroke or other serious cardiac event, compared with patients who took a placebo.
The company’s shares surged on the news in 2018. The shares closed up 4.9% at $24.12 after Friday’s announcement. Amarin is based in Dublin, Ireland, but operates out of Bridgewater, N.J.
A monthly supply of the gram dose of Vascepa carries a list price of $303.65 before insurance kicks in.
https://www.wsj.com/articles/fda-approves-fish-oil-derived-d…
FDA Approves Fish-Oil-Derived Drug for Use Preventing Heart Attacks, Strokes
Vascepa becomes new agent to help millions of heart-disease patients who have high cholesterol despite taking medicine for it
By Jared S. Hopkins
Dec. 13, 2019 5:48 pm ET
The U.S. Food and Drug Administration on Friday expanded the approved use of a fish-oil-derived drug to reduce the likelihood of heart attacks and strokes in high-risk patients.
The drug, Vascepa from Amarin Corp. PLC, now becomes a new tool for reducing the risk of heart attacks, strokes and deaths in millions of heart-disease or diabetes patients with elevated triglycerides while opening up a multibillion-dollar commercial opportunity for its maker. The expanded label could mean Vascepa sales surpass $3 billion,
 analysts say. Last year’s sales approached $230 million.
analysts say. Last year’s sales approached $230 million.Vascepa was approved in the U.S. in 2012 to treat adults with severe hypertriglyceridemia, or very high levels of triglycerides, which are fats that circulate in the blood k of heart disease.
High triglyceride levels can signal the presence of metabolic abnormalities that can damage the heart and blood vessels. About four million Americans have severe hypertriglyceridemia, according to Amarin, and 70 million have elevated triglyceride levels.
The FDA said Vascepa could be taken on top of cholesterol drugs known as statins. Patients must have either heart disease, or diabetes and at least two additional risk factors for cardiovascular disease.
“For the first time, physicians, patients and payers have an FDA-approved treatment option beyond cholesterol lowering that has been demonstrated to significantly reduce major adverse cardiovascular events when used on top of a statin,” Amarin Chief Executive John Thero said.
A large late-stage study, commissioned by Amarin, showed that Vascepa can reduce the risk of cardiovascular-disease events in patients with elevated triglyceride levels who are at high risk and on cholesterol-lowering drugs.
Most patients in the study had already had a heart attack, stroke or other cardiovascular event. The rest were at high risk: They had Type 2 diabetes and at least one other risk factor, such as tobacco use.
The FDA acted earlier than the expected Dec. 28 date, the latest in a series of recent earlier-than-expected decisions from the FDA.
In November, a panel of outside advisers voted 16 to 0 in recommending the agency enlarge the approved use of the drug. The FDA wasn’t bound to follow the advice of its advisory committee, but it normally does.
The FDA rejected Amarin’s first request for a broader label in 2013, prompting the company to sponsor the latest study. In 2018, Amarin announced data from that study, which showed high-risk patients who took Vascepa in addition to a statin experienced a 25% reduction in risk of a heart attack, stroke or other serious cardiac event, compared with patients who took a placebo.
The company’s shares surged on the news in 2018. The shares closed up 4.9% at $24.12 after Friday’s announcement. Amarin is based in Dublin, Ireland, but operates out of Bridgewater, N.J.
A monthly supply of the gram dose of Vascepa carries a list price of $303.65 before insurance kicks in.
https://www.wsj.com/articles/fda-approves-fish-oil-derived-d…
Sehr guter Artikel:
Amarin eyes blockbuster Vascepa sales as FDA grants heart-helping label expansion
by Kyle Blankenship | Dec 13, 2019 4:45pm
Amarin Vascepa pill
The Vascepa label expansion will likely spur even more buyout speculation for Amarin. (Amarin)
Teilen
Facebook
Twitter
LinkedIn
Email
Print
Amarin has been on a scorching run with its fish-oil derivative Vascepa after a major cardiovascular outcomes trial last year set a possible blockbuster trajectory for the drug. Now, the FDA has voted to give Vascepa a major label boost with the addition of CV risk reduction––and those blockbuster sales could be on the near horizon.
The FDA approved Vascepa as an add-on to statins to reduce the risk of cardiovascular events in patients with elevated triglycerides who have either established CV disease or diabetes with two additional CV risk factors, the administration said Friday.
The indication settles a lingering question around Vascepa's expanded label of whether the FDA would limit the drug's patient pool to only those with a preexisting CV disease or expand it to the much larger population of patients at high risk of disease.
With a broader label in hand, Vascepa is now poised to become the blockbuster drug Amarin hoped it would become after a major outcomes trial last year showed the drug on top of statins cut the risk of CV events by 25% in patients with abnormally high triglyceride levels.
RELATED: Blockbuster in sight? Amarin's Vascepa sails through FDA panel vote toward new CV nod
Amarin raised its 2020 sales forecast to between $600 million and $700 million as it plans to roll out an expanded sales team of 800 by mid-January, CEO John Thero said Friday. With the new label, Thero said Amarin was expecting peak sales estimates in the "multiple billions."
"We are creating a market here and going where people have not gone before," he said. "Our focus is on improving patient care and executing on our plan."
Vascepa has now been approved for a possible patient population that runs into the tens of millions, Thero said.
"This is an indication that gives physicians tools to treat their high-risk patients, whether those are patients with an established CV disease or diabetes with other CV risk factors," he said. "This is a lot of patients."
The FDA's approval follows an advisory committee's unanimous vote in November to recommend the drug's expanded label. In that review, committee members were tasked with investigating concerns that the control arm of Vascepa's outcomes trial, dubbed Reduce-It, may have had a skewing effect on the drug's results.
The trial tested Vascepa as an add-on to statin therapy, compared with the mineral oil placebo paired with statins. The FDA examined whether that mineral oil interfered with patients' ability to absorb statin therapy and artificially raised patients' cholesterol levels, but it called the study "inconclusive," John Sharretts, acting deputy director of the FDA's division of metabolism and endocrinology products, said in a briefing at the time.
RELATED: Amarin for $20B? Novartis' MedCo deal drives fresh buyout rumors for Vascepa's maker
With Vascepa now a likely blockbuster candidate, already sky-high speculation about Amarin's acquisition by a major player will likely continue apace.
Following the committee vote and Novartis' $9.7 billion buyout of The Medicines Company and its PCSK9 cholesterol buster candidate, inclisiran, rumors––however fanciful––of a possible $20 billion Amarin acquisition made the rounds among investors.
And Vascepa's label expansion made Amarin "an even more interesting asset in a consolidating space," Cantor analyst Louise Chen said in a Friday note to investors.
In the meantime, Amarin has expressed its intention to go it alone without a major partner.
In July, Thero told investors Amarin was prepared to take on the increased demand for Vascepa with a sales hiring goal of 800 employees and a planned direct-to-consumer advertising campaign upon expansion. The drugmaker said the company expects to reach 70,000 to 80,000 healthcare professionals when its sales force is fully fleshed out and is seeing higher-than-expected access to doctors in the meantime.
Read more on
cardiovascular outcomes cardiovascular disease fish oil drug labels Amarin Vascepa
Amarin eyes blockbuster Vascepa sales as FDA grants heart-helping label expansion
by Kyle Blankenship | Dec 13, 2019 4:45pm
Amarin Vascepa pill
The Vascepa label expansion will likely spur even more buyout speculation for Amarin. (Amarin)
Teilen
Amarin has been on a scorching run with its fish-oil derivative Vascepa after a major cardiovascular outcomes trial last year set a possible blockbuster trajectory for the drug. Now, the FDA has voted to give Vascepa a major label boost with the addition of CV risk reduction––and those blockbuster sales could be on the near horizon.
The FDA approved Vascepa as an add-on to statins to reduce the risk of cardiovascular events in patients with elevated triglycerides who have either established CV disease or diabetes with two additional CV risk factors, the administration said Friday.
The indication settles a lingering question around Vascepa's expanded label of whether the FDA would limit the drug's patient pool to only those with a preexisting CV disease or expand it to the much larger population of patients at high risk of disease.
With a broader label in hand, Vascepa is now poised to become the blockbuster drug Amarin hoped it would become after a major outcomes trial last year showed the drug on top of statins cut the risk of CV events by 25% in patients with abnormally high triglyceride levels.
RELATED: Blockbuster in sight? Amarin's Vascepa sails through FDA panel vote toward new CV nod
Amarin raised its 2020 sales forecast to between $600 million and $700 million as it plans to roll out an expanded sales team of 800 by mid-January, CEO John Thero said Friday. With the new label, Thero said Amarin was expecting peak sales estimates in the "multiple billions."
"We are creating a market here and going where people have not gone before," he said. "Our focus is on improving patient care and executing on our plan."
Vascepa has now been approved for a possible patient population that runs into the tens of millions, Thero said.
"This is an indication that gives physicians tools to treat their high-risk patients, whether those are patients with an established CV disease or diabetes with other CV risk factors," he said. "This is a lot of patients."
The FDA's approval follows an advisory committee's unanimous vote in November to recommend the drug's expanded label. In that review, committee members were tasked with investigating concerns that the control arm of Vascepa's outcomes trial, dubbed Reduce-It, may have had a skewing effect on the drug's results.
The trial tested Vascepa as an add-on to statin therapy, compared with the mineral oil placebo paired with statins. The FDA examined whether that mineral oil interfered with patients' ability to absorb statin therapy and artificially raised patients' cholesterol levels, but it called the study "inconclusive," John Sharretts, acting deputy director of the FDA's division of metabolism and endocrinology products, said in a briefing at the time.
RELATED: Amarin for $20B? Novartis' MedCo deal drives fresh buyout rumors for Vascepa's maker
With Vascepa now a likely blockbuster candidate, already sky-high speculation about Amarin's acquisition by a major player will likely continue apace.
Following the committee vote and Novartis' $9.7 billion buyout of The Medicines Company and its PCSK9 cholesterol buster candidate, inclisiran, rumors––however fanciful––of a possible $20 billion Amarin acquisition made the rounds among investors.

And Vascepa's label expansion made Amarin "an even more interesting asset in a consolidating space," Cantor analyst Louise Chen said in a Friday note to investors.
In the meantime, Amarin has expressed its intention to go it alone without a major partner.
In July, Thero told investors Amarin was prepared to take on the increased demand for Vascepa with a sales hiring goal of 800 employees and a planned direct-to-consumer advertising campaign upon expansion. The drugmaker said the company expects to reach 70,000 to 80,000 healthcare professionals when its sales force is fully fleshed out and is seeing higher-than-expected access to doctors in the meantime.
Read more on
cardiovascular outcomes cardiovascular disease fish oil drug labels Amarin Vascepa
Sehe ich auch so Hexe, geil, jetzt kommt noch die Kanada Zulassung und es kann jederzeit ein Buyout kommen, was glaubst Du?
Antwort auf Beitrag Nr.: 62.159.558 von Cyberhexe am 14.12.19 14:36:21kleine Korrektur:
Vascepa wird für Patienten zugelassen, bei denen bereits eine Herz-Kreislauf-Erkrankung festgestellt wurde (Herzinfarkt, Angina pectoris ...) oder für diejenigen, die Diabetes und zwei Risikofaktoren für Herzinfarkte haben. Vascepa ist nun für Patienten zugelassen, deren Triglyceridgehalt im Blut 150 Milligramm pro Deziliter übersteigt - Amarin hatte auf einen Minimalwert von 135 mg/dL gehofft.
Vascepa wird für Patienten zugelassen, bei denen bereits eine Herz-Kreislauf-Erkrankung festgestellt wurde (Herzinfarkt, Angina pectoris ...) oder für diejenigen, die Diabetes und zwei Risikofaktoren für Herzinfarkte haben. Vascepa ist nun für Patienten zugelassen, deren Triglyceridgehalt im Blut 150 Milligramm pro Deziliter übersteigt - Amarin hatte auf einen Minimalwert von 135 mg/dL gehofft.
Antwort auf Beitrag Nr.: 62.157.644 von asthmamoah am 14.12.19 06:25:06Vascepa wird für Patienten zugelassen, bei denen bereits eine Herz-Kreislauf-Erkrankung festgestellt wurde (Herzinfarkt, Angina pectoris ...) oder für diejenigen, die Diabetes und zwei Risikofaktoren für Herzinfarkte haben. Vascepa ist für Patienten zugelassen, deren Triglycerid im Blut haben 150 Milligramm pro Deziliter übersteigt - Amarin hatte auf eine Schwelle oberhalb 135 mg/dL gehofft.
"Die FDA erkennt an, dass es einen Bedarf an zusätzlichen medizinischen Behandlungen für Herz-Kreislauf-Erkrankungen gibt", sagte Dr. John Sharretts, stellvertretender Direktor der Division of Metabolism and Endocrinology Products im Center for Drug Evaluation and Research der FDA, in einer Erklärung. "Die heutige Zulassung wird Patienten mit erhöhten Triglyceriden und anderen wichtigen Risikofaktoren, einschließlich Herzerkrankungen, Schlaganfall und Diabetes, eine ergänzende Behandlungsmöglichkeit bieten, die dazu beitragen kann, das Risiko von Herz-Kreislauf-Erkrankungen zu verringern."
Trotz dieser Einschränkungen könnte die Verwendung von Vascepa dramatisch zunehmen. Vor der Zulassung haben die Ergebnisse einer im vergangenen November veröffentlichten wissenschaftlichen Studie dazu geführt, dass sich der Umsatz von Amarin im dritten Quartal dieses Jahres gegenüber dem Vorjahr auf 112 Millionen US-Dollar verdoppelt hat. Ich rechne mit einer weiteren signifikanten Umsatzsteigerung. Vascepa wird relativ schnell ein Blockbuster werden, also > 1 Milliarde$ Jahresumsatz generieren.
"Die FDA erkennt an, dass es einen Bedarf an zusätzlichen medizinischen Behandlungen für Herz-Kreislauf-Erkrankungen gibt", sagte Dr. John Sharretts, stellvertretender Direktor der Division of Metabolism and Endocrinology Products im Center for Drug Evaluation and Research der FDA, in einer Erklärung. "Die heutige Zulassung wird Patienten mit erhöhten Triglyceriden und anderen wichtigen Risikofaktoren, einschließlich Herzerkrankungen, Schlaganfall und Diabetes, eine ergänzende Behandlungsmöglichkeit bieten, die dazu beitragen kann, das Risiko von Herz-Kreislauf-Erkrankungen zu verringern."
Trotz dieser Einschränkungen könnte die Verwendung von Vascepa dramatisch zunehmen. Vor der Zulassung haben die Ergebnisse einer im vergangenen November veröffentlichten wissenschaftlichen Studie dazu geführt, dass sich der Umsatz von Amarin im dritten Quartal dieses Jahres gegenüber dem Vorjahr auf 112 Millionen US-Dollar verdoppelt hat. Ich rechne mit einer weiteren signifikanten Umsatzsteigerung. Vascepa wird relativ schnell ein Blockbuster werden, also > 1 Milliarde$ Jahresumsatz generieren.
Closing Nasdaq nachbörslich 26,70 USD 👍😃
Nur so ein Gefühl zu später Stunde aber ich denke der nächste Squeeze steht am Montag an. Ich sollte dafür aber zur gebührenden Begleitung dieses Ereignisses mal auf was anderes umsteigen, mir ist schon ganz schlecht vom ganzen Popcorn *grins*, hat irgendjemand konstruktive Vorschläge 🙃 ?
Antwort auf Beitrag Nr.: 62.157.077 von bernie55 am 13.12.19 22:33:56
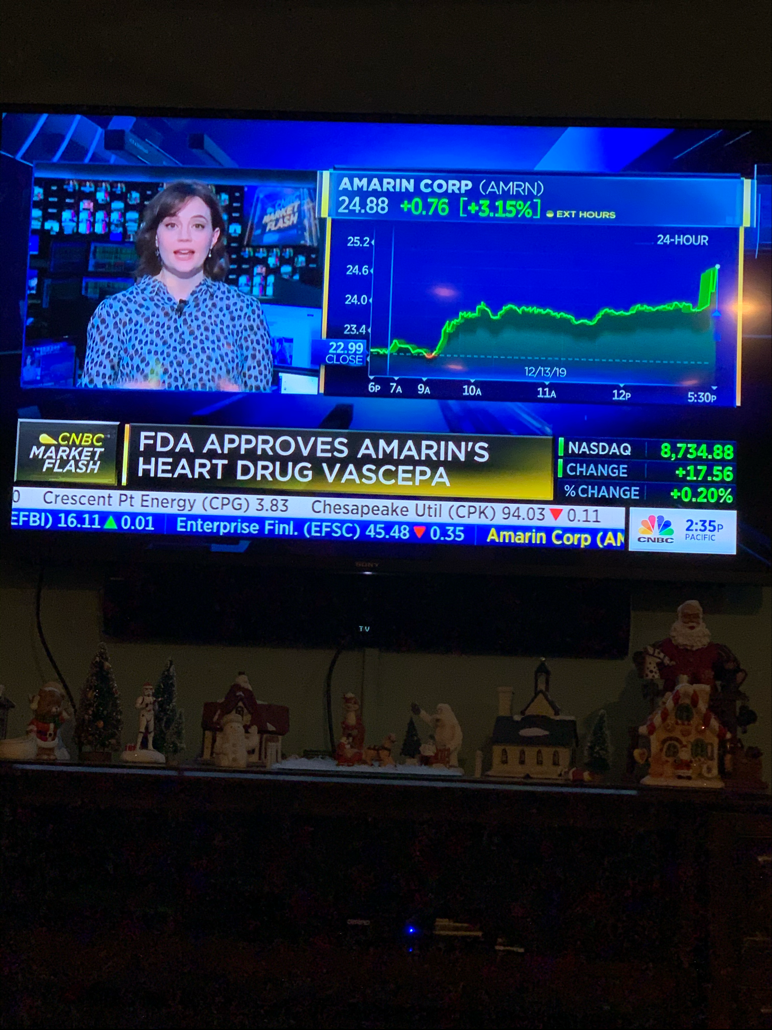
Zitat von bernie55: FDA approves use of drug to reduce risk of cardiovascular events in certain adult patient groups
https://www.fda.gov/news-events/press-announcements/fda-appr…

Antwort auf Beitrag Nr.: 62.157.335 von bernie55 am 13.12.19 23:26:10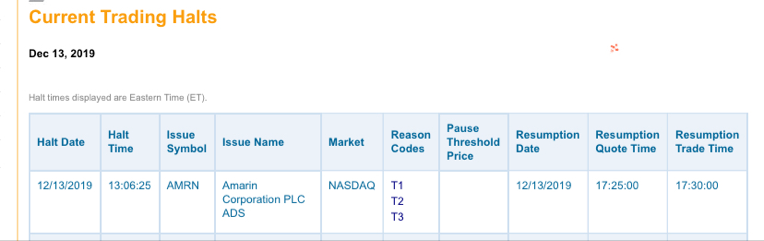

Amarin - The Science Of Lipid Therapy















