ALS - Biotech machts möglich? - 500 Beiträge pro Seite
eröffnet am 13.12.10 17:23:26 von
neuester Beitrag 06.01.11 12:48:19 von
neuester Beitrag 06.01.11 12:48:19 von
Beiträge: 9
ID: 1.161.954
ID: 1.161.954
Aufrufe heute: 0
Gesamt: 346
Gesamt: 346
Aktive User: 0
Top-Diskussionen
| Titel | letzter Beitrag | Aufrufe |
|---|---|---|
| vor 1 Stunde | 2399 | |
| vor 47 Minuten | 1955 | |
| vor 1 Stunde | 1827 | |
| 08.05.24, 11:56 | 1818 | |
| vor 48 Minuten | 1333 | |
| vor 51 Minuten | 1282 | |
| vor 1 Stunde | 1085 | |
| gestern 17:20 | 853 |
Meistdiskutierte Wertpapiere
| Platz | vorher | Wertpapier | Kurs | Perf. % | Anzahl | ||
|---|---|---|---|---|---|---|---|
| 1. | 1. | 18.772,85 | +0,46 | 131 | |||
| 2. | 3. | 0,2170 | +3,33 | 125 | |||
| 3. | Neu! | 8,2570 | +96,67 | 108 | |||
| 4. | 4. | 156,46 | -2,31 | 103 | |||
| 5. | 14. | 5,7540 | -2,18 | 56 | |||
| 6. | 2. | 0,2980 | -3,87 | 50 | |||
| 7. | 5. | 2,3720 | -7,54 | 49 | |||
| 8. | 7. | 6,8000 | +2,38 | 38 |
Cytokinetics, Incorporated (Nasdaq: CYTK) gab heute den erfolgreichen Abschluss seiner Phase-IIa - Studie:
"Evidence of Effect" (EOE) klinische Studie mit CK-2017357 bei Patienten mit Amyotrophe Lateralsklerose (ALS) bekant, auch als Lou-Gehrig-Krankheit genannt, während einer mündlichen Präsentation, die in der Clinical Trials Session auf dem 21. International Symposium on ALS / MND in Orlando, Florida.
http://www.cytokinetics.com/press_releases/release/pr_129219…
South San Francisco, CA - December 13, 2010
Cytokinetics, Incorporated (Nasdaq:CYTK) announced the successful completion of its Phase IIa “Evidence of Effect” (EoE) clinical trial of CK-2017357 in patients with amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig’s disease, during an oral presentation included in the Clinical Trials Session at the 21st International Symposium on ALS/MND in Orlando, Florida.
A presentation titled, “A Phase 2A, Double-Blind, Randomized, Placebo-Controlled, Single-dose, Crossover Study of the Selective Fast Skeletal Muscle Troponin Activator, CK-2017357, in Patients with ALS” was made by Jeremy M. Shefner, MD, PhD, Professor and Chair of the Department of Neurology at the Upstate Medical University at the State University of New York. CK-2017357 is the lead drug candidate from the company's skeletal muscle contractility program.
“We are pleased that this clinical trial has successfully generated results in patients with ALS that are both encouraging and consistent with data generated in our preclinical studies and our Phase I clinical trials with CK-2017357. Importantly, this clinical trial has now generated clinically relevant hypotheses that warrant further exploration in proof-of-concept clinical trials in patients with ALS,” stated Andrew A. Wolff, MD, FACC, Cytokinetics’ Senior Vice President of Clinical Research and Development and Chief Medical Officer. “We are now planning to proceed into a multiple-dose clinical trial to understand how longer-term dosing of CK-2017357 may impact the functional status of these patients.”
“This study suggests that CK-2017357 may play a significant role in the treatment of patients with ALS,” stated Dr. Shefner. “We assessed multiple variables associated with muscle function in these patients. Based on these encouraging results, I believe that CK-2017357 may offer a new way of thinking about symptom relief and quality of life for these patients. These results support further study of CK-2017357 to evaluate the potential for sustained functional benefit in patients with ALS.”
"Evidence of Effect" (EOE) klinische Studie mit CK-2017357 bei Patienten mit Amyotrophe Lateralsklerose (ALS) bekant, auch als Lou-Gehrig-Krankheit genannt, während einer mündlichen Präsentation, die in der Clinical Trials Session auf dem 21. International Symposium on ALS / MND in Orlando, Florida.
http://www.cytokinetics.com/press_releases/release/pr_129219…
South San Francisco, CA - December 13, 2010
Cytokinetics, Incorporated (Nasdaq:CYTK) announced the successful completion of its Phase IIa “Evidence of Effect” (EoE) clinical trial of CK-2017357 in patients with amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig’s disease, during an oral presentation included in the Clinical Trials Session at the 21st International Symposium on ALS/MND in Orlando, Florida.
A presentation titled, “A Phase 2A, Double-Blind, Randomized, Placebo-Controlled, Single-dose, Crossover Study of the Selective Fast Skeletal Muscle Troponin Activator, CK-2017357, in Patients with ALS” was made by Jeremy M. Shefner, MD, PhD, Professor and Chair of the Department of Neurology at the Upstate Medical University at the State University of New York. CK-2017357 is the lead drug candidate from the company's skeletal muscle contractility program.
“We are pleased that this clinical trial has successfully generated results in patients with ALS that are both encouraging and consistent with data generated in our preclinical studies and our Phase I clinical trials with CK-2017357. Importantly, this clinical trial has now generated clinically relevant hypotheses that warrant further exploration in proof-of-concept clinical trials in patients with ALS,” stated Andrew A. Wolff, MD, FACC, Cytokinetics’ Senior Vice President of Clinical Research and Development and Chief Medical Officer. “We are now planning to proceed into a multiple-dose clinical trial to understand how longer-term dosing of CK-2017357 may impact the functional status of these patients.”
“This study suggests that CK-2017357 may play a significant role in the treatment of patients with ALS,” stated Dr. Shefner. “We assessed multiple variables associated with muscle function in these patients. Based on these encouraging results, I believe that CK-2017357 may offer a new way of thinking about symptom relief and quality of life for these patients. These results support further study of CK-2017357 to evaluate the potential for sustained functional benefit in patients with ALS.”
http://www.streetinsider.com/Analyst+Comments/Canaccord+Genu…
Kursziel : 6$
Canaccord Genuity maintains a 'Buy' on Cytokinetics (NASDAQ: CYTK), PT $6.
Canaccord analyst says, "CK357 Phase 2 ALS efficacy data shows promise of real clinical benefit in ALS. We think CK357 has a strong mechanistic rationale in ALS and muscle-wasting diseases, and very promising new Phase 2 efficacy data. We think CYTK’s partner Amgen will continue to advance Phase 2 omecamtiv in heart failure trials in 2011. Our $6 price target is based on a pNPV analysis."
For more ratings news on Cytokinetics click here and for the rating history of Cytokinetics click here.
Shares of Cytokinetics closed at $2.93 yesterday, with a 52 week range of $2.03-$3.80.
Kursziel : 6$
Canaccord Genuity maintains a 'Buy' on Cytokinetics (NASDAQ: CYTK), PT $6.
Canaccord analyst says, "CK357 Phase 2 ALS efficacy data shows promise of real clinical benefit in ALS. We think CK357 has a strong mechanistic rationale in ALS and muscle-wasting diseases, and very promising new Phase 2 efficacy data. We think CYTK’s partner Amgen will continue to advance Phase 2 omecamtiv in heart failure trials in 2011. Our $6 price target is based on a pNPV analysis."
For more ratings news on Cytokinetics click here and for the rating history of Cytokinetics click here.
Shares of Cytokinetics closed at $2.93 yesterday, with a 52 week range of $2.03-$3.80.
Was für ein grässlicher Umsatz (...und Kurs) nach diesen News hier in Old-Germany:

in New-York sieht es kurstechnisch aber auch nicht wesentlich besser aus...
Hier stimmt aber zumindestens das Interesse:
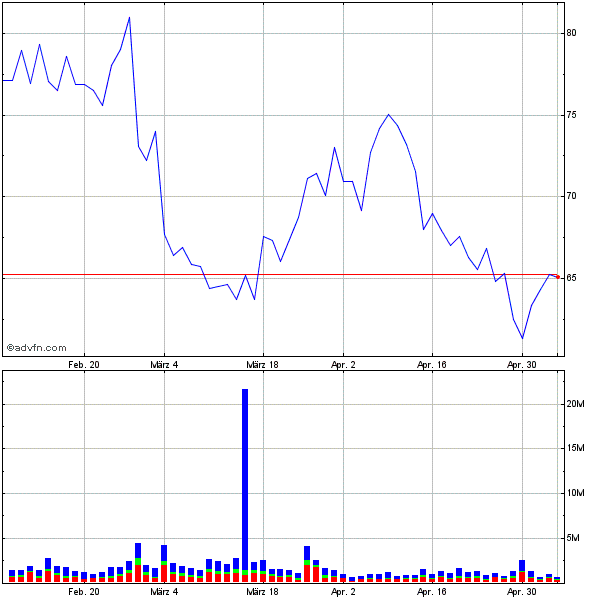
in New-York sieht es kurstechnisch aber auch nicht wesentlich besser aus...
Hier stimmt aber zumindestens das Interesse:
.... ich glaube wir werden ne ganze Schüssel Geduld mitbringen müssen ...
Die 2,- $ Barriere hält (noch) .... und hoffe, die wird noch ne harte Nuss ...
Ich wünsche Euch allen ein erfolgreiches- und gesundes Raketen-Jahr 2011 mit Cyto.

Die 2,- $ Barriere hält (noch) .... und hoffe, die wird noch ne harte Nuss ...
Ich wünsche Euch allen ein erfolgreiches- und gesundes Raketen-Jahr 2011 mit Cyto.

allen Investierten eine gesundes neues Jahr und ein "glückliche Händchen", welches man auch mal braucht ..... 
Zum Anfang des Jahres gleich mal eine Analyse von Equities Research, mit dem Votum "Halten":
.... aber ich glaube , die hat auch schon Moos vom letzten Jahr angesetzt...
http://www.americanbankingnews.com/2011/01/01/cytokinetics-i…

Zum Anfang des Jahres gleich mal eine Analyse von Equities Research, mit dem Votum "Halten":
.... aber ich glaube , die hat auch schon Moos vom letzten Jahr angesetzt...

http://www.americanbankingnews.com/2011/01/01/cytokinetics-i…
Auch das nächste Votum steht auf "Halten":
http://www.americanconsumernews.com/2011/01/zacks-investment…
Zacks Investment Research Analysts Give Cytokinetics, Inc. (NASDAQ: CYTK) a “Neutral” Rating
January 3rd, 2011 • Related • Filed Under • by ACN Staff
Filed Under: Stock Market News
Zacks Investment Research research analysts reiterated a “neutral” rating for shares of Cytokinetics, Inc. (NASDAQ: CYTK) in a research note issued to investors on Tuesday.
Cytokinetics, Inc. (NASDAQ: CYTK)’s stock opened at 2.09 on Monday. Cytokinetics, Inc. has a 52 week range of $2.01 to $3.78. The stock’s 50-day moving average is $2.25 and its 200-day moving average is $2.42. Analysts predict on average that Cytokinetics, Inc. will post $-0.20 earnings per share next quarter. The company has a market cap of $137.7 million and a P/E (price-to-earnings ratio) of N/A.
http://www.americanconsumernews.com/2011/01/zacks-investment…
Zacks Investment Research Analysts Give Cytokinetics, Inc. (NASDAQ: CYTK) a “Neutral” Rating
January 3rd, 2011 • Related • Filed Under • by ACN Staff
Filed Under: Stock Market News
Zacks Investment Research research analysts reiterated a “neutral” rating for shares of Cytokinetics, Inc. (NASDAQ: CYTK) in a research note issued to investors on Tuesday.
Cytokinetics, Inc. (NASDAQ: CYTK)’s stock opened at 2.09 on Monday. Cytokinetics, Inc. has a 52 week range of $2.01 to $3.78. The stock’s 50-day moving average is $2.25 and its 200-day moving average is $2.42. Analysts predict on average that Cytokinetics, Inc. will post $-0.20 earnings per share next quarter. The company has a market cap of $137.7 million and a P/E (price-to-earnings ratio) of N/A.
Ankündigung des Beginns der Phase IIa - Studie von CK-2017357
"....Tuesday said it opened enrollment in a Phase IIa "Evidence of Effect" or EoE clinical trial of CK-2017357 in patients with generalized myasthenia gravis or MG.
CK-2017357 is a fast skeletal muscle troponin activator and is the lead drug candidate that has emerged from the company's skeletal muscle contractility program. ..."
http://www.rttnews.com/Content/QuickFacts.aspx?Id=1518786&SM…
"....Tuesday said it opened enrollment in a Phase IIa "Evidence of Effect" or EoE clinical trial of CK-2017357 in patients with generalized myasthenia gravis or MG.
CK-2017357 is a fast skeletal muscle troponin activator and is the lead drug candidate that has emerged from the company's skeletal muscle contractility program. ..."
http://www.rttnews.com/Content/QuickFacts.aspx?Id=1518786&SM…
23.148 Optionsscheine (min 50.000,-$) mit einem Wandelpreis von 2,16 $ / Aktie
für den 57 Jahre alten Direktor von CYTK Herrn Heidrich A. Grant zum 01.03.2011
(so eine kurzfristige Option sieht man eigentlich selten .... )
Der Preis des Werpapiers für Herrn Grant liegt bei 0,864 $!
http://xml.10kwizard.com/filing_raw.php?repo=tenk&ipage=7325…
Ob das ein "Dankeschön" - Boni für den Beginn der Phase 2a - ALS Studie sein soll?
für den 57 Jahre alten Direktor von CYTK Herrn Heidrich A. Grant zum 01.03.2011
(so eine kurzfristige Option sieht man eigentlich selten .... )
Der Preis des Werpapiers für Herrn Grant liegt bei 0,864 $!
http://xml.10kwizard.com/filing_raw.php?repo=tenk&ipage=7325…
Ob das ein "Dankeschön" - Boni für den Beginn der Phase 2a - ALS Studie sein soll?
Beitrag zu dieser Diskussion schreiben
Zu dieser Diskussion können keine Beiträge mehr verfasst werden, da der letzte Beitrag vor mehr als zwei Jahren verfasst wurde und die Diskussion daraufhin archiviert wurde.
Bitte wenden Sie sich an feedback@wallstreet-online.de und erfragen Sie die Reaktivierung der Diskussion oder starten Sie eine neue Diskussion.
Meistdiskutiert
| Wertpapier | Beiträge | |
|---|---|---|
| 100 | ||
| 75 | ||
| 69 | ||
| 47 | ||
| 47 | ||
| 42 | ||
| 36 | ||
| 26 | ||
| 18 | ||
| 17 |





















