Aclaris Therapeutics (ACRS) - Marktkap: $43 M - Reboundplay (Seite 234)
eröffnet am 13.09.19 08:31:34 von
neuester Beitrag 01.02.24 00:59:35 von
neuester Beitrag 01.02.24 00:59:35 von
Beiträge: 2.342
ID: 1.311.638
ID: 1.311.638
Aufrufe heute: 0
Gesamt: 287.963
Gesamt: 287.963
Aktive User: 0
ISIN: US00461U1051 · WKN: A1412H · Symbol: ACRS
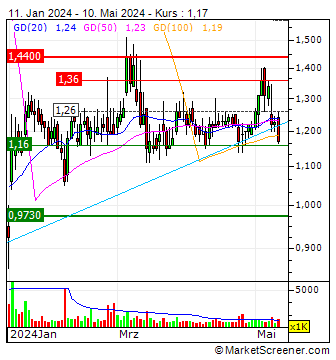
1,1900
USD
0,00 %
0,0000 USD
Letzter Kurs 02:00:00 Nasdaq
Neuigkeiten
07.05.24 · globenewswire |
30.04.24 · globenewswire |
27.02.24 · globenewswire |
16.01.24 · globenewswire |
Werte aus der Branche Pharmaindustrie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 0,7019 | +71,15 | |
| 0,8000 | +45,45 | |
| 3,0000 | +42,18 | |
| 0,6100 | +25,70 | |
| 2,0600 | +24,85 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 20,360 | -11,48 | |
| 1,2900 | -11,64 | |
| 1,1060 | -14,79 | |
| 4,0400 | -14,95 | |
| 23,700 | -19,52 |
Beitrag zu dieser Diskussion schreiben
Wo liegt euer EK hier?
Antwort auf Beitrag Nr.: 61.478.438 von Sugar2000 am 13.09.19 15:31:35Das ist ALLES Geschichte seit der änderung der Strategie man stellt die eigne Vermarktung ein was ziemlich viel Geld verschlungen und will jetzt alles verpartnern deshalb werden die kosten massiv fallen . Das steht doch alles in der Nachricht warum liest man das nicht ?
Robbins Arroyo LLP Reminds Shareholders That Aclaris Therapeutics, Inc. (ACRS) Sued for Misleading
Shareholders
SAN DIEGO & WAYNE, Pa. -- September 9, 2019
Shareholder rights law firm Robbins Arroyo LLP reminds shareholders that a
purchaser of Aclaris Therapeutics, Inc. (NASDAQ: ACRS) filed a class action
complaint against the company's officers and directors for alleged violations of
the Securities Exchange Act of 1934 between May 8, 2018 and June 20, 2019.
Aclaris, a pharmaceutical company, identifies, develops, and commercializes
therapies for dermatological and immune-inflammatory diseases. Its lead
product is ESKATA, a hydrogen peroxide topical solution that treats seborrheic
keratosis.
According to the complaint, in May 2018, Aclaris announced the launch of ESKATA, touting the product as the "first and only FDA-approved topical treatment for seborrheic keratosis." Despite positive expectations, Aclaris acknowledged in its 2017 10-K that ESKATA was subject to post-marketing restrictions and that failure to comply with regulations could lead to penalties. In its subsequent filings with the SEC, Aclaris touted its revenue from its sale of ESKATA and consistently assured that there were no material changes in its risk factors. However, in reality, Aclaris' advertising materials for ESKATA minimized its risks and overstated its efficacy, leaving the company vulnerable to regulatory scrutiny. This reality was realized on June 20, 2019, when the FDA issued a letter in response to a direct-to-consumer video interview with a paid Aclaris spokesperson, stating that Aclaris' advertisement for ESKATA "[made] false or misleading claims" about the product's risks and efficacy by failing to mention risk of serious eye disorders or severe skin reactions. Since this news, Aclaris' share price has fallen $3.99, almost 80%.
If you purchased Aclaris Therapeutics, Inc. (ACRS) securities between May 8, 2018 and June 20, 2019, you have until September 30, 2019, to ask the court to be appointed lead plaintiff for the class.
Shareholders
SAN DIEGO & WAYNE, Pa. -- September 9, 2019
Shareholder rights law firm Robbins Arroyo LLP reminds shareholders that a
purchaser of Aclaris Therapeutics, Inc. (NASDAQ: ACRS) filed a class action
complaint against the company's officers and directors for alleged violations of
the Securities Exchange Act of 1934 between May 8, 2018 and June 20, 2019.
Aclaris, a pharmaceutical company, identifies, develops, and commercializes
therapies for dermatological and immune-inflammatory diseases. Its lead
product is ESKATA, a hydrogen peroxide topical solution that treats seborrheic
keratosis.
According to the complaint, in May 2018, Aclaris announced the launch of ESKATA, touting the product as the "first and only FDA-approved topical treatment for seborrheic keratosis." Despite positive expectations, Aclaris acknowledged in its 2017 10-K that ESKATA was subject to post-marketing restrictions and that failure to comply with regulations could lead to penalties. In its subsequent filings with the SEC, Aclaris touted its revenue from its sale of ESKATA and consistently assured that there were no material changes in its risk factors. However, in reality, Aclaris' advertising materials for ESKATA minimized its risks and overstated its efficacy, leaving the company vulnerable to regulatory scrutiny. This reality was realized on June 20, 2019, when the FDA issued a letter in response to a direct-to-consumer video interview with a paid Aclaris spokesperson, stating that Aclaris' advertisement for ESKATA "[made] false or misleading claims" about the product's risks and efficacy by failing to mention risk of serious eye disorders or severe skin reactions. Since this news, Aclaris' share price has fallen $3.99, almost 80%.
If you purchased Aclaris Therapeutics, Inc. (ACRS) securities between May 8, 2018 and June 20, 2019, you have until September 30, 2019, to ask the court to be appointed lead plaintiff for the class.
Allerdings ist der FCF heftigst negativ! Allein im ersten Halbjahr wurden 111 Mio. USD verbrannt...
Wir haben ebenfalls ein negatives EBITDA in Höhe von -135 Mio. USD!
Die SG&A Kosten sind mir persönlich mit 70 Mio. USD zu hoch. R&D zusätzliche 73 Mio. USD...
Reichen die 115 Mio. Cash so lange aus?
Wir haben ebenfalls ein negatives EBITDA in Höhe von -135 Mio. USD!
Die SG&A Kosten sind mir persönlich mit 70 Mio. USD zu hoch. R&D zusätzliche 73 Mio. USD...
Reichen die 115 Mio. Cash so lange aus?
Also das verstehe ich nicht....
Aber so ist Börse. Ich bin immer erst ein wenig skeptisch und suche das Haar in der Suppe. Stelle aber mal einen Fuß rein..
Danke für den thread
Aber so ist Börse. Ich bin immer erst ein wenig skeptisch und suche das Haar in der Suppe. Stelle aber mal einen Fuß rein..
Danke für den thread
Antwort auf Beitrag Nr.: 61.477.589 von Jack48 am 13.09.19 13:46:59Eine Phase 2 Studie ist im Juni gescheitert aber kurze Zeit später wurden positive P2 Daten für dieselbe indication gemeldet (Oralversion positiv / Topical gescheitert) trotzdem ist die Aktie gefallen weil höchstwahrscheinlich Insider schon wussten das ein Strategiewechsel bevorsteht . Für uns gut sonst hätten wir die Aktie niemals so günstig bekommen .
Aclaris Therapeutics Announces Phase 2 Clinical Trial of ATI-502 Topical in Patients with Alopecia Areata Did Not Meet Endpoints
https://investor.aclaristx.com/news-releases/news-release-de…
Aclaris Therapeutics Announces Phase 2 Clinical Trial of ATI-501 Oral in Patients with Alopecia Areata Met Primary Endpoint
https://investor.aclaristx.com/news-releases/news-release-de…
Aclaris Therapeutics Announces Phase 2 Clinical Trial of ATI-502 Topical in Patients with Alopecia Areata Did Not Meet Endpoints
https://investor.aclaristx.com/news-releases/news-release-de…
Aclaris Therapeutics Announces Phase 2 Clinical Trial of ATI-501 Oral in Patients with Alopecia Areata Met Primary Endpoint
https://investor.aclaristx.com/news-releases/news-release-de…
Antwort auf Beitrag Nr.: 61.477.559 von Biohero am 13.09.19 13:43:35Was war der Grund für den Absturz im Juni?
Antwort auf Beitrag Nr.: 61.477.274 von Biohero am 13.09.19 13:08:16Nur um zu verdeutlichen wie abnormal unterbewertet die Aktie ist : man bekommt sie nicht nur 60% unter Cash man kriegt sogar noch 2 Zugelassene Produkte eines davon von Allergan für $65+ Million einlizenziert plus ein weiteres für das in kürze die final Phase 3 Ergebnisse veröffentlicht werden . Allein Rhofade von Allergan ist mindestens soviel Wert wie die aktuelle Bewertung und dann kommt noch die 60% unter Cash notierung schon übel unterbewertet
Antwort auf Beitrag Nr.: 61.477.175 von Biohero am 13.09.19 12:58:18 Cash : $115 M ausreichend bis 2021 weiteres Cash durch geplante Auslizenzierungen und Royalties eingerechnet
Cash : $115 M ausreichend bis Q3 2021 weiteres Cash durch geplante Auslizenzierungen und Royalties NICHT eingerechnet ...
Aclaris Is Worth A Look After Positive Data And Strategic Review Potential
https://seekingalpha.com/article/4284936-aclaris-worth-look-…
Pipeline
https://www.aclaristx.com/pipeline/
Cash : $115 M ausreichend bis Q3 2021 weiteres Cash durch geplante Auslizenzierungen und Royalties NICHT eingerechnet ...
Aclaris Is Worth A Look After Positive Data And Strategic Review Potential
https://seekingalpha.com/article/4284936-aclaris-worth-look-…
Pipeline
https://www.aclaristx.com/pipeline/
Antwort auf Beitrag Nr.: 61.474.463 von liquidx am 13.09.19 08:31:34titel ein etwas ungünstig geraten aber okay...
Besser könnte ein Rebound Play nicht aussehen die Aktie mehr als nur Unterbewertet mehr kann ich dazu nicht sagen einfach eine Riesenchance hier schnell geld zu verdienen ...
Aclaris Therapeutics (ACRS)
Marktkap $44.6 M
Cash : $115 M ausreichend bis 2021 weiteres Cash durch geplante Auslizenzierungen und Royalties eingerechnet
eingerechnet
Kurs: 1.08
Aktienanzahl: 41,3 M
Aclaris Therapeutics to Acquire Worldwide Rights to RHOFADE® from Allergan
https://www.globenewswire.com/news-release/2018/10/15/162151…
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-de…
Aclaris anticipates that its current cash and cash equivalents will be sufficient to fund its operations into the third quarter of 2021
Actively Seeking a Commercialization Partner for RHOFADE® (oxymetazoline hydrochloride) cream, 1%
Aclaris will also seek strategic partners for ESKATA® (hydrogen peroxide) topical solution, 40%
Aclaris is actively seeking a strategic partner to commercialize its drug candidate A-101 45% Topical Solution,an investigational compound being developed as a potential treatment for verruca vulgaris (common warts). The Company’s two ongoing Phase 3 pivotal clinical trials, THWART-1 and THWART-2, in which A-101 45% Topical Solution is being evaluated as a potential treatment for common warts, are progressing as planned. Aclaris has completed enrollment of more than 1,000 patients across these two trials, and data from both trials are expected in the second half of 2019.
Aclaris is actively seeking a development and commercialization partner for its drug candidates, ATI-501 (oral) and ATI-502 (topical), which are investigational Janus Kinase (JAK) 1/3 inhibitor compounds for the potential treatment of alopecia.
Morgan Stanley reports 5% passive stake in Aclaris Therapeutics...08/29,2019
https://thefly.com/landingPageNews.php?id=2956902
Morgan Stanley reports 5% passive stake in Aclaris Therapeutics In a regulatory filing, Morgan Stanley (MS) disclosed a 5% stake in Aclaris (ACRS), which represents about 2M shares. The filing does not allow for activism.
Größten Aktionäre:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
Besser könnte ein Rebound Play nicht aussehen die Aktie mehr als nur Unterbewertet mehr kann ich dazu nicht sagen einfach eine Riesenchance hier schnell geld zu verdienen ...
Aclaris Therapeutics (ACRS)
Marktkap $44.6 M
Cash : $115 M ausreichend bis 2021 weiteres Cash durch geplante Auslizenzierungen und Royalties
Kurs: 1.08
Aktienanzahl: 41,3 M
Aclaris Therapeutics to Acquire Worldwide Rights to RHOFADE® from Allergan
https://www.globenewswire.com/news-release/2018/10/15/162151…
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-de…
Aclaris anticipates that its current cash and cash equivalents will be sufficient to fund its operations into the third quarter of 2021
Actively Seeking a Commercialization Partner for RHOFADE® (oxymetazoline hydrochloride) cream, 1%
Aclaris will also seek strategic partners for ESKATA® (hydrogen peroxide) topical solution, 40%
Aclaris is actively seeking a strategic partner to commercialize its drug candidate A-101 45% Topical Solution,an investigational compound being developed as a potential treatment for verruca vulgaris (common warts). The Company’s two ongoing Phase 3 pivotal clinical trials, THWART-1 and THWART-2, in which A-101 45% Topical Solution is being evaluated as a potential treatment for common warts, are progressing as planned. Aclaris has completed enrollment of more than 1,000 patients across these two trials, and data from both trials are expected in the second half of 2019.
Aclaris is actively seeking a development and commercialization partner for its drug candidates, ATI-501 (oral) and ATI-502 (topical), which are investigational Janus Kinase (JAK) 1/3 inhibitor compounds for the potential treatment of alopecia.
Morgan Stanley reports 5% passive stake in Aclaris Therapeutics...08/29,2019
https://thefly.com/landingPageNews.php?id=2956902
Morgan Stanley reports 5% passive stake in Aclaris Therapeutics In a regulatory filing, Morgan Stanley (MS) disclosed a 5% stake in Aclaris (ACRS), which represents about 2M shares. The filing does not allow for activism.
Größten Aktionäre:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022





















