Opko Health - Der nächste Blockbuster (Seite 298)
eröffnet am 10.12.13 19:42:20 von
neuester Beitrag 07.05.24 22:55:13 von
neuester Beitrag 07.05.24 22:55:13 von
Beiträge: 3.051
ID: 1.189.336
ID: 1.189.336
Aufrufe heute: 5
Gesamt: 261.286
Gesamt: 261.286
Aktive User: 0
ISIN: US68375N1037 · WKN: A0MUUJ · Symbol: XCY
1,2500
USD
-3,85 %
-0,0500 USD
Letzter Kurs 02:00:00 Nasdaq
Neuigkeiten
07.05.24 · globenewswire |
03.05.24 · globenewswire |
05.03.24 · globenewswire |
27.02.24 · globenewswire |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,8450 | +146,00 | |
| 0,5700 | +55,23 | |
| 0,7200 | +47,03 | |
| 5,4500 | +41,56 | |
| 1,0000 | +33,33 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 13,800 | -13,86 | |
| 4,1900 | -14,49 | |
| 0,9235 | -16,88 | |
| 5,2500 | -19,23 | |
| 2,5600 | -70,32 |
Beitrag zu dieser Diskussion schreiben
www.baus.org.uk/_userfiles/pages/files/AGM/BAUS2018-Abstracts.pdf
Journal of Clinical Urology Formerly British Journal of Medical and Surgical Urology
P6-6 The 4Kscore predicts adverse pathology at radical prostatectomy in men diagnosed at biopsy with gleason 6
Galvin D3, Haese A1, Hou D2, Eggener S2
1University Clinic Eppendorf, Martini-Klinik Prostate Cancer Centre,
Hamburg, Germany, 2University of Chicago, Department of Surgery,
Chicago, United States of America, 3Mater and St Vincent’s Hospitals, Dublin, Ireland
Introduction: Men at biopsy (Bx) with Gleason 6 prostate cancer (PCa) would benefit from better understanding their risk for adverse pathology (Gleason score t7 or Gleason 6 with tpT3a). Our study objective was to determine if a preoperative 4Kscore from men with Gleason 6 could predict adverse pathology at radical prostatectomy (RP). Methods: Serum samples from 177 men with Bx-detected Gleason 6 PCa who underwent RP at Martini Klinik (MK) from 2003 to 2013 were obtained from the MK biorepository and the 4Kscore calculated. Bx and RP
pathology were read by MK. Primary outcome was adverse RP pathology. 4Kscore and other clinical and pathologic characteristics were evaluated for their association with adverse RP pathology.
Results: 4Kscore, prostate volume, % positive cores, and total tumor length were significantly associated with adverse RP pathology (p d0.002). Total PSA, %fPSA ratio, DRE status and biopsy T stage were not associated (pt0.2). Age and no previous biopsy trended towards significance
(p=0.08 and 0.07, respectively). On multivariate modelling, 4Kscore (OR=1.75 per 20% increase, 95% CI 1.19-2.57, p=0.004) and prostate volume (OR=0.67 per 20cc of volume, 95% CI 0.54-0.95, p=0.020) were independently associated. Comparing a 4Kscore cut off of 7.5%
with other risk indices (NCCN, D’Amico and CAPRA) demonstrated 4Kscore had 80% sensitivity, 40% specificity, 63% PPV, and 72% NPV. 4Kscore t7.5% did not miss detection of Gleason 8 PCa and missed 7% (1 of 14) pT3a/b.
Conclusions: The 4Kscore was significantly associatedwith adverse pathology at radical prostatectomy in men with Bx-proven Gleason 6 PCa
Journal of Clinical Urology Formerly British Journal of Medical and Surgical Urology
P6-6 The 4Kscore predicts adverse pathology at radical prostatectomy in men diagnosed at biopsy with gleason 6
Galvin D3, Haese A1, Hou D2, Eggener S2
1University Clinic Eppendorf, Martini-Klinik Prostate Cancer Centre,
Hamburg, Germany, 2University of Chicago, Department of Surgery,
Chicago, United States of America, 3Mater and St Vincent’s Hospitals, Dublin, Ireland
Introduction: Men at biopsy (Bx) with Gleason 6 prostate cancer (PCa) would benefit from better understanding their risk for adverse pathology (Gleason score t7 or Gleason 6 with tpT3a). Our study objective was to determine if a preoperative 4Kscore from men with Gleason 6 could predict adverse pathology at radical prostatectomy (RP). Methods: Serum samples from 177 men with Bx-detected Gleason 6 PCa who underwent RP at Martini Klinik (MK) from 2003 to 2013 were obtained from the MK biorepository and the 4Kscore calculated. Bx and RP
pathology were read by MK. Primary outcome was adverse RP pathology. 4Kscore and other clinical and pathologic characteristics were evaluated for their association with adverse RP pathology.
Results: 4Kscore, prostate volume, % positive cores, and total tumor length were significantly associated with adverse RP pathology (p d0.002). Total PSA, %fPSA ratio, DRE status and biopsy T stage were not associated (pt0.2). Age and no previous biopsy trended towards significance
(p=0.08 and 0.07, respectively). On multivariate modelling, 4Kscore (OR=1.75 per 20% increase, 95% CI 1.19-2.57, p=0.004) and prostate volume (OR=0.67 per 20cc of volume, 95% CI 0.54-0.95, p=0.020) were independently associated. Comparing a 4Kscore cut off of 7.5%
with other risk indices (NCCN, D’Amico and CAPRA) demonstrated 4Kscore had 80% sensitivity, 40% specificity, 63% PPV, and 72% NPV. 4Kscore t7.5% did not miss detection of Gleason 8 PCa and missed 7% (1 of 14) pT3a/b.
Conclusions: The 4Kscore was significantly associatedwith adverse pathology at radical prostatectomy in men with Bx-proven Gleason 6 PCa
Antwort auf Beitrag Nr.: 58.198.904 von tomsylver am 12.07.18 17:23:21Mal schauen, wie Vifor Rayaldee vermarktet...
Rayaldee wurde in Kanada genehmigt. https://pdf.hres.ca/dpd_pm/00046289.PDF
Antwort auf Beitrag Nr.: 58.080.517 von faultcode am 27.06.18 17:29:44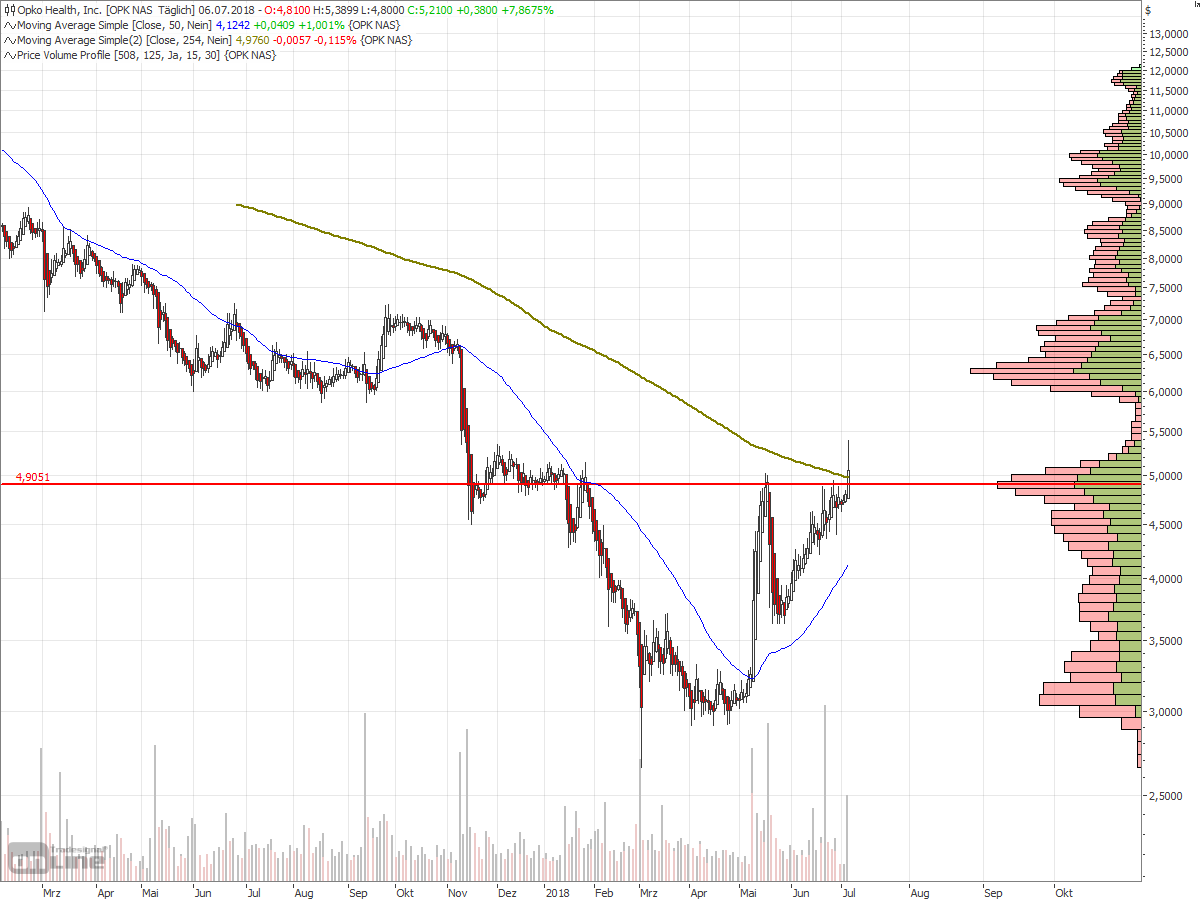
Grund: keiner
gut, der CEO hat am 3.7.18 dazugekauft - aber für seine Verhältnisse sehr bescheiden mit 10k Stück --> Symbolkauf:
https://backend.otcmarkets.com/otcapi/company/sec-filings/12…
drüber

Grund: keiner

gut, der CEO hat am 3.7.18 dazugekauft - aber für seine Verhältnisse sehr bescheiden mit 10k Stück --> Symbolkauf:
https://backend.otcmarkets.com/otcapi/company/sec-filings/12…
Antwort auf Beitrag Nr.: 57.802.847 von vonHS am 21.05.18 17:33:46Warum sollte der Test zu teuer sein?
Der Test verringert die Anzahl der Biopsien deutlich. Eine Biopsie kostet ein vielfachen des Tests. Zudem können bei den sonst unnötigen Biopsien Folgekrankheiten entstehen mit natürlich Folgekosten.
Für mich persönlich: bevor bei mir eine Biospie gemacht würde, würde ich lieber einmal nach Warschau oder Barcelona düsen und dort einen Test machen lassen, um mich dieser Prozedur nicht zu unterziehen...
Der Test verringert die Anzahl der Biopsien deutlich. Eine Biopsie kostet ein vielfachen des Tests. Zudem können bei den sonst unnötigen Biopsien Folgekrankheiten entstehen mit natürlich Folgekosten.
Für mich persönlich: bevor bei mir eine Biospie gemacht würde, würde ich lieber einmal nach Warschau oder Barcelona düsen und dort einen Test machen lassen, um mich dieser Prozedur nicht zu unterziehen...
Antwort auf Beitrag Nr.: 58.080.517 von faultcode am 27.06.18 17:29:44
=>
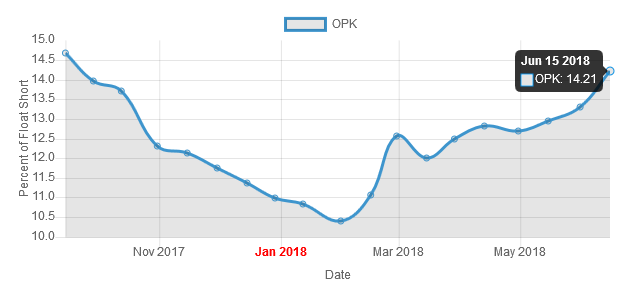
https://www.shortpainbot.com/?s=opk[/" target="_blank" rel="nofollow ugc noopener">https://www.shortpainbot.com/?s=opk[/
Shorties
...bauen wieder aus - und der Kurs steigt 
=>

https://www.shortpainbot.com/?s=opk[/" target="_blank" rel="nofollow ugc noopener">https://www.shortpainbot.com/?s=opk[/
Antwort auf Beitrag Nr.: 57.958.482 von faultcode am 11.06.18 15:40:42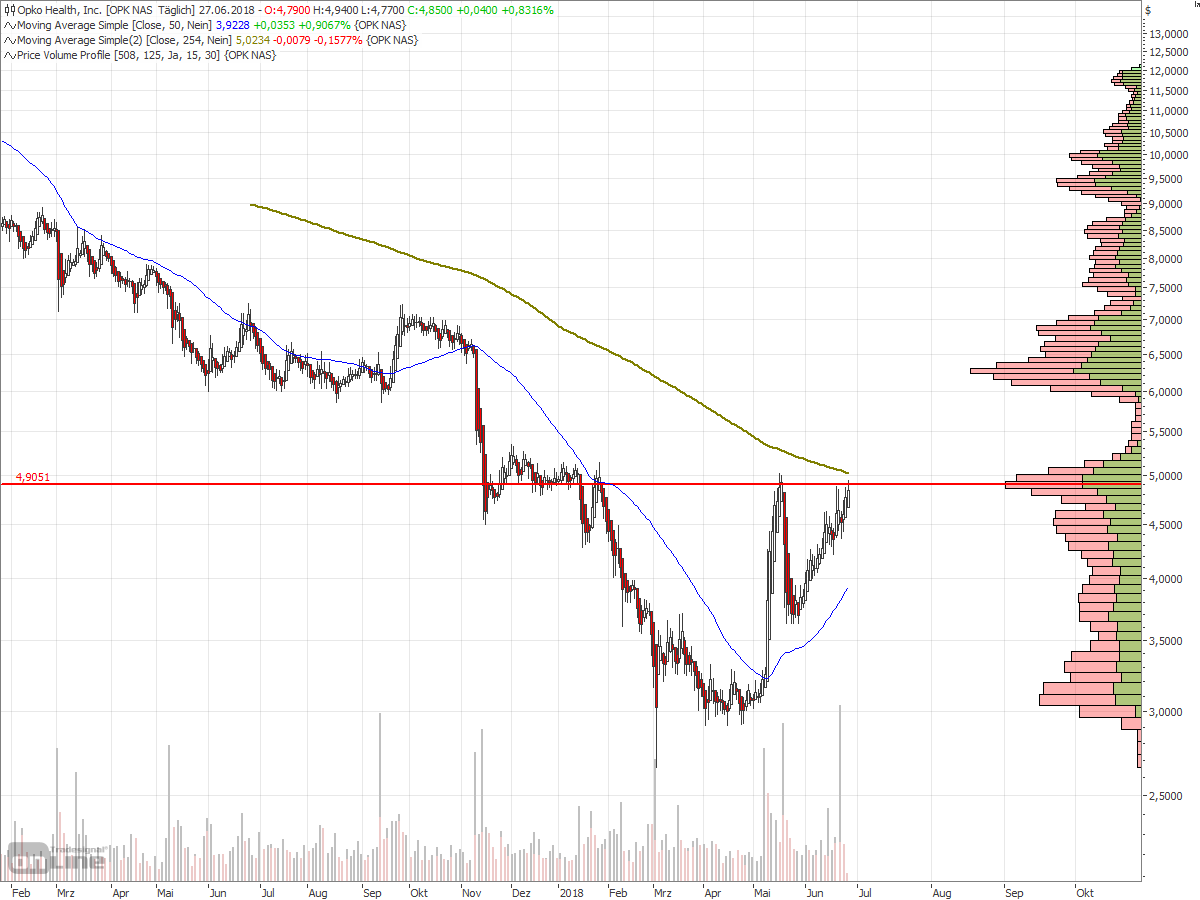
=> wenn die USD4.9 nachhaltig überschritten werden können (trotz aller fundamentaler Zweifel, die nach wie vor bestehen), dann war es eine klassische Untertassen-Formation (mit Hilfe von Shorties):
http://www.bullchart.de/ta/formationen/index8.php
=>
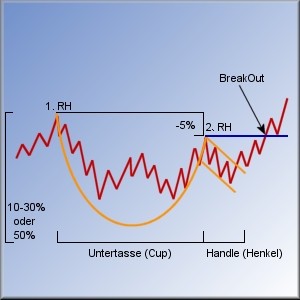
Untertasse

=> wenn die USD4.9 nachhaltig überschritten werden können (trotz aller fundamentaler Zweifel, die nach wie vor bestehen), dann war es eine klassische Untertassen-Formation (mit Hilfe von Shorties):
http://www.bullchart.de/ta/formationen/index8.php
=>

OPKO Health Completes Enrollment in Phase 2b Study of OPK88003 to Treat Type 2 Diabetes and Obesity
http://investor.opko.com/news-releases/news-release-details/…=>
MIAMI, June 11, 2018 (GLOBE NEWSWIRE) -- OPKO Health, Inc. (NASDAQ:OPK) today announced it has completed enrollment of 110 subjects in a Phase 2b dose escalation trial of OPK88003 to treat type 2 diabetes and obesity.
OPK88003 is a once weekly injectable oxyntomodulin medicine with GLP-1 and glucagon dual agonist activity. The primary efficacy endpoint is the reduction of HbA1c levels over a 30-week period; weight loss and improvement of blood lipid profiles are secondary endpoints.
Based on preclinical and Phase 2 data, the use of the dual agonist, OPK88003, once-weekly with an optimized dosing regimen, could more effectively improve glucose control, increase weight loss and improve the lipid profile in type 2 diabetics compared with the currently available GLP-1 medicines.
"We are pleased to announce complete enrollment in the Phase 2b dose escalation trial of OPK88003 two months after we initiated the study. Our confidence in this compound is supported by the earlier Phase 2 data, and it is our hope that the outcome of the current study along with our existing body of data will prepare us for a pivotal Phase 3 program,” stated Phillip Frost, M.D., Chairman and Chief Executive Officer of OPKO Health. “We believe the dual agonist activity of OPK88003 may better control glucose and improve long term cardiovascular outcomes in the growing population of type 2 diabetics worldwide."
According to the American Diabetes Association (ADA), approximately 30 million people in the U.S. have type 2 diabetes and 1.5 million Americans are diagnosed each year. The ADA estimates the annual cost of care for diabetic patients to be $327 billion, including direct medical costs and reduced productivity.
=> langsam wird guter Rat teuer hier:

Hier gibt es noch eine Bericht zu Opko:
http://www.globes.co.il/en/article-recommendation-against-me…
Außerdem hat Opko eine Frist bis zum 05.07.2018 erhalten, um zu den Feststellungen bei Medicare Stellung zu nehmen.
Welche Auswirkungen das Problem auf Opko hat, weiß man wohl gar nicht so genau:
"Opko is not reporting exactly what its revenue from the test was, probably because it was not enough to justify reporting it."
Fakt ist, dass der CEO aber weiterhin kräftig kauft:
http://investor.opko.com/static-files/fe6c320c-cbb9-4ee0-917…
http://www.globes.co.il/en/article-recommendation-against-me…
Außerdem hat Opko eine Frist bis zum 05.07.2018 erhalten, um zu den Feststellungen bei Medicare Stellung zu nehmen.
Welche Auswirkungen das Problem auf Opko hat, weiß man wohl gar nicht so genau:
"Opko is not reporting exactly what its revenue from the test was, probably because it was not enough to justify reporting it."
Fakt ist, dass der CEO aber weiterhin kräftig kauft:
http://investor.opko.com/static-files/fe6c320c-cbb9-4ee0-917…
Antwort auf Beitrag Nr.: 57.799.340 von SmartCap am 20.05.18 19:12:14Der Link zum Twitter-Bild von Hindenburg Research vom 18.05.2018 lautet:
https://twitter.com/HindenburgRes/status/997493269662699520
Um das Problem beim Namen zu nennen: der Krebstest von Opko ist einfach zu teuer!
https://twitter.com/HindenburgRes/status/997493269662699520
Um das Problem beim Namen zu nennen: der Krebstest von Opko ist einfach zu teuer!





















