Pieris Pharmaceuticals - Die letzten 30 Beiträge
eröffnet am 06.01.17 13:11:27 von
neuester Beitrag 28.08.23 08:45:50 von
neuester Beitrag 28.08.23 08:45:50 von
Beiträge: 104
ID: 1.244.291
ID: 1.244.291
Aufrufe heute: 0
Gesamt: 11.896
Gesamt: 11.896
Aktive User: 0
ISIN: US7207952026 · WKN: A40A3W · Symbol: PIRS
11,610
USD
-2,68 %
-0,320 USD
Letzter Kurs 02:00:00 NYSE
Neuigkeiten
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,9000 | +59,66 | |
| 7,4250 | +45,30 | |
| 0,5922 | +44,44 | |
| 11,890 | +43,25 | |
| 4,7450 | +35,57 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 8,0100 | -12,85 | |
| 9,8600 | -15,58 | |
| 2,5000 | -17,76 | |
| 2,1050 | -22,32 | |
| 1,6052 | -30,21 |
Beitrag zu dieser Diskussion schreiben
Neue Woche, ich erwarte einen signifikanten Kursanstieg bis zum Ende der Woche...Das Interesse wächst und nach der gesunden Korrektur in der letzten Woche werden die Karten neu gemischt. Viel Erfolg
Antwort auf Beitrag Nr.: 74.372.432 von tupper am 24.08.23 12:05:19Denke bis zum Gap 0,89 $ wird es hier mindestens getrieben 🤙 darauf baut ja der mittlerweile gute Hype…Vol steigt täglich, ab 13.00 Uhr sind wir schlauer, wo die Reise hingeht 💪
Antwort auf Beitrag Nr.: 74.372.405 von Phoenix369 am 24.08.23 12:01:35auch Zufallsgriff, aber schön dass es noch ein paar gibt 😂
Achtung Tipp Fehler : US-Umsätze wäre richtig!
Antwort auf Beitrag Nr.: 74.372.183 von Ben26 am 24.08.23 11:27:29Sind doch noch genügend dabei. Da will sich nach dem Erlebnis des brutalen Absturzes eben niemand zu weit aus dem Fenster lehnen.
Der Chart sieht inzwischen natürlich riskant aus, aber wen interessiert noch der Chart, falls es zu einer Übernahme kommt.
Absturz befürchte ich momentan nicht. Konsolidierung wäre denkbar, kann aber auch sein, dass der "Wahnsinn" einfachh weiter geht.
Der Chart sieht inzwischen natürlich riskant aus, aber wen interessiert noch der Chart, falls es zu einer Übernahme kommt.
Absturz befürchte ich momentan nicht. Konsolidierung wäre denkbar, kann aber auch sein, dass der "Wahnsinn" einfachh weiter geht.
könnte weiter steigen!
Bin seit dieser Woche mit dabei. Bin im Stockwits-Forum auf diese Aktie aufmerksam gemacht worden. Man darf dort natürlich nicht alles glauben. Ich schaue mir immer die US-Unsätze an. Seit 16.8. beachtlich mehr Umsätze wie davor. Da könnte was im Busch sein. Nur meine Meinung.
Antwort auf Beitrag Nr.: 74.369.270 von faultcode am 23.08.23 20:29:31Bin ich nicht alleine 😂?
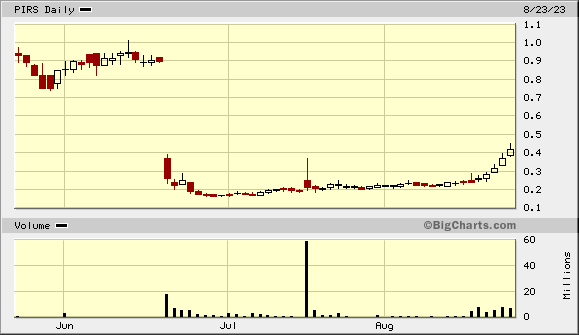
und was nun?
Schade das es hier so ruhig ist 🤙
Antwort auf Beitrag Nr.: 74.362.175 von tupper am 22.08.23 17:38:44So sehe ich es auch 👌👍
Gut, dass die Gap-Spekulation auch noch ein bisschen Substanz hat. BVF hat immerhin 20% der Aktien, bei whalewisdom stand sogar 25%. An EK sind noch 40 Mio da = 0,40 pS und Milestones könnten noch von Servier, Boston Pharma und Seagen kommen.
Eine Übernahme könnte ich mir bei der MK auch gut vorstellen.
Es gab schon riskantere Spekulationen.
Eine Übernahme könnte ich mir bei der MK auch gut vorstellen.
Es gab schon riskantere Spekulationen.
Knappe 40% haben wir bereits und ab 0,35 $ ist das Gap bis 0,90 $ offen - eine Menge Potenzial..
Der Trend ist sehr schön, sollte über mittlere Sicht 30%-60% bei stetigem Newsflow bringen. Es gibt ja einiges was demnächst berichtet werden könnte ✌️
Position gestartet
17.7.
Pieris Pharmaceuticals Provides Strategic Update and Announces Restructuring
https://www.accesswire.com/768538/Pieris-Pharmaceuticals-Pro…
...
Pieris Pharmaceuticals, Inc. (Nasdaq:PIRS), a clinical-stage biotechnology company advancing novel biotherapeutics through its proprietary Anticalin® technology platform for respiratory diseases and cancer, today provided a strategic update following recent events that have impacted its inhaled respiratory franchise, including AstraZeneca's discontinuation of enrollment of the Phase 2a study for elarekibep.
AstraZeneca has now informed the Company of its decision to terminate the parties' R&D collaboration agreement and hand back elarekibep along with discontinuing the remaining discovery program.
Pieris' management and board of directors have assessed several strategic options, which will include focusing on execution of new or expanded partnerships to advance its therapeutic programs, including cinrebafusp alfa (PRS-343), PRS-220 and PRS-400.
While it explores potential transactions, Pieris will prioritize capital preservation, with cash, cash equivalents and investments totaling approximately $54.9 million as of June 30, 2023. As part of its cash preservation plan, Pieris initiated a corporate restructuring that will result in a reduction of the Company's workforce by approximately 70%.
These and other cost-saving measures are expected to maximize the opportunity to pursue a range of transactions across both its respiratory and immuno-oncology franchises and its discovery platform. Pieris has retained Stifel, Nicolaus & Company, Inc. as its exclusive financial advisor to evaluate a range of strategic options.
These strategic options may include an acquisition, merger, reverse merger, other business combination, sale of assets, financing alternatives, licensing, or other strategic transactions involving the Company. There can be no assurance of a transaction, a successful outcome of these efforts, or the form or timing of any such outcome. The Company does not intend to make any further disclosures regarding the strategic review process unless and until a specific course of action is approved by the Company's board of directors or until the Company determines that further disclosure is appropriate.
"We are pursuing strategic options across three main areas following the recent developments that have impacted our ability to independently advance our respiratory programs," commented President and CEO Stephen Yoder. "One track is accelerating partnering discussions of PRS-220 and PRS-400. A second focal area is diligently selecting the best possible development partner and deal structure to re-initiate clinical development of cinrebafusp alfa, our former lead immuno-oncology asset, which has shown 100% ORR in five patients in a HER2+ gastric cancer trial that was discontinued for strategic reasons. Third, we will explore whether our balance sheet, position as a public company, and other assets are of strategic value to a range of third parties." Mr. Yoder continued, "While the challenges we recently experienced across our respiratory franchise have forced us to make very difficult personnel decisions, I cannot express enough gratitude to our departing colleagues for their dedication, collaborative spirit and integrity."
...
Pieris Pharmaceuticals Provides Strategic Update and Announces Restructuring
https://www.accesswire.com/768538/Pieris-Pharmaceuticals-Pro…
...
Pieris Pharmaceuticals, Inc. (Nasdaq:PIRS), a clinical-stage biotechnology company advancing novel biotherapeutics through its proprietary Anticalin® technology platform for respiratory diseases and cancer, today provided a strategic update following recent events that have impacted its inhaled respiratory franchise, including AstraZeneca's discontinuation of enrollment of the Phase 2a study for elarekibep.
AstraZeneca has now informed the Company of its decision to terminate the parties' R&D collaboration agreement and hand back elarekibep along with discontinuing the remaining discovery program.
Pieris' management and board of directors have assessed several strategic options, which will include focusing on execution of new or expanded partnerships to advance its therapeutic programs, including cinrebafusp alfa (PRS-343), PRS-220 and PRS-400.
While it explores potential transactions, Pieris will prioritize capital preservation, with cash, cash equivalents and investments totaling approximately $54.9 million as of June 30, 2023. As part of its cash preservation plan, Pieris initiated a corporate restructuring that will result in a reduction of the Company's workforce by approximately 70%.
These and other cost-saving measures are expected to maximize the opportunity to pursue a range of transactions across both its respiratory and immuno-oncology franchises and its discovery platform. Pieris has retained Stifel, Nicolaus & Company, Inc. as its exclusive financial advisor to evaluate a range of strategic options.
These strategic options may include an acquisition, merger, reverse merger, other business combination, sale of assets, financing alternatives, licensing, or other strategic transactions involving the Company. There can be no assurance of a transaction, a successful outcome of these efforts, or the form or timing of any such outcome. The Company does not intend to make any further disclosures regarding the strategic review process unless and until a specific course of action is approved by the Company's board of directors or until the Company determines that further disclosure is appropriate.
"We are pursuing strategic options across three main areas following the recent developments that have impacted our ability to independently advance our respiratory programs," commented President and CEO Stephen Yoder. "One track is accelerating partnering discussions of PRS-220 and PRS-400. A second focal area is diligently selecting the best possible development partner and deal structure to re-initiate clinical development of cinrebafusp alfa, our former lead immuno-oncology asset, which has shown 100% ORR in five patients in a HER2+ gastric cancer trial that was discontinued for strategic reasons. Third, we will explore whether our balance sheet, position as a public company, and other assets are of strategic value to a range of third parties." Mr. Yoder continued, "While the challenges we recently experienced across our respiratory franchise have forced us to make very difficult personnel decisions, I cannot express enough gratitude to our departing colleagues for their dedication, collaborative spirit and integrity."
...
Pieris sollte man noch nicht abschreiben, die haben jede Menge Patente.
https://patents.justia.com/assignee/pieris-pharmaceuticals-g…
https://patents.justia.com/assignee/pieris-pharmaceuticals-g…
Antwort auf Beitrag Nr.: 74.038.473 von watchingtheflood am 21.06.23 16:12:38So ist es, muß man mit umgehen, aber das Jahr ist noch jung.
Antwort auf Beitrag Nr.: 74.037.411 von biogenetic am 21.06.23 13:56:06Man kann jetzt nicht behaupten, dass Du zuletzt vom Glück verfolgt wurdest ....
Nun ja, Biotech hat seine eigenen harten Gesetze.
Nun ja, Biotech hat seine eigenen harten Gesetze.
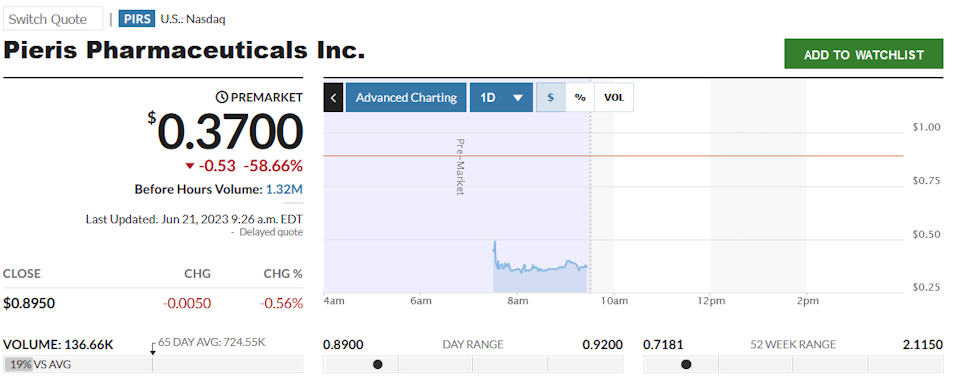
Das war es hier. Bin raus, sehe keinen Mehrwert im Rest der Pipeline.
21.6.
Pieris Pharmaceuticals Announces AstraZeneca Discontinuation of Phase 2a Trial of Elarekibep (PRS-060/AZD1402) Due to New Non-Clinical Safety Findings From 13-week Toxicology Study
https://www.wallstreet-online.de/nachricht/17065536-pieris-p…
...
Pieris Pharmaceuticals Announces AstraZeneca Discontinuation of Phase 2a Trial of Elarekibep (PRS-060/AZD1402) Due to New Non-Clinical Safety Findings From 13-week Toxicology Study
https://www.wallstreet-online.de/nachricht/17065536-pieris-p…
...
https://www.accesswire.com/756232/Pieris-Pharmaceuticals-Ann…
Pieris Pharmaceuticals Announces Presentation of Promising Preclinical Data for PRS-220 at ATS 2023 International Conference
BOSTON, MA / ACCESSWIRE / May 21, 2023 / Pieris Pharmaceuticals, Inc. (Nasdaq:PIRS), a clinical-stage biotechnology company advancing novel biotherapeutics through its proprietary Anticalin® technology platform for respiratory diseases, cancer, and other indications, announced the presentation of preclinical data from the Company's inhaled connective tissue growth factor (CTGF) antagonist program, PRS-220, for idiopathic pulmonary fibrosis (IPF) at the annual American Thoracic Society (ATS) International Conference being held in Washington, D.C. May 19-24, 2023.
The poster will be presented on May 21, 2023, from 11:30 AM to 1:15 PM (Session A67, P1052) and demonstrates that PRS-220 achieved proof of concept in a silica-induced lung fibrosis mouse model when delivered to the lung by inhalation and outperformed a systemically administered CTGF-targeting monoclonal antibody, reinforcing the transformative potential of a local intervention in this pathway. A copy of the presentation can be viewed HERE.
The poster also includes preclinical data demonstrating a favorable profile of PRS-220 for inhaled delivery and CTGF targeting in the lung, including a superior lung biodistribution profile compared to a systemically administered anti-CTGF monoclonal antibody, a lung PK profile supporting once or twice daily dosing in humans, and desired aerosol performance upon nebulization to effectively target pulmonary tissue of interest.
"Targeting CTGF is a clinically validated treatment approach for IPF, as shown by trial data from the intravenously delivered monoclonal antibody pamrevlumab. Based on the pulmonary pathophysiology associated with IPF, we believe that directly inhibiting CTGF in the lung with an inhaled therapy could result in more efficient target saturation, superior clinical efficacy, and greater convenience for patients," said Shane Olwill Ph.D., Chief Development Officer at Pieris. "Our ongoing PRS-220 Phase 1 healthy volunteer study is on track to provide data in the second half of this year, and we are eager to initiate development in IPF patients to further evaluate the clinical potential of this promising investigational medicine."
About PRS-220:
PRS-220 is an inhaled Anticalin protein targeting CTGF for the treatment of IPF and other fibrotic lung diseases. Previously reported preclinical data for PRS-220 demonstrated superior on-target potency compared to pamrevlumab, an intravenously infused CTGF antagonist in late-stage clinical development. Pieris continues to benefit from a meaningful grant from the Bavarian government, which supports early-stage development of this program.
...
Pieris Pharmaceuticals Announces Presentation of Promising Preclinical Data for PRS-220 at ATS 2023 International Conference
BOSTON, MA / ACCESSWIRE / May 21, 2023 / Pieris Pharmaceuticals, Inc. (Nasdaq:PIRS), a clinical-stage biotechnology company advancing novel biotherapeutics through its proprietary Anticalin® technology platform for respiratory diseases, cancer, and other indications, announced the presentation of preclinical data from the Company's inhaled connective tissue growth factor (CTGF) antagonist program, PRS-220, for idiopathic pulmonary fibrosis (IPF) at the annual American Thoracic Society (ATS) International Conference being held in Washington, D.C. May 19-24, 2023.
The poster will be presented on May 21, 2023, from 11:30 AM to 1:15 PM (Session A67, P1052) and demonstrates that PRS-220 achieved proof of concept in a silica-induced lung fibrosis mouse model when delivered to the lung by inhalation and outperformed a systemically administered CTGF-targeting monoclonal antibody, reinforcing the transformative potential of a local intervention in this pathway. A copy of the presentation can be viewed HERE.
The poster also includes preclinical data demonstrating a favorable profile of PRS-220 for inhaled delivery and CTGF targeting in the lung, including a superior lung biodistribution profile compared to a systemically administered anti-CTGF monoclonal antibody, a lung PK profile supporting once or twice daily dosing in humans, and desired aerosol performance upon nebulization to effectively target pulmonary tissue of interest.
"Targeting CTGF is a clinically validated treatment approach for IPF, as shown by trial data from the intravenously delivered monoclonal antibody pamrevlumab. Based on the pulmonary pathophysiology associated with IPF, we believe that directly inhibiting CTGF in the lung with an inhaled therapy could result in more efficient target saturation, superior clinical efficacy, and greater convenience for patients," said Shane Olwill Ph.D., Chief Development Officer at Pieris. "Our ongoing PRS-220 Phase 1 healthy volunteer study is on track to provide data in the second half of this year, and we are eager to initiate development in IPF patients to further evaluate the clinical potential of this promising investigational medicine."
About PRS-220:
PRS-220 is an inhaled Anticalin protein targeting CTGF for the treatment of IPF and other fibrotic lung diseases. Previously reported preclinical data for PRS-220 demonstrated superior on-target potency compared to pamrevlumab, an intravenously infused CTGF antagonist in late-stage clinical development. Pieris continues to benefit from a meaningful grant from the Bavarian government, which supports early-stage development of this program.
...
Jetze wo die MC gut runtergekommen ist, wegen dem AZ Zusatzjahr und Kohla ran muss, macht doch Yoder klar, dass er eventuell die IO Anticalin Sparte versilbern will.
Seht ihr da gute Chancen? Wieviel könnten die dadurch erdealen? is ein ipo denkbar?
Seht ihr da gute Chancen? Wieviel könnten die dadurch erdealen? is ein ipo denkbar?
Pieris Pharmaceuticals Announces Presentation of Positive Clinical Data for Cinrebafusp Alpa (PRS-343) At 2023 AACR Annual Meeting
https://www.pieris.com/investors/news-events/press-releases/…
"The ability to drive response in patients who progressed on the most potent currently available therapies provides further evidence of the differentiation of cinrebafusp alfa in the HER2 landscape as well as the overall 4-1BB franchise. Beyond cinrebafusp alfa, we look forward to the progression of-and future data read-outs for-the broader 4-1BB franchise, including our PD-L1/4-1BB bispecific in collaboration with Servier in Phase 1 studies, our CD228/4-1BB bispecific with Seagen in Phase 1 studies, and our GPC-3/4-1BB bispecific with Boston Pharmaceuticals entering the clinic shortly."
https://www.pieris.com/investors/news-events/press-releases/…
"The ability to drive response in patients who progressed on the most potent currently available therapies provides further evidence of the differentiation of cinrebafusp alfa in the HER2 landscape as well as the overall 4-1BB franchise. Beyond cinrebafusp alfa, we look forward to the progression of-and future data read-outs for-the broader 4-1BB franchise, including our PD-L1/4-1BB bispecific in collaboration with Servier in Phase 1 studies, our CD228/4-1BB bispecific with Seagen in Phase 1 studies, and our GPC-3/4-1BB bispecific with Boston Pharmaceuticals entering the clinic shortly."
Verzögerung bei AstraZeneca
https://www.pieris.com/investors/news-events/press-releases/…
KE oder lizensiert Yoder wieder was aus.
https://www.pieris.com/investors/news-events/press-releases/…
KE oder lizensiert Yoder wieder was aus.
Is Pieris Pharmaceuticals Inc (PIRS) a Leader in the Biotechnology Industry?
The 60 rating InvestorsObserver gives to Pieris Pharmaceuticals Inc (PIRS) stock puts it near the top of the Biotechnology industry. In addition to scoring higher than 72 percent of stocks in the Biotechnology industry, PIRS’s 60 overall rating means the stock scores better than 60 percent of all stocks.Quelle: https://www.investorsobserver.com/news/stock-update/is-pieri…
10.1.
Pieris Pharmaceuticals Announces $5 Million Milestone from Seagen for Initiation of Phase 1 Trial of CD228 x 4-1BB Bispecific Molecule (Mabcalin SGN-BB228 (PRS-346)
https://www.accesswire.com/734629/Pieris-Pharmaceuticals-Ann…
...
Pieris Pharmaceuticals Announces $5 Million Milestone from Seagen for Initiation of Phase 1 Trial of CD228 x 4-1BB Bispecific Molecule (Mabcalin SGN-BB228 (PRS-346)
https://www.accesswire.com/734629/Pieris-Pharmaceuticals-Ann…
...
Antwort auf Beitrag Nr.: 72.931.909 von biogenetic am 17.12.22 00:14:31Wobei Übernahme kann es eigentlich nicht sein, wenn es bis May 2024 zurückgehalten wird.
Sehr spannend.
Sehr spannend.
Confidential Treatment Order
Informationen werden zurückgehalten die einen Wettbewerbsnachteil hätten. https://www.sec.gov/Archives/edgar/data/1583648/999999999722…
Könnte eine Übernahme sein mit einem bereits geeigneten Preis, damit es nicht zu einem Bieter Wettstreit kommt.
Oder aber es geht um die zwei bisher zurückgehaltenen Projekte von Astra Zeneca.
Pieris Pharmaceuticals





















