Immunogen: Mirvetuximab soravtansine ein zukünftiger Blockbuster (Seite 2)
eröffnet am 27.01.16 18:48:47 von
neuester Beitrag 30.11.23 18:48:17 von
neuester Beitrag 30.11.23 18:48:17 von
Beiträge: 395
ID: 1.225.530
ID: 1.225.530
Aufrufe heute: 0
Gesamt: 43.395
Gesamt: 43.395
Aktive User: 0
ISIN: US45253H1014 · WKN: 878613 · Symbol: IMU
29,00
EUR
+0,35 %
+0,10 EUR
Letzter Kurs 09.02.24 Tradegate
Neuigkeiten
| Titel |
|---|
01.02.24 · Business Wire (engl.) |
03.01.24 · Business Wire (engl.) |
10.12.23 · Business Wire (engl.) |
05.12.23 · Business Wire (engl.) |
05.12.23 · Business Wire (engl.) |
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 4,9600 | +41,71 | |
| 5,3000 | +28,64 | |
| 1,3000 | +26,21 | |
| 1,1000 | +22,22 | |
| 16,550 | +14,14 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 2,1200 | -18,46 | |
| 9,6000 | -18,64 | |
| 2,0850 | -19,19 | |
| 1,4600 | -24,56 | |
| 1,7000 | -31,17 |
Beitrag zu dieser Diskussion schreiben
Seit einiger Zeit geht's immer abwärts, schade.
Hab mich wohl hier mit der Investition vertan.
🤔
LG der 🐋
Hab mich wohl hier mit der Investition vertan.
🤔
LG der 🐋
Antwort auf Beitrag Nr.: 66.980.756 von Malecon am 12.02.21 16:46:23Hallo Weiß jemand warum ist die letzten Tage so starnberg Abging mit emotionen ich hab keine
richtigen Nachrichten dazu gefunden danke der Orka
richtigen Nachrichten dazu gefunden danke der Orka
Ein schöner Schub nach oben heute:
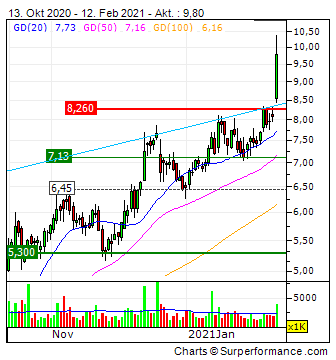
🎵
Antwort auf Beitrag Nr.: 62.961.116 von rheinwein am 11.03.20 12:57:17
ich würde sagen: JA allerdings sollte man permanent beobachten (können) !!
Zitat von rheinwein: Mal eine Frage an die Profis:
Im Finanzreport vom Febr. stand:
ANTICIPATED 2020 EVENTS
- Initiate pivotal SORAYA trial in the first quarter of 2020 and continue enrollment in the confirmatory Phase 3 MIRASOL trial.
- Open an additional platinum-sensitive investigator sponsored trial evaluating mirvetuximab in combination with carboplatin.
- Present initial data from the Phase 1b FORWARD II platinum-agnostic doublet cohort evaluating mirvetuximab in combination with Avastin (bevacizumab) in mid-2020 and updated data from the FORWARD II platinum-sensitive triplet cohort evaluating mirvetuximab in combination with carboplatin and bevacizumab in the fall of 2020.
- Continue enrollment with IMGN632 monotherapy in relapsed AML, ALL, BPDCN, and MRD+ AML expansion cohorts and in combinations in AML.
- Present IMGN632 BPDCN and AML combination and MRD+ monotherapy data at ASH in December.
- File IND for IMGC936 in the first half of 2020.
- Transition next generation anti-folate receptor alpha (FRα) ADC, IMGN151, to pre-clinical development in mid-2020.
ImmunoGen Reports Recent Progress and 2019 Financial Results | wallstreet-online.de - Vollständiger Artikel unter:
https://www.wallstreet-online.de/nachricht/12171103-immunoge…
Lohnt sich ein Einstieg bei der Pipeline?
ich würde sagen: JA allerdings sollte man permanent beobachten (können) !!
Mal eine Frage an die Profis:
Im Finanzreport vom Febr. stand:
ANTICIPATED 2020 EVENTS
- Initiate pivotal SORAYA trial in the first quarter of 2020 and continue enrollment in the confirmatory Phase 3 MIRASOL trial.
- Open an additional platinum-sensitive investigator sponsored trial evaluating mirvetuximab in combination with carboplatin.
- Present initial data from the Phase 1b FORWARD II platinum-agnostic doublet cohort evaluating mirvetuximab in combination with Avastin (bevacizumab) in mid-2020 and updated data from the FORWARD II platinum-sensitive triplet cohort evaluating mirvetuximab in combination with carboplatin and bevacizumab in the fall of 2020.
- Continue enrollment with IMGN632 monotherapy in relapsed AML, ALL, BPDCN, and MRD+ AML expansion cohorts and in combinations in AML.
- Present IMGN632 BPDCN and AML combination and MRD+ monotherapy data at ASH in December.
- File IND for IMGC936 in the first half of 2020.
- Transition next generation anti-folate receptor alpha (FRα) ADC, IMGN151, to pre-clinical development in mid-2020.
ImmunoGen Reports Recent Progress and 2019 Financial Results | wallstreet-online.de - Vollständiger Artikel unter:
https://www.wallstreet-online.de/nachricht/12171103-immunoge…
Lohnt sich ein Einstieg bei der Pipeline?
Im Finanzreport vom Febr. stand:
ANTICIPATED 2020 EVENTS
- Initiate pivotal SORAYA trial in the first quarter of 2020 and continue enrollment in the confirmatory Phase 3 MIRASOL trial.
- Open an additional platinum-sensitive investigator sponsored trial evaluating mirvetuximab in combination with carboplatin.
- Present initial data from the Phase 1b FORWARD II platinum-agnostic doublet cohort evaluating mirvetuximab in combination with Avastin (bevacizumab) in mid-2020 and updated data from the FORWARD II platinum-sensitive triplet cohort evaluating mirvetuximab in combination with carboplatin and bevacizumab in the fall of 2020.
- Continue enrollment with IMGN632 monotherapy in relapsed AML, ALL, BPDCN, and MRD+ AML expansion cohorts and in combinations in AML.
- Present IMGN632 BPDCN and AML combination and MRD+ monotherapy data at ASH in December.
- File IND for IMGC936 in the first half of 2020.
- Transition next generation anti-folate receptor alpha (FRα) ADC, IMGN151, to pre-clinical development in mid-2020.
ImmunoGen Reports Recent Progress and 2019 Financial Results | wallstreet-online.de - Vollständiger Artikel unter:
https://www.wallstreet-online.de/nachricht/12171103-immunoge…
Lohnt sich ein Einstieg bei der Pipeline?
Antwort auf Beitrag Nr.: 60.582.072 von Cyberhexe am 16.05.19 09:56:57Der Markt ist entzückt !
Antwort auf Beitrag Nr.: 59.996.485 von Cyberhexe am 01.03.19 13:42:21
...FDA verlangt eine weitere Pivotalstudie!
ImmunoGen Provides Regulatory Update on Mirvetuximab Soravtansine Monotherapy in Ovarian Cancer
May 15, 2019 at 6:30 AM EDT
PDF Version
Conference Call to be Held at 8:00 a.m. ET Today
WALTHAM, Mass.--(BUSINESS WIRE)--May 15, 2019-- ImmunoGen, Inc., (Nasdaq: IMGN), a leader in the expanding field of antibody-drug conjugates (ADCs) for the treatment of cancer, today announced the United States Food and Drug Administration (FDA) has recommended that the Company conduct a new Phase 3 randomized trial to evaluate the safety and efficacy of mirvetuximab soravtansine in patients with high folate receptor alpha (FRα)-positive, platinum-resistant ovarian cancer as part of a Type C meeting held this week.
...FDA verlangt eine weitere Pivotalstudie!
ImmunoGen Provides Regulatory Update on Mirvetuximab Soravtansine Monotherapy in Ovarian Cancer
May 15, 2019 at 6:30 AM EDT
PDF Version
Conference Call to be Held at 8:00 a.m. ET Today
WALTHAM, Mass.--(BUSINESS WIRE)--May 15, 2019-- ImmunoGen, Inc., (Nasdaq: IMGN), a leader in the expanding field of antibody-drug conjugates (ADCs) for the treatment of cancer, today announced the United States Food and Drug Administration (FDA) has recommended that the Company conduct a new Phase 3 randomized trial to evaluate the safety and efficacy of mirvetuximab soravtansine in patients with high folate receptor alpha (FRα)-positive, platinum-resistant ovarian cancer as part of a Type C meeting held this week.
sieht nicht gut aus:
ImmunoGen Announces Top-Line Results from Phase 3 FORWARD I Study of Mirvetuximab Soravtansine in Ovarian Cancer
Trial Did Not Meet Primary Endpoint of Progression-Free Survival
Efficacy Signal Seen in High Folate Receptor Alpha Patients; Additional Analyses to be Conducted
Favorable Tolerability Profile Confirmed
Combination Regimens to be Evaluated as an Independent Path Forward to Support Registration in Ovarian Cancer
Conference Call to be Held at 8 a.m. ET
ImmunoGen Announces Top-Line Results from Phase 3 FORWARD I Study of Mirvetuximab Soravtansine in Ovarian Cancer
Trial Did Not Meet Primary Endpoint of Progression-Free Survival
Efficacy Signal Seen in High Folate Receptor Alpha Patients; Additional Analyses to be Conducted
Favorable Tolerability Profile Confirmed
Combination Regimens to be Evaluated as an Independent Path Forward to Support Registration in Ovarian Cancer
Conference Call to be Held at 8 a.m. ET
Marker Therapeutics Exits ASH An Optimist. Should That Be Enough For You?
https://seekingalpha.com/article/4227821-marker-therapeutics…
https://seekingalpha.com/article/4227821-marker-therapeutics…
Joschka???
30.11.23 · dpa-AFX · Ford Motor |
30.11.23 · wallstreetONLINE NewsUpdate · Immunogen |
30.11.23 · dpa-AFX · Ford Motor |
30.11.23 · dpa-AFX · Ford Motor |



















