Evotec 566480, wohin geht die Reise??? (Seite 6630)
eröffnet am 12.01.07 11:23:52 von
neuester Beitrag 23.05.24 16:38:46 von
neuester Beitrag 23.05.24 16:38:46 von
Beiträge: 82.262
ID: 1.104.790
ID: 1.104.790
Aufrufe heute: 4.447
Gesamt: 16.300.230
Gesamt: 16.300.230
Aktive User: 1
ISIN: DE0005664809 · WKN: 566480 · Symbol: EVT
9,2300
EUR
-2,69 %
-0,2550 EUR
Letzter Kurs 19:50:13 Tradegate
Neuigkeiten
| TitelBeiträge |
|---|
22.05.24 · wallstreetONLINE Redaktion |
| Evotec Aktien ab 5,80 Euro handeln - Ohne versteckte Kosten!Anzeige |
17:50 Uhr · wO Newsflash |
11:03 Uhr · dpa-AFX Analysen |
Werte aus der Branche Biotechnologie
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,2780 | +53,79 | |
| 1,3560 | +52,87 | |
| 2,1800 | +27,49 | |
| 0,7000 | +22,79 | |
| 10.777,50 | +19,75 |
| Wertpapier | Kurs | Perf. % |
|---|---|---|
| 1,9500 | -15,22 | |
| 2,0500 | -15,29 | |
| 0,8069 | -15,95 | |
| 45,40 | -17,45 | |
| 1,9600 | -17,65 |
Beitrag zu dieser Diskussion schreiben
Noch etwas zu den Gründen der Kinaxo Übernahme, mit der man nicht nur die Onkologie Sparte Evotecs stärken wollte, sondern vielleicht vor allem auf dem Biomarker Segment noch einiges vor hat. Das klingt nach guten Wachstums Chancen.
http://www.genengnews.com/keywordsandtools/print/3/23715/
Analysis & Insight : Jul 25, 2011
Companies with Cancer Therapeutic
Candidates Ramp Up Companion
Diagnostic Efforts
As regulatory framework gets fine-tuned, firms realize the importance of
having a biomarker strategy.
Patricia F. Dimond, Ph.D.
Biopharma companies are more focused on developing companion diagnostics for
cancer drugs despite challenges and initial trepidations over this business model.
Indeed, FDA’s recently issued draft guidance on companion tests is testament to the
fact that the field is advancing and more regulatory clarity is needed.
Therapeutic firms are pursuing two strategies in developing diagnostics: through inhouse
work or through external partnerships with diagnostics companies. Depending
on the availability of a clear-cut biomarker, the ideal is to start diagnostic
development early in the clinical development cycle of a drug. The aim is to have a
test ready for use in Phase III drug testing.
Firms are making the investment in companion diagnostics with the belief that it will
ultimately impact the overall cost of drug development. “While there is some up-front
investment, in the ideal scenario co-development of diagnostics will reduce drug
development costs,” David Reese, M.D., Amgen’s vp of medical sciences, told GEN.
“By allowing you to target much earlier in the drug development process patients
that are likely to respond, you can demonstrate the value of the drug early on.”
Developing Dx and Rx Together
Roche’s recent advances with its metastatic melanoma candidate, vemurafenib
developed through its Genentech subsidiary, and its corresponding companion
diagnostic, the Cobas 4800 BRAF V600 Mutation Test developed through Roche
Diagnostics, provides an example of a successful internal collaboration.
Vemurafenib is being co-developed under a 2006 license and collaboration
agreement between Roche/Genentech and Plexxikon, the original drug developer.
The NDA and MAA for the drug, submitted last May, covers the treatment of
individuals with BRAF V600 mutation-positive metastatic melanoma.
The Cobas 4800 BRAF V600 Mutation Test, a PCR-based companion diagnostic, is
designed to identify people whose tumors carry the BRAF V600 mutation. About
60% of melanomas have this mutation; melanoma cells lacking it are not inhibited by
the drug, which stimulates normal BRAF and can promote tumor growth.
Commenting on the Phase III trial (BRIM3) evaluating the drug, Paul Brown, head of
Roche Molecular Systems, said, “In BRIM3 our investigational test enabled rapid
and accurate identification of eligible patients with metastatic melanoma.” The study
met its co-primary endpoints of overall survival and progression-free survival. Risk of
death was reduced by 63% and risk of disease progression by 74%.
One of the key factors in the successful, side-by-side development of vemurafenib
and its companion diagnostic was that Plexxikon was in the Bay area, Walter Koch,
Ph.D., vp, head of global research at Roche Molecular Diagnostics, told GEN. “Peter
Hirth of Plexxikon had reached out to us in 2005, told us what Plexxikon was
working on, and asked if we could develop a test.
“Our diagnostics agreement predated that of Roche Pharma, and having that
diagnostic relationship in place played a key role in our pharmaceutical colleagues
getting involved in it. Once Roche had decided to in-license and develop the drug, a
lot of planning had started in 2006 to coordinate the development of the drug and
the test and to have the test available for the clinical trials.”
Expected Challenges
Commenting on hurdles with companion diagnostic development for vemurafenib,
Dr. Koch said the first challenge was a “generic one. Drug development and
diagnostic development are fundamentally different, and aligning the processes so
that both the therapeutic and the test can be filed with the FDA simultaneously works
better if you have both teams at the same table together.
“This was greatly facilitated in our case because we were all the same company.”
While he believes it can be done with external partners, Dr. Koch noted that it would
be more complicated.
The second challenge he mentioned was linking hardware, software, and test
reagents. “We were working with a different generation platform in 2005, then had to
change the platform during the development process. We moved to a newer
generation instrument, and so had to revalidate, re-tune, and re-optimize the
reagents so they worked with the new instrument and software.”
Despite inherent challenges, Dr. Koch said that “in Roche and in most pharma
companies, personalized healthcare is the way we are moving forward. We believe
we will greatly improve the efficacy of therapeutics when we can identify appropriate
patients. We are seeing early success rates that suggest that companion diagnostics
are a far better way to improve therapeutics and success rates of clinical trials.”
Having Biomarker Data Early
Vemurafenib stands apart from other cancer drugs with companion tests because
knowledge of specific mutations affecting drug response was available early in the
drug’s development cycle. Dr. Koch explained that while there are multiple examples
of cancer drugs with specific biomarker tests that characterize the diseases,
knowledge of specific mutations affecting drug response became available after the
drugs were developed. “This substantially changes the market,” Dr. Koch said.
Drugs approved along with a companion diagnostic include Eli Lilly’s Erbitux and
Amgen’s Vectibix, both anti-epidermal growth factor receptor (EGFR) antibodies.
The original companion diagnostic, EGFR pharmDx, which detected EGRF status in
patients, was developed by partnering with DAKO.
Evidence that KRAS mutation status would affect how subjects responded to anti-
EGFR treatments became apparent only after clinical trials that formed the basis of
NDA and MAA submissions were completed. Amgen and Imclone analyzed the
existing information and found that about 43% of patients, those with KRAS
mutations, did not respond to the EGRF inhibitors.
“Therefore, if you had a diagnostic test for the KRAS mutations, you would have
40% of patients who wouldn’t get the therapy but who would have been treated prior
to that knowledge.”
But, Dr. Koch said, many drugs are evolving along the lines of BRAF, “where an
activating mutation in an oncogene makes sense to target specifically or knowledge
of a pathway downstream portends failure for the drug and suggests other
development strategies.”
Amgen has bolstered work on identifying biomarkers that will allow the company to
predict which patients will or won’t respond to a given drug. Generally the company
identifies a diagnostic company with appropriate expertise to partner with depending
on the type of test needed.
“We believe it’s an essential part of the drug development process,” Dr. Reese told
GEN. “Every product we put into humans has a biomarker team formed typically one
to two years before the first patient is dosed with a drug.” He explained that the
teams have two mandates. “One is to potentially develop pharmacodynamic
biomarkers that tell you whether the drug is doing what you think it should be doing.
For example, if you are targeting a receptor in cancer, is your drug getting in at an
appropriate level and shutting the receptor off?
“The second mandate of that team is to identify those patients likely to respond or
not respond to the therapy. Our goal is to get the right drug to the right patient as
early in the process as possible, starting that effort even with preclinical work.”
Dr. Reese explained that what potentially differentiates Amgen from companies
developing companion diagnostics for their drug candidates is “the scale of the effort
and the formation of cross-functional teams with the sole mandate of biomarker
development.”
Integrating Companion Dx Development
“Patient selection can take many forms, depending on the clinical scenario and
really has to be customized to each setting,” Dr. Reese remarked. “There is no one
size fits all development process. We have the philosophy that the science and the
biology drive what we do and shape what we do in the clinic.”
Companion diagnostics offer the perfect means of fulfilling the promise of
personalized medicine. They have, however, been viewed as disruptors of the
biopharmaceutical market, as the tests could restrict the size of treatable
populations.
Increasingly, however, pharma and biotech companies are realizing that they cannot
ignore companion diagnostics. With FDA’s draft guidance on this sector suggesting
that drugs and their corresponding tests must be approved simultaneously,
investment in either internal strategies or external partnerships are ramping up.
Grüße
http://www.genengnews.com/keywordsandtools/print/3/23715/
Analysis & Insight : Jul 25, 2011
Companies with Cancer Therapeutic
Candidates Ramp Up Companion
Diagnostic Efforts
As regulatory framework gets fine-tuned, firms realize the importance of
having a biomarker strategy.
Patricia F. Dimond, Ph.D.
Biopharma companies are more focused on developing companion diagnostics for
cancer drugs despite challenges and initial trepidations over this business model.
Indeed, FDA’s recently issued draft guidance on companion tests is testament to the
fact that the field is advancing and more regulatory clarity is needed.
Therapeutic firms are pursuing two strategies in developing diagnostics: through inhouse
work or through external partnerships with diagnostics companies. Depending
on the availability of a clear-cut biomarker, the ideal is to start diagnostic
development early in the clinical development cycle of a drug. The aim is to have a
test ready for use in Phase III drug testing.
Firms are making the investment in companion diagnostics with the belief that it will
ultimately impact the overall cost of drug development. “While there is some up-front
investment, in the ideal scenario co-development of diagnostics will reduce drug
development costs,” David Reese, M.D., Amgen’s vp of medical sciences, told GEN.
“By allowing you to target much earlier in the drug development process patients
that are likely to respond, you can demonstrate the value of the drug early on.”
Developing Dx and Rx Together
Roche’s recent advances with its metastatic melanoma candidate, vemurafenib
developed through its Genentech subsidiary, and its corresponding companion
diagnostic, the Cobas 4800 BRAF V600 Mutation Test developed through Roche
Diagnostics, provides an example of a successful internal collaboration.
Vemurafenib is being co-developed under a 2006 license and collaboration
agreement between Roche/Genentech and Plexxikon, the original drug developer.
The NDA and MAA for the drug, submitted last May, covers the treatment of
individuals with BRAF V600 mutation-positive metastatic melanoma.
The Cobas 4800 BRAF V600 Mutation Test, a PCR-based companion diagnostic, is
designed to identify people whose tumors carry the BRAF V600 mutation. About
60% of melanomas have this mutation; melanoma cells lacking it are not inhibited by
the drug, which stimulates normal BRAF and can promote tumor growth.
Commenting on the Phase III trial (BRIM3) evaluating the drug, Paul Brown, head of
Roche Molecular Systems, said, “In BRIM3 our investigational test enabled rapid
and accurate identification of eligible patients with metastatic melanoma.” The study
met its co-primary endpoints of overall survival and progression-free survival. Risk of
death was reduced by 63% and risk of disease progression by 74%.
One of the key factors in the successful, side-by-side development of vemurafenib
and its companion diagnostic was that Plexxikon was in the Bay area, Walter Koch,
Ph.D., vp, head of global research at Roche Molecular Diagnostics, told GEN. “Peter
Hirth of Plexxikon had reached out to us in 2005, told us what Plexxikon was
working on, and asked if we could develop a test.
“Our diagnostics agreement predated that of Roche Pharma, and having that
diagnostic relationship in place played a key role in our pharmaceutical colleagues
getting involved in it. Once Roche had decided to in-license and develop the drug, a
lot of planning had started in 2006 to coordinate the development of the drug and
the test and to have the test available for the clinical trials.”
Expected Challenges
Commenting on hurdles with companion diagnostic development for vemurafenib,
Dr. Koch said the first challenge was a “generic one. Drug development and
diagnostic development are fundamentally different, and aligning the processes so
that both the therapeutic and the test can be filed with the FDA simultaneously works
better if you have both teams at the same table together.
“This was greatly facilitated in our case because we were all the same company.”
While he believes it can be done with external partners, Dr. Koch noted that it would
be more complicated.
The second challenge he mentioned was linking hardware, software, and test
reagents. “We were working with a different generation platform in 2005, then had to
change the platform during the development process. We moved to a newer
generation instrument, and so had to revalidate, re-tune, and re-optimize the
reagents so they worked with the new instrument and software.”
Despite inherent challenges, Dr. Koch said that “in Roche and in most pharma
companies, personalized healthcare is the way we are moving forward. We believe
we will greatly improve the efficacy of therapeutics when we can identify appropriate
patients. We are seeing early success rates that suggest that companion diagnostics
are a far better way to improve therapeutics and success rates of clinical trials.”
Having Biomarker Data Early
Vemurafenib stands apart from other cancer drugs with companion tests because
knowledge of specific mutations affecting drug response was available early in the
drug’s development cycle. Dr. Koch explained that while there are multiple examples
of cancer drugs with specific biomarker tests that characterize the diseases,
knowledge of specific mutations affecting drug response became available after the
drugs were developed. “This substantially changes the market,” Dr. Koch said.
Drugs approved along with a companion diagnostic include Eli Lilly’s Erbitux and
Amgen’s Vectibix, both anti-epidermal growth factor receptor (EGFR) antibodies.
The original companion diagnostic, EGFR pharmDx, which detected EGRF status in
patients, was developed by partnering with DAKO.
Evidence that KRAS mutation status would affect how subjects responded to anti-
EGFR treatments became apparent only after clinical trials that formed the basis of
NDA and MAA submissions were completed. Amgen and Imclone analyzed the
existing information and found that about 43% of patients, those with KRAS
mutations, did not respond to the EGRF inhibitors.
“Therefore, if you had a diagnostic test for the KRAS mutations, you would have
40% of patients who wouldn’t get the therapy but who would have been treated prior
to that knowledge.”
But, Dr. Koch said, many drugs are evolving along the lines of BRAF, “where an
activating mutation in an oncogene makes sense to target specifically or knowledge
of a pathway downstream portends failure for the drug and suggests other
development strategies.”
Amgen has bolstered work on identifying biomarkers that will allow the company to
predict which patients will or won’t respond to a given drug. Generally the company
identifies a diagnostic company with appropriate expertise to partner with depending
on the type of test needed.
“We believe it’s an essential part of the drug development process,” Dr. Reese told
GEN. “Every product we put into humans has a biomarker team formed typically one
to two years before the first patient is dosed with a drug.” He explained that the
teams have two mandates. “One is to potentially develop pharmacodynamic
biomarkers that tell you whether the drug is doing what you think it should be doing.
For example, if you are targeting a receptor in cancer, is your drug getting in at an
appropriate level and shutting the receptor off?
“The second mandate of that team is to identify those patients likely to respond or
not respond to the therapy. Our goal is to get the right drug to the right patient as
early in the process as possible, starting that effort even with preclinical work.”
Dr. Reese explained that what potentially differentiates Amgen from companies
developing companion diagnostics for their drug candidates is “the scale of the effort
and the formation of cross-functional teams with the sole mandate of biomarker
development.”
Integrating Companion Dx Development
“Patient selection can take many forms, depending on the clinical scenario and
really has to be customized to each setting,” Dr. Reese remarked. “There is no one
size fits all development process. We have the philosophy that the science and the
biology drive what we do and shape what we do in the clinic.”
Companion diagnostics offer the perfect means of fulfilling the promise of
personalized medicine. They have, however, been viewed as disruptors of the
biopharmaceutical market, as the tests could restrict the size of treatable
populations.
Increasingly, however, pharma and biotech companies are realizing that they cannot
ignore companion diagnostics. With FDA’s draft guidance on this sector suggesting
that drugs and their corresponding tests must be approved simultaneously,
investment in either internal strategies or external partnerships are ramping up.
Grüße
Möglicherweise schichtet der ein oder andere auch von EVO zu MOR um. MOR hat in den letzten Tagen relative Stärke aufgebaut, das HJ.-Ergebnis am Freitag müßte aufgrund des extrem starken 1.Q. sehr gut ausfallen. Wäre eine Erklärung.
Heute wird er ganz bewusst unter 3,35€ gedrückt... Hoffe der Gesamtmarkt kommt langsam mal in die Gänge.
Bleibt wie immer spannend bei Evotec.
Bleibt wie immer spannend bei Evotec.
Ja, ist wirklich traurig zu sehen wie der Kurs bei EVO so leicht zu manipulieren ist!
Der 45K Block liegt immer noch im Orderbuch. Wird Zeit das den mal jemand wegkauft 

Kursverlauf vom 21.03.2008 bis 22.07.2011 (log. Kerzendarstellung / 1 Kerze = 1 Woche)
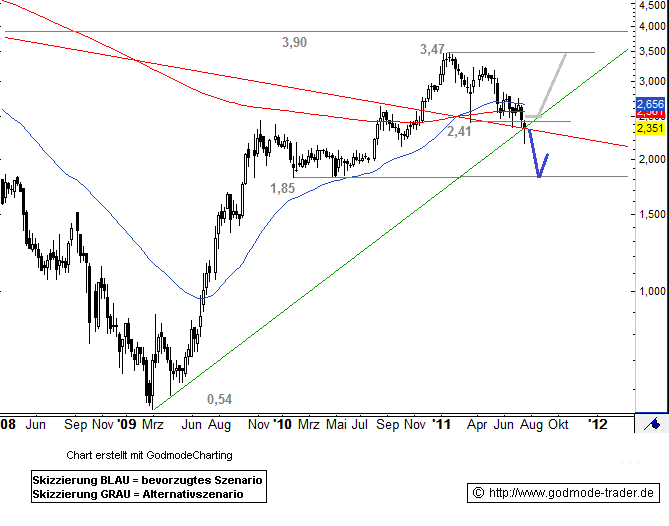

22.07.2011
EVOTEC - Bullen wehren sich, aber ...
Börse: Xetra in Euro / Kursstand: 2,35 Euro
Rückblick: Die Aktie Evotec erreichte im Januar 2011 ein Hoch bei 3,47 Euro. Damit endete bisher eine langfristige Rally, die im März 2009 nach einem Tief 0,54 Euro eingesetzt hatte. Anschließend fiel der Wert auf 2,41 Euro zurück. Damit kam es zu einem Pullback an den gebrochenen Abwärtstrend ab September 2003. In dieser Woche fällt die Aktie allerdings unter 2,41 Euro und damit auch in den Abwärtstrend wieder zurück. Allerding bildet der Wert scheinbar eine bullische Wochenkerze aus.
Charttechnischer Ausblick: Gelingt der Aktie von Evotec eine dynamische Rückkehr über 2,46 Euro auf Wochenschlusskursbasis, dann wäre eine Rally in Richtung 3,47 Euro zu erwarten.
Sollte sich der Wert allerdings unter 2,47 Euro etablieren, wäre eine Abwärtsbewegung in Richtung 1,85 Euro zu erwarten.
EVOTEC - Bullen wehren sich, aber ...
Börse: Xetra in Euro / Kursstand: 2,35 Euro
Rückblick: Die Aktie Evotec erreichte im Januar 2011 ein Hoch bei 3,47 Euro. Damit endete bisher eine langfristige Rally, die im März 2009 nach einem Tief 0,54 Euro eingesetzt hatte. Anschließend fiel der Wert auf 2,41 Euro zurück. Damit kam es zu einem Pullback an den gebrochenen Abwärtstrend ab September 2003. In dieser Woche fällt die Aktie allerdings unter 2,41 Euro und damit auch in den Abwärtstrend wieder zurück. Allerding bildet der Wert scheinbar eine bullische Wochenkerze aus.
Charttechnischer Ausblick: Gelingt der Aktie von Evotec eine dynamische Rückkehr über 2,46 Euro auf Wochenschlusskursbasis, dann wäre eine Rally in Richtung 3,47 Euro zu erwarten.
Sollte sich der Wert allerdings unter 2,47 Euro etablieren, wäre eine Abwärtsbewegung in Richtung 1,85 Euro zu erwarten.
Schlusskurs über 2,41€ wäre fein gewesen... wird wohl nichts.
Zitat von depulep: nur noch 13 hgandelstage bis zu den zahlen
wenn ubs fonds die aktie nennen und im blick haben braucht mann sich wegen der kursprügel nicht zu wundern
das nach den zahlen neue Höchstände ereicht werden ist relativ unwahrscheinlich
ich hoffe nur das die überteibung nach unten auch mal nach oben geht
die frage is nicht ob sondern wann
Aus charttechnischer Sicht wurde immerhin schon mal der Widerstand, der bei 2,35 € lag durchbrochen. Kurse über 2,40 und mehr sind jetzt gut möglich.
http://www.4investors.de/php_fe/index.php?sektion=stock&ID=4…
warum sollten nach den Zahlen keine neuen Höchststände möglich sein?
17:50 Uhr · wO Newsflash · Carl Zeiss Meditec |
05:10 Uhr · kapitalerhoehungen.de · Evotec |
22.05.24 · dpa-AFX · Analog Devices |
22.05.24 · dpa-AFX · Analog Devices |
22.05.24 · dpa-AFX · Evotec |
22.05.24 · dpa-AFX · Evotec |
22.05.24 · dpa-AFX · BMW |
22.05.24 · dpa-AFX · Evotec |


























