Rohstoffe, Angebot/Nachfrage - 500 Beiträge pro Seite (Seite 5)
eröffnet am 11.09.14 08:32:17 von
neuester Beitrag 02.11.19 22:08:46 von
neuester Beitrag 02.11.19 22:08:46 von
Beiträge: 2.052
ID: 1.198.959
ID: 1.198.959
Aufrufe heute: 2
Gesamt: 67.820
Gesamt: 67.820
Aktive User: 0
Top-Diskussionen
| Titel | letzter Beitrag | Aufrufe |
|---|---|---|
| vor 1 Stunde | 7311 | |
| vor 36 Minuten | 6116 | |
| heute 19:29 | 5490 | |
| vor 1 Stunde | 4840 | |
| vor 15 Minuten | 4830 | |
| vor 42 Minuten | 3429 | |
| vor 15 Minuten | 2395 | |
| vor 14 Minuten | 1933 |
Meistdiskutierte Wertpapiere
| Platz | vorher | Wertpapier | Kurs | Perf. % | Anzahl | ||
|---|---|---|---|---|---|---|---|
| 1. | 1. | 18.767,00 | +0,17 | 131 | |||
| 2. | 3. | 0,2170 | +3,33 | 125 | |||
| 3. | Neu! | 9,0700 | +60,82 | 108 | |||
| 4. | 4. | 168,12 | -2,24 | 103 | |||
| 5. | 14. | 5,6750 | -3,62 | 56 | |||
| 6. | 2. | 0,2980 | -3,87 | 50 | |||
| 7. | 5. | 2,5800 | -6,18 | 49 | |||
| 8. | 7. | 6,7880 | +1,97 | 38 |
Antwort auf Beitrag Nr.: 59.848.129 von Popeye82 am 12.02.19 01:23:44
http://www.mining-journal.com/leadership/news/1356320/kefi-t…

http://www.mining-journal.com/leadership/news/1356320/kefi-t…
Antwort auf Beitrag Nr.: 58.741.459 von Popeye82 am 19.09.18 18:57:40
http://www.mining-journal.com/copper-news/news/1356686/china…
http://www.mining-journal.com/copper-news/news/1356686/china…
Antwort auf Beitrag Nr.: 59.901.725 von Popeye82 am 18.02.19 16:34:06
http://stockhead.com.au/resources/the-world-is-now-running-o…

http://stockhead.com.au/resources/the-world-is-now-running-o…
Antwort auf Beitrag Nr.: 59.746.851 von Popeye82 am 30.01.19 16:51:21
http://www.mining-journal.com/profit-amp-loss/news/1356886/g…
http://www.glencore.com/media-and-insights/news/Furthering-o…
http://www.handelsblatt.com/unternehmen/industrie/glencore-d…
http://www.mining-journal.com/profit-amp-loss/news/1356886/g…
http://www.glencore.com/media-and-insights/news/Furthering-o…
http://www.handelsblatt.com/unternehmen/industrie/glencore-d…
Antwort auf Beitrag Nr.: 59.863.552 von Popeye82 am 13.02.19 15:36:11
http://www.mining-journal.com/regulation/news/1357879/brazil…

http://www.mining-journal.com/regulation/news/1357879/brazil…
Antwort auf Beitrag Nr.: 59.919.878 von Popeye82 am 20.02.19 13:17:36
http://energypost.eu/the-race-to-phase-out-coal-a-nation-by-…
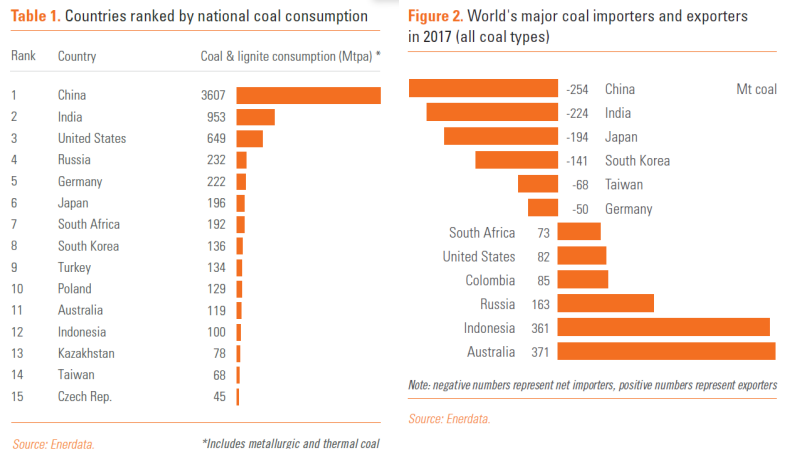
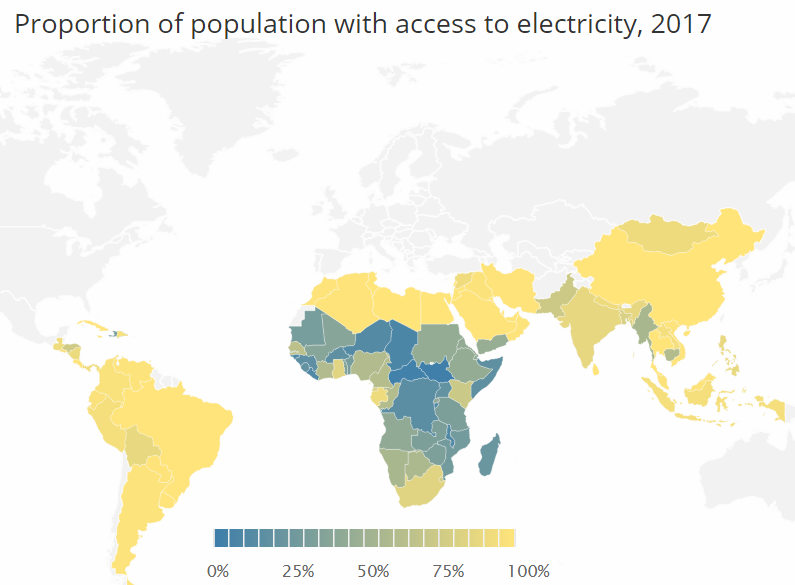
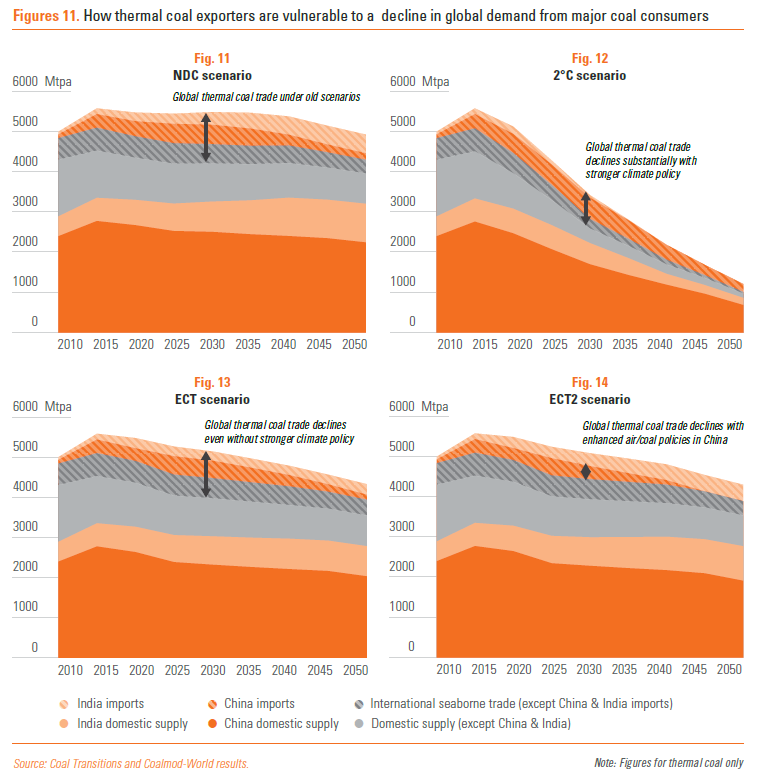
http://energypost.eu/the-race-to-phase-out-coal-a-nation-by-…



Antwort auf Beitrag Nr.: 60.057.032 von Popeye82 am 09.03.19 03:00:42
http://www.jwnenergy.com/article/2019/3/morgan-stanley-sees-…
http://www.jwnenergy.com/article/2019/3/morgan-stanley-sees-…
Antwort auf Beitrag Nr.: 60.115.615 von Popeye82 am 15.03.19 21:05:02
http://www.wallstreet-online.de/nachricht/11364606-kohleauss…
http://www.wallstreet-online.de/nachricht/11364606-kohleauss…
Antwort auf Beitrag Nr.: 60.291.177 von Popeye82 am 05.04.19 19:36:20
http://reneweconomy.com.au/wesfarmers-dumps-coal-and-turns-t…
http://reneweconomy.com.au/wesfarmers-dumps-coal-and-turns-t…
Antwort auf Beitrag Nr.: 60.488.873 von Popeye82 am 05.05.19 06:39:40Artikel "BPH sees early end for Thermal coal, plugs into electric Future" kann man sich auch gut durchlesen.
"Pentagon seeks funds to reduce U.S. Reliance on China's rare earth metals".
"The UK needs twice the current Global cobalt production, to hit its 2050 EV Target. .....and the Rest?".
Reuben Adams.
Stockhead.
Reuben Adams.
Stockhead.
"U.S. outlines critical mineral Strategy; DOC publishes report mandated by Trump executive order".
Shane Lasley.
miningnewsnorth[.com]
Shane Lasley.
miningnewsnorth[.com]
!
Dieser Beitrag wurde von MadMod moderiert. Grund: Spam
"world zinc stocks approach a 12-year low".
Mining Journal.
Mining Journal.
"[Tesla] might get into the mining business".
Jun 13.
Mining Journal.
Jun 13.
Mining Journal.
"LME choses Fastmarkets as Lithium pricing partner"; The London Metal Exchange(LME) hast chosen Pricing reporting Ageny Fastmarkets, as Lithium pricing partner, to promote Market uptake of a transparent, and Representative, global Lithium Price
Jun 13.
Mining Journal.
Jun 13.
Mining Journal.
Facts 'n' Figures: World Copper mine production falls 1.8% in Early 2019, says ICSG".
Jun 13.
John Cumming.
Mining Journal.
Jun 13.
John Cumming.
Mining Journal.
Antwort auf Beitrag Nr.: 60.488.873 von Popeye82 am 05.05.19 06:39:40
http://www.mining.com/trump-gift-to-coal-industry-wont-halt-…
http://www.mining.com/trump-gift-to-coal-industry-wont-halt-…
Antwort auf Beitrag Nr.: 60.923.047 von Popeye82 am 30.06.19 16:04:31
http://www.ferc.gov/legal/staff-reports/2019/apr-energy-infr…
http://www.ferc.gov/legal/staff-reports/2019/apr-energy-infr…
Antwort auf Beitrag Nr.: 60.923.221 von Popeye82 am 30.06.19 16:55:33
http://www.mining.com/chile-goes-carbon-neutral-begins-closi…

http://news.trust.org/item/20190608104230-yomtd/
http://www.mining.com/chile-goes-carbon-neutral-begins-closi…

http://news.trust.org/item/20190608104230-yomtd/
!
Dieser Beitrag wurde von MadMod moderiert. Grund: Spam, Werbung
Neutrino-Voltaic: Strom aus kosmischer Strahlung – Seit Jahren unerwünschte Realität
Elektrischer Strom ist ein essenzieller Bestandteil der modernen Gesellschaft. Trotz über ein Jahrhundert der Forschung gibt es keine unendliche Energiequelle, oder? Strom aus kosmischer Strahlung existiert seit Jahren - nur können Ölkonzerne damit keine Milliardengewinne erwirtschaften.
https://www.epochtimes.de/genial/tech/neutrino-voltaic-strom…
Elektrischer Strom ist ein essenzieller Bestandteil der modernen Gesellschaft. Trotz über ein Jahrhundert der Forschung gibt es keine unendliche Energiequelle, oder? Strom aus kosmischer Strahlung existiert seit Jahren - nur können Ölkonzerne damit keine Milliardengewinne erwirtschaften.
https://www.epochtimes.de/genial/tech/neutrino-voltaic-strom…
Antwort auf Beitrag Nr.: 60.924.967 von Popeye82 am 01.07.19 02:52:52
https://www.mining-journal.com/view-from-the-west-end/opinio…
https://www.mining-journal.com/view-from-the-west-end/opinio…
Beitrag zu dieser Diskussion schreiben
Zu dieser Diskussion können keine Beiträge mehr verfasst werden, da der letzte Beitrag vor mehr als zwei Jahren verfasst wurde und die Diskussion daraufhin archiviert wurde.
Bitte wenden Sie sich an feedback@wallstreet-online.de und erfragen Sie die Reaktivierung der Diskussion oder starten Sie eine neue Diskussion.
Meistdiskutiert
| Wertpapier | Beiträge | |
|---|---|---|
| 139 | ||
| 114 | ||
| 108 | ||
| 104 | ||
| 56 | ||
| 53 | ||
| 51 | ||
| 35 | ||
| 33 | ||
| 32 |
| Wertpapier | Beiträge | |
|---|---|---|
| 28 | ||
| 22 | ||
| 22 | ||
| 22 | ||
| 22 | ||
| 19 | ||
| 19 | ||
| 18 | ||
| 17 | ||
| 15 |